Tabla-Conversión De Unidades
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Pressure, Its Units of Measure and Pressure References
_______________ White Paper Pressure, Its Units of Measure and Pressure References Viatran Phone: 1‐716‐629‐3800 3829 Forest Parkway Fax: 1‐716‐693‐9162 Suite 500 [email protected] Wheatfield, NY 14120 www.viatran.com This technical note is a summary reference on the nature of pressure, some common units of measure and pressure references. Read this and you won’t have to wait for the movie! PRESSURE Gas and liquid molecules are in constant, random motion called “Brownian” motion. The average speed of these molecules increases with increasing temperature. When a gas or liquid molecule collides with a surface, momentum is imparted into the surface. If the molecule is heavy or moving fast, more momentum is imparted. All of the collisions that occur over a given area combine to result in a force. The force per unit area defines the pressure of the gas or liquid. If we add more gas or liquid to a constant volume, then the number of collisions must increase, and therefore pressure must increase. If the gas inside the chamber is heated, the gas molecules will speed up, impact with more momentum and pressure increases. Pressure and temperature therefore are related (see table at right). The lowest pressure possible in nature occurs when there are no molecules at all. At this point, no collisions exist. This condition is known as a pure vacuum, or the absence of all matter. It is also possible to cool a liquid or gas until all molecular motion ceases. This extremely cold temperature is called “absolute zero”, which is -459.4° F. -
Unit 1 an Introduction to Fluids
UNIT 1 AN INTRODUCTION TO FLUIDS If the rock is not too heavy, you might just _ it up and carry it. When a person moves a rock, the energy to move the rock is suppl ied by the 6. When a rock fall s from a cliff, then it is gravity that is supplying the _ For a fluid to flow, there (must/need not) be a source of energy. 8. When a pump is forcing fluids through a well-bore, the source of energy for flow is the _ 9. The source of energy for a flowing well is re servoir pressure. A flowing well produces because of the of the fluids in the reservoir. 10. Any substance that can flow and that has no definite shape is a fluid. The oil in this tank (has a definite shape/assumes the shape of the tank), Gas (flows/does not flow) and has (a definite/ an indefinite) shape. 12. Anything that flows and has an indefinite shape is a (liquid/ gas/fluid), 15. The oil and gas found in a reservoir are made up of hydrogen atoms and carbon atoms. Hydrocarbons are substances made up only of atoms of ______ and atoms of _ 17. Water is made up of hydrogen atoms and oxygen atoms (H2O)· 18. Different oils and petroleum gases are made up of different combinations of hydrogen and carbon atoms. 21. A substance, water for example, can exist as a gas, a liquid, or a solid. We can change the liquid water to a gas by adding even more 23 Molecules have attractive forces which hold them together. -

Metric System Conversion Factors1 J
AGR39 Metric System Conversion Factors1 J. Bryan Unruh, Barry J. Brecke, and Ramon G. Leon-Gonzalez2 Area Equivalents 1 Hectare (ha) 2 1 Acre (A) = 10,000 square meters (m ) 2 = 100 are (a) = 43,560 square feet (ft ) = 2.471 acres (A) = 4,840 square yards (yd2) = 0.405 hectares (ha) 1 Square Foot (ft) = 160 square rods (rd2) 2 = 4,047 square meters (m2) = 144 square inches (in ) = 929.03 square centimeters (cm2) 2 1 Acre-inch (ac-in) = 0.0929 square meters (m ) 3 = 102.8 cubic meters (m ) 1 Square Mile (mi) = 27,154 gallons, US (gal) 2 = 3,630 cubic feet (ft3) = 27,878,400 square feet (ft ) = 3,097,600 square yards (yd2) 2 1 Are (a) = 640 square acres (A ) = 2,589,988.11 square meters (m2) = 100 square meters (m2) 2 = 119.6 square yards (yd ) 1 Square Rod (rd) = 0.025 acre (A) = 39,204 square inches (in2) = 272.25 square feet (ft2) 1 Cubic Foot (ft) 2 3 = 30.25 square yards (yds ) = 1,728 cubic inches (in ) = 25.3 square meters (m2) = 0.037 cubic yards (yds3) 3 = 0.02832 cubic meters (cm ) 1 Square Yard (yd) = 28,320 cubic centimeters (cm3) = 9 square feet (ft2) 2 1 Cubic Yard (yd) = 0.836 square meters (m ) = 27 cubic feet (ft3) = 0.764 cubic meters (m3) 1. This document is AGR39, one of a series of the Environmental Horticulture Department, UF/IFAS Extension. Original publication date November 1993. Revised December 2014. Reviewed December 2017. Visit the EDIS website at http://edis.ifas.ufl.edu. -

Manual of Style
Manual Of Style 1 MANUAL OF STYLE TABLE OF CONTENTS CHAPTER 1 GENERAL PROVISIONS ...............3 101.0 Scope .............................................. 3 102.0 Codes and Standards..................... 3 103.0 Code Division.................................. 3 104.0 Table of Contents ........................... 4 CHAPTER 2 ADMINISTRATION .........................6 201.0 Administration ................................. 6 202.0 Chapter 2 Definitions ...................... 6 203.0 Referenced Standards Table ......... 6 204.0 Individual Chapter Administrative Text ......................... 6 205.0 Appendices ..................................... 7 206.0 Installation Standards ..................... 7 207.0 Extract Guidelines .......................... 7 208.0 Index ............................................... 9 CHAPTER 3 TECHNICAL STYLE .................... 10 301.0 Technical Style ............................. 10 302.0 Technical Rules ............................ 10 Table 302.3 Possible Unenforceable and Vague Terms ................................ 10 303.0 Health and Safety ........................ 11 304.0 Rules for Mandatory Documents .................................... 11 305.0 Writing Mandatory Requirements ............................... 12 Table 305.0 Typical Mandatory Terms............. 12 CHAPTER 4 EDITORIAL STYLE ..................... 14 401.0 Editorial Style ................................ 14 402.0 Definitions ...................................... 14 403.0 Units of Measure ............................ 15 404.0 Punctuation -

Fundamentals of Hydraulics: Pressure
PUBLISHED BY THE NATIONAL ENVIRONMENTAL SERVICES CENTER FundamentalsBy Zane Satterfield, of P.Hydraulics: E., NESC Engineering Scientist Pressure Summary Hydraulics is the branch of engineering that focuses on the practical problems of collecting, storing, measuring, transporting, controlling, and using water and other liquids. This Tech Brief is the first of two that will discuss some fundamental hydraulic problems and will focus primarily on pressure. The second will discuss flow. Why is understanding hydraulics important? cubic foot (62.4 lb/ft3); so, one cubic foot (1ft x 1ft x 1ft) of water weighs 62.4 lbs. There are 7.48 gallons in The science of hydraulics is as old as civilization itself. a cubic foot, with each gallon weighing approximately For centuries, engineers have succeeded in making water 8.34 pounds, so 7.48 gal x 8.34 lbs/gal = 62.4 pounds. flow from one place to another with as few hitches as possible. When problems do occur, they are usually In the metric system, the term for weight (force due related to the hydraulics involved in pipe flow. to gravity) is called a Newton (N), and the unit weight of water is 9,800 Newtons per cubic meter (9,800 Liquids in motion produce forces and pressure whenever N/m3). More appropriately, this is expressed as 9.8 the velocity, flow direction, or elevation changes. kilonewtons per cubic meter (9.8 kN/m3), where the Knowing pipe pressure and flow at certain points along prefix kilo stands for 1,000. the pipe’s path can help determine pipe size and capac- ity. -

The International System of Units (SI) - Conversion Factors For
NIST Special Publication 1038 The International System of Units (SI) – Conversion Factors for General Use Kenneth Butcher Linda Crown Elizabeth J. Gentry Weights and Measures Division Technology Services NIST Special Publication 1038 The International System of Units (SI) - Conversion Factors for General Use Editors: Kenneth S. Butcher Linda D. Crown Elizabeth J. Gentry Weights and Measures Division Carol Hockert, Chief Weights and Measures Division Technology Services National Institute of Standards and Technology May 2006 U.S. Department of Commerce Carlo M. Gutierrez, Secretary Technology Administration Robert Cresanti, Under Secretary of Commerce for Technology National Institute of Standards and Technology William Jeffrey, Director Certain commercial entities, equipment, or materials may be identified in this document in order to describe an experimental procedure or concept adequately. Such identification is not intended to imply recommendation or endorsement by the National Institute of Standards and Technology, nor is it intended to imply that the entities, materials, or equipment are necessarily the best available for the purpose. National Institute of Standards and Technology Special Publications 1038 Natl. Inst. Stand. Technol. Spec. Pub. 1038, 24 pages (May 2006) Available through NIST Weights and Measures Division STOP 2600 Gaithersburg, MD 20899-2600 Phone: (301) 975-4004 — Fax: (301) 926-0647 Internet: www.nist.gov/owm or www.nist.gov/metric TABLE OF CONTENTS FOREWORD.................................................................................................................................................................v -
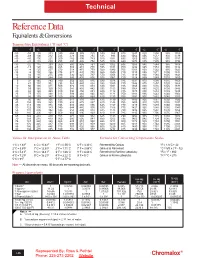
Reference Data Equivalents & Conversions
Technical Reference Data Equivalents & Conversions Temperature Equivalents ( °F and °C) °C °F °C °F °C °F °C °F °C °F °C °F °C °F °C °F -50 -58 95 203 240 464 385 725 530 986 675 1247 820 1508 965 1769 -45 -49 100 212 245 473 390 734 535 995 680 1256 825 1517 970 1778 -40 -40 105 221 250 482 395 743 540 1004 685 1265 830 1526 975 1787 -35 -31 110 230 255 491 400 752 545 1013 690 1274 835 1535 980 1796 -30 -22 115 239 260 500 405 761 550 1022 695 1283 840 1544 985 1805 -25 -13 120 248 265 509 410 770 555 1031 700 1292 845 1553 990 1814 -20 -4 125 257 270 518 415 779 560 1040 705 1301 850 1562 995 1823 -15 -5 130 266 275 527 420 788 565 1049 710 1310 855 1571 1000 1832 -10 14 135 275 280 536 425 797 570 1058 715 1319 860 1580 1005 1841 -5 23 140 284 285 545 430 806 575 1067 720 1328 865 1589 1010 1850 0 32 145 293 290 554 435 815 580 1076 725 1337 870 1598 1015 1859 5 41 150 302 295 563 440 824 585 1085 730 1346 875 1607 1020 1868 10 50 155 311 300 572 445 833 590 1094 735 1355 880 1616 1025 1877 15 59 160 320 305 581 450 842 595 1103 740 1364 885 1625 1030 1886 20 68 165 329 310 590 455 851 600 1112 745 1373 890 1634 1035 1895 25 77 170 338 315 599 460 860 605 1121 750 1382 895 1643 1040 1904 30 86 175 347 320 608 465 869 610 1130 755 1391 900 1652 1045 1913 35 95 180 356 325 617 470 878 615 1139 760 1400 905 1661 1050 1922 40 104 185 365 330 626 475 887 620 1148 765 1409 910 1670 1055 1931 45 113 190 374 335 635 480 896 625 1157 770 1418 915 1679 1060 1940 50 122 195 383 340 644 485 905 630 1166 775 1427 920 1688 1065 1949 55 131 200 -

Pressure Dependence of Senseair´S NDIR Sensors
Pressures Dependence of SenseAir´s NDIR sensors Pressure Dependence of SenseAir´s NDIR sensors Pressure affects the reading of CO 2 sensors. This is because of the physics principle used to measure CO 2 concentration. The NDIR type of sensors measure the IR radiation absorbed by CO 2 molecules, which corresponds to the mole concentration. And when pressure increases, the number of molecules in a given volume also increases linearly. Therefore, all CO 2 sensors have a dependence on pressure. How atmosphere affects the reading of CO 2 Regulation by using ABC function sensors? SenseAir sensors make use of the ABC algorithm to The composition of air is unchanged until an elevation of compensate for any long-term drift due to e.g. aging of approximately 10.000 m, but the atmospheric pressure of components and initial inaccuracy due to e.g. air varies with altitude, that at high altitudes there’s lower transportation. If we located a CO 2 at 2000 m, as figure 2 pressure than at sea level. This means also that the shows, using Senseair’s ABC function can effectively number of molecules per volume unit decreases, adjust the reading close to true reading which minimizes according to the Ideal gas law. the difference generated by pressure sensitivity. Since an NDIR sensor in fact measures the number of molecules per volume unit, it will show a lower reading at higher altitudes. But the unit used by the sensor to express CO 2 concentration is ppm by volume and the volume fraction occupied by CO 2 is the same regardless of pressure, which means that the choice of ppm by volume as unit of measurement for CO 2 sensors introduces a deviation that can be compensated for, if needed. -
TG19 JUNE 2009 Version
MSL Technical Guide 19 Measuring Atmospheric Pressure with a Barometer Barometric Pressure Units Summary The pascal (Pa) is the metric (SI) unit of pressure This technical guide explains how to use a digital ba- and is defined as a force of 1 newton acting over an area 2 rometer to measure atmospheric pressure in a labora- of 1 square metre so 1 Pa = 1 N/m [1]. One pascal is a tory or industrial setting. It starts with a brief discussion small pressure so measurements are usually reported in of the atmosphere and then talks about barometers, their kPa (1000 Pa) or MPa (1000 kPa). stability and calibration requirements, along with how to Barometric pressure measurements are often re- work out the uncertainty of an air pressure measure- ported in hectopascal (hPa). Hecto means times 100 so ment. 1 hPa = 100 Pa. The prefix hecto is used in barometry as the numerical value of the pressure in hPa is the Introduction same as that measured in millibar (mbar), 1 mbar = 1 hPa = 100 Pa. Many historical meteorological A barometer measures the air pressure of the Earth’s records of atmospheric pressure were recorded in mbar. atmosphere. The atmospheric pressure is generated by There are many other barometric pressure units, the Earth’s gravity acting on the mass of air in the at- some of which are shown in the table below, along with mosphere. The forces involved are surprisingly large, a their symbol and relationship to the pascal. surface pressure of 100 kPa means the force at the base 2 of a column of the atmosphere, cross-section area 1 m , is equivalent to a mass of 10,000 kg. -

Using Manometers to Precisely Measure Pressure, Flow and Level
Using Manometers to Precisely Measure Pressure, Flow and Level Precision Measurement Since 1911 ..• A1eriam Instrument l lrl [ aScott Fetzer compaii] Table of Contents Manometer Principles .... ........................................... .... :. 2 Indicating Fluids .. .......................... .. .............. ... .. .......... ... 5 Manometer Corrections ........................................ .......... 6 Digital Manometers ...................................... .. ................. 9 Applications Guide .. ............. ................... .-............... ... .. 12 Glossary of Pressure Terms ....... .. ................................. 16 Pressure Conversions ........................ Inside Back Cover 1 Manometer Principles The manometer, one of the earliest pressure measur ing instruments, when used properly is very accurate. NIST recognizes the U tube manometer as a primary standard due to its inherent accuracy and simplicity of 3 operation. The manometer has no moving parts sub ject to wear, age, or fatigue. Manometers operate on 2 the Hydrostatic Balance Principle: a liquid column of known height will exert a known pressure when the 1 weight per unit volume of the liquid is known. The fundamental relationship for pressure expressed by a 0 liquid column is 1 2 p = differential pressure 3 P 1 = pressure at the low pressure connection P2 = pressure at the high pressure connection p density of the liquid g acceleration of gravity h height of the liquid column Figure 1 In all forms of manometers (U tubes, well-types, and manometer has a uniform tube, the center one has an inclines) there are two liquid surfaces. Pressure deter enlarged leg and the right-hand one has a irregular minations are made by how the fluid moves when leg. Manometers in Figure 3 are open to atmosphere pressures are applied to each surface. For gauge pres on both legs so the indicating fluid level in both legs sure, P2 is equal to zero (atmospheric reference), is the same. -

EPA Requirements & Refrigerant Handling
Course 5: EPA Requirements & Refrigerant Handling November 4 & 6, 2014 1 Federal Clean Air Act (sc. 608) All persons who maintain, service, repair, or dispose of appliances that contain regulated refrigerants, be cerGfied in proper refrigerant handling techniques. If EPA regulaons change aer a technician becomes cerGfied, it is the responsibility of the technician to comply with any future changes. 2 Technician Certification 4 Categories of Technician Cer1ficaon Type I: Persons who maintain, service or repair small appliances must be cerGfied as Type I technicians. Type II: Persons, who maintain, service, repair or dispose of high or very high-pressure appliances, except small appliances and motor vehicle air condiGoning systems, must be cerGfied as Type II technicians. 3 Technician Certification (cont.) 4 Categories of Technician Cer1ficaon Type III: Persons, who maintain, service, repair, or dispose of low-pressure appliances must be cerGfied as Type III technicians. Type IV (Universal): Persons, who maintain, service or repair both low and high-pressure equipment, as well as small appliances, must be cerGfied as Universal technicians. 4 Refrigeration Definition Heat is a form of energy. Refrigeraon is the movement of heat from an area where it is not wanted to an area where it is less objecGonable. For example, a refrigerator removes heat from the inside of the cabinet and transfers it to the outside. 5 Gauge Manifold Set One of the most important tools to the HVAC&R technician is the gauge manifold set. The compound gauge (BLUE) and the high pressure gauge (RED) are connected to the manifold, and the manifold is then connected by hoses to access ports to measure system pressures. -

Fed-Std-376B
i Fed-Std-376B Foreword This standard was developed by the Standards and Metric Practices Subcommittee of the Metrication Operating Committee, which operates under the Interagency Council on Metric Policy. It is the basic Federal standard that lists metric units recommended for use throughout the Federal government, and is specified in the Federal Standardization Handbook, issued by the General Services Administration in accordance with 41 CFR 101-29. Before issue, it was coordinated with the departments and agencies of the Interagency Council on Metric Policy. The General Services Administration has authorized the use of this Federal standard by all Federal agencies. Civilian Agency Coordinating Activity: Federal Supply Service, General Services Administration Military Agency Coordinating Activity: Standardization Program, Office of the Assistant Secretary (Production and Logistics), Department of Defense Preparing Activity: Metric Program, National Institute of Standards and Technology, Technology Administration, Department of Commerce Changes When a federal agency determines that there is a need for a revision of this standard, a written request for revision should be submitted to the General Services Administration, Federal Supply Service, Environmental and Engineering Policy Division (FCRE), Washington, DC 20406. The request shall include data that support the proposed change. The Metric Program, National Institute of Standards and Technology, as custodian of this standard, will coordinate all proposed changes with the Metrication