Amide Prodrugs of Nsaids
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Contrasting Effects of Diclofenac and Ibuprofen on Active Imatinib Uptake Into Leukaemic Cells British Journal of Cancer, 2012; 106(11):1772-1778
PUBLISHED VERSION Wang, Jueqiong; Hughes, Timothy Peter; Kok, Chung Hoow; Saunders, Verity Ann; Frede, Amity; Groot- Obbink, Kelvin Stuart; Osborn, M.; Somogyi, Andrew Alexander; D'Andrea, Richard James; White, Deborah Lee Contrasting effects of diclofenac and ibuprofen on active imatinib uptake into leukaemic cells British Journal of Cancer, 2012; 106(11):1772-1778 © 2012 Cancer Research UK From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/ The electronic version of this article is the complete one and can be found online at: http://www.nature.com/bjc/journal/v106/n11/full/bjc2012173a.html PERMISSIONS http://www.nature.com/bjc/authors/submit.html#open BJC publishes BJC OPEN articles under a Creative Commons Attribution-Non commercial-Share Alike 3.0 Unported licence. This licence allows users to: share - to copy, distribute and transmit the work remix - to adapt the work Under the following conditions: attribution - users must attribute the work in the manner specified by the author or licensor (but not in any way that suggests an endorsement ) non commercial - users may not use this work for commercial purposes share alike - If users alter, transform, or build upon this work, they may distribute the resulting work only under the same or similar license to this one 20th May 2013 http://hdl.handle.net/2440/72375 British Journal of Cancer -

Fenbufen | Medchemexpress
Inhibitors Product Data Sheet Fenbufen • Agonists Cat. No.: HY-B1138 CAS No.: 36330-85-5 Molecular Formula: C₁₆H₁₄O₃ • Molecular Weight: 254.28 Screening Libraries Target: COX; Caspase Pathway: Immunology/Inflammation; Apoptosis Storage: Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month SOLVENT & SOLUBILITY In Vitro DMSO : 50 mg/mL (196.63 mM; Need ultrasonic) H2O : 0.67 mg/mL (2.63 mM; Need ultrasonic) Mass Solvent 1 mg 5 mg 10 mg Concentration Preparing 1 mM 3.9327 mL 19.6634 mL 39.3267 mL Stock Solutions 5 mM 0.7865 mL 3.9327 mL 7.8653 mL 10 mM 0.3933 mL 1.9663 mL 3.9327 mL Please refer to the solubility information to select the appropriate solvent. In Vivo 1. Add each solvent one by one: 10% DMSO >> 90% (20% SBE-β-CD in saline) Solubility: 2.5 mg/mL (9.83 mM); Suspended solution; Need ultrasonic 2. Add each solvent one by one: 10% DMSO >> 90% corn oil Solubility: ≥ 2.5 mg/mL (9.83 mM); Clear solution BIOLOGICAL ACTIVITY Description Fenbufen (CL-82204) is an orally active non-steroidal anti-inflammatory drug (NSAID), with analgetic and antipyretic effects. Fenbufen has potent activity in a variety of animal model, including carageenin edema, UV erythema and adjuvant arthritis. Fenbufen has inhibitory activities against COX-1 and COX-2 with IC50s of 3.9 μM and 8.1 μM, respectively. Fenbufen is a caspases (caspase-1, 3, 4, 5, 9) inhibitor[1][2][3][4][5]. -

Long-Term Use of Anti-Inflammatory Drugs and Risk of Atrial Fibrillation
ORIGINAL INVESTIGATION Long-term Use of Anti-inflammatory Drugs and Risk of Atrial Fibrillation Raffaele De Caterina, MD, PhD; Ana Ruigo´mez, MD, PhD; Luı´s Alberto Garcı´a Rodrı´guez, MD, MSc Background: Previous reports have described an asso- ratio [RR], 2.49; 95% confidence interval [CI], 1.56- ciation between the use of corticosteroids (steroidal anti- 3.97). However, we also found that the current use of inflammatory drugs [SAIDs]) and the risk of atrial fibril- NSAIDs was associated with an increased risk of chronic lation (AF). We sought to determine the existence of a AF (RR, 1.44; 95% CI, 1.08-1.91). Such risk was further similar association for non-SAIDs (NSAIDs). increased among long-term users with a treatment du- ration of longer than 1 year (RR, 1.80; 95% CI, 1.20- Methods: We identified patients aged 40 to 89 years with 2.72). The increased risk of chronic AF was not ex- a first-ever diagnosis of AF in 1996 in a United King- plained by the occurrence of heart failure. The use of dom primary care database and classified them as hav- NSAIDs was not associated with paroxysmal AF. ing paroxysmal or chronic AF. After validation with their primary care physicians, 1035 patients were confirmed Conclusions: The use of NSAIDs, as for SAIDs, is asso- as having incident chronic AF and 525 as having parox- ciated with an increased risk of chronic AF. Because the ysmal AF. Two separate nested case-control analyses es- use of anti-inflammatory drugs in general is a marker for timated the risk of first-time chronic and paroxysmal AF underlying inflammatory disorders, inflammation may among users of SAIDs and NSAIDs. -

Inflammatory Drugs
Solubility of Nonsteroidal Anti- diflunisal, etoricoxib, fenbufen, fentiazac, flufenamic inflammatory Drugs (NSAIDs) in acid, flurbiprofen, ibuprofen, indomethacin, ketopro- Neat Organic Solvents and Organic fen, ketorolac, lornoxicam, mefenamic acid, melox- Solvent Mixtures iam, nabumetone, naproxen, niflumic acid, nimesulide, phenylbutazone, piroxicam, rofecoxib, sodium diclof- William E. Acree enac, sodium ibuprofen, sodium naproxen, sodium J. Phys. Chem. Ref. Data 43, 023102 (2014) salicylate, tenoxicam, tolfenamic acid, and valdecoxib. IUPAC-NIST Solubility Data Series, Volume 102 http://dx.doi.org/10.1063/1.4869683 This IUPAC-NIST Solubility Data Series volume reviews experimentally determined solubility data for 33 non- To access recent volumes in the Solubility Data Series, visit http://jpcrd.aip.org/ and steroidal anti-inflammatory drugs (NSAIDs) dissolved search IUPAC-NIST Solubility Data Series in neat organic solvents and well-defined binary and ternary organic solvent mixtures retrieved from the published chemical and pharmaceutical litera- ture covering the period from 1980 to the beginning of 2014. Except for aspirin (2-acetoxybenzoic acid) and salicylic acid (2-hydroxybenzoic acid), very little Provisional Recommendations physical and chemical property data are available Provisional Recommendations are drafts of IUPAC in the published literature for NSAIDs prior to 1980. recommendations on terminology, nomenclature, and Solubility data are compiled and critically reviewed symbols made widely available to allow interested -

Rheological and Mechanical Analyses of Felbinac Cataplasms by Using Box–Behnken Design
pharmaceutics Article Rheological and Mechanical Analyses of Felbinac Cataplasms by Using Box–Behnken Design Jie Yang, Yishen Zhu * ID , Yongqin Diao and Caiyun Yin College of Biotechnology and Pharmaceutical Engineering, Nanjing Tech University, Nanjing 211816, China; [email protected] (J.Y.); [email protected] (Y.D.); [email protected] (C.Y.) * Correspondence: [email protected]; Tel.: +86-189-0066-0563; Fax: +86-25-5813-9910 Received: 1 June 2018; Accepted: 5 July 2018; Published: 11 July 2018 Abstract: Felbinac, an active pharmaceutical ingredient (API) used clinically for the treatment of osteoarthritis, has poor solubility. Felbinac cataplasm product design was investigated using rheological and mechanical analyses. Experiments using a response surface methodology based on Box–Behnken design (BBD) incorporated three independent variables: the proportions of partially neutralized polyacrylate (NP800), dihydroxyaluminum aminoacetate (DAAA), and felbinac. Statistically significant quadratic models obtained using BBD demonstrated optimal NP-800, DAAA, and felbinac cataplasm proportions of 4.78–5.75%, 0.30–0.59%, and 0.70–0.90%, respectively. Felbinac cataplasms exhibited “gel-like” mechanical property with predominantly elastic behavior. Rheological studies correlated increasing NP-800 and DAAA concentrations with increased complex modulus (G*) values that were inversely related to peeling strength. Frequency sweep and creep tests revealed decreasing tan θ values with increasing NP-800 and DAAA concentrations. G’ and G” values were higher for higher NP-800 and DAAA levels, although G” values decreased with increasing DAAA concentration. Response surface methodology was applied to develop mathematical models. Variance analysis showed that the quadratic model effectively predicted felbinac and matrix material interactions, with two verification samples upholding model predictions. -

(12) United States Patent (10) Patent No.: US 6,183,779 B1 10 11 12
USOO6183779B1 (12) United States Patent (10) Patent No.: US 6,183,779 B1 Ouali et al. (45) Date of Patent: Feb. 6, 2001 (54) STABILIZED PHARMACEUTICAL 5,698,225 12/1997 Gimet et al. ......................... 424/475 COMPOSITION OF ANONSTEROIDAL ANTI-NFLAMMATORY AGENT AND A FOREIGN PATENT DOCUMENTS PROSTAGLANDIN 91/16895 11/1991 (WO). 99/12524 3/1999 (WO). (75) Inventors: Aomar Ouali, Boisbriand; Abul Kalam 99/65496 12/1999 (WO). Azad, Montreal, both of (CA) 00/01368 1/2000 (WO). 00/15200 3/2000 (WO). (73) Assignee: Pharmascience Inc., Montreal (CA) OTHER PUBLICATIONS (*) Notice: Under 35 U.S.C. 154(b), the term of this Searle HealthNet Prescribing Information for Arthrotec(R) patent shall be extended for 0 days. (downloaded from http://www.searlehealthnet.com/pi/ar throtec.html on Oct. 27, 1998). (21) Appl. No.: 09/273,692 Information for the Patient: ControtecTM. (22) Filed: Mar. 22, 1999 * cited by examiner Primary Examiner James M. Spear (51) Int. Cl. ............................... A61K 9/22; A61 K9/24; (74) Attorney, Agent, or Firm-Dianne E. Reed; J. Elin A61 K9/52; A61 K9/54 Hartrum; Reed & Associates (52) U.S. Cl. .......................... 424/472; 424/451; 424/457; 424/458; 424/465; 424/468; 424/470; 424/474; (57) ABSTRACT 424/489; 514/772.3; 514/781; 514/970 A pharmaceutical composition is provided for the oral (58) Field of Search ..................................... 424/472, 474, administration of an NSAID and a prostaglandin. The com 424/451, 464, 465, 489, 470, 457, 468, position is a solid dosage form wherein the NSAID is 458 enterically coated and the prostaglandin is present along with an effective Stabilizing amount of a prostaglandin (56) References Cited Stabilizing agent Such as hydroxypropyl methylcellulose or U.S. -

Stembook 2018.Pdf
The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances FORMER DOCUMENT NUMBER: WHO/PHARM S/NOM 15 WHO/EMP/RHT/TSN/2018.1 © World Health Organization 2018 Some rights reserved. This work is available under the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 IGO licence (CC BY-NC-SA 3.0 IGO; https://creativecommons.org/licenses/by-nc-sa/3.0/igo). Under the terms of this licence, you may copy, redistribute and adapt the work for non-commercial purposes, provided the work is appropriately cited, as indicated below. In any use of this work, there should be no suggestion that WHO endorses any specific organization, products or services. The use of the WHO logo is not permitted. If you adapt the work, then you must license your work under the same or equivalent Creative Commons licence. If you create a translation of this work, you should add the following disclaimer along with the suggested citation: “This translation was not created by the World Health Organization (WHO). WHO is not responsible for the content or accuracy of this translation. The original English edition shall be the binding and authentic edition”. Any mediation relating to disputes arising under the licence shall be conducted in accordance with the mediation rules of the World Intellectual Property Organization. Suggested citation. The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances. Geneva: World Health Organization; 2018 (WHO/EMP/RHT/TSN/2018.1). Licence: CC BY-NC-SA 3.0 IGO. Cataloguing-in-Publication (CIP) data. -

ARCI Uniform Classification Guidelines for Foreign Substances, Or Similar State Regulatory Guidelines, Shall Be Assigned Points As Follows
DRUG TESTING STANDARDS AND PRACTICES PROGRAM. Uniform Classification Guidelines for Foreign Substances And Recommended Penalties Model Rule. January, 2019 (V.14.0) © ASSOCIATION OF RACING COMMISSIONERS INTERNATIONAL – 2019. Association of Racing Commissioners International 2365 Harrodsburg Road- B450 Lexington, Kentucky, USA www.arci.com Page 1 of 66 Preamble to the Uniform Classification Guidelines of Foreign Substances The Preamble to the Uniform Classification Guidelines was approved by the RCI Drug Testing and Quality Assurance Program Committee (now the Drug Testing Standards and Practices Program Committee) on August 26, 1991. Minor revisions to the Preamble were made by the Drug Classification subcommittee (now the Veterinary Pharmacologists Subcommittee) on September 3, 1991. "The Uniform Classification Guidelines printed on the following pages are intended to assist stewards, hearing officers and racing commissioners in evaluating the seriousness of alleged violations of medication and prohibited substance rules in racing jurisdictions. Practicing equine veterinarians, state veterinarians, and equine pharmacologists are available and should be consulted to explain the pharmacological effects of the drugs listed in each class prior to any decisions with respect to penalities to be imposed. The ranking of drugs is based on their pharmacology, their ability to influence the outcome of a race, whether or not they have legitimate therapeutic uses in the racing horse, or other evidence that they may be used improperly. These classes of drugs are intended only as guidelines and should be employed only to assist persons adjudicating facts and opinions in understanding the seriousness of the alleged offenses. The facts of each case are always different and there may be mitigating circumstances which should always be considered. -

(CD-P-PH/PHO) Report Classification/Justifica
COMMITTEE OF EXPERTS ON THE CLASSIFICATION OF MEDICINES AS REGARDS THEIR SUPPLY (CD-P-PH/PHO) Report classification/justification of - Medicines belonging to the ATC group M01 (Antiinflammatory and antirheumatic products) Table of Contents Page INTRODUCTION 6 DISCLAIMER 8 GLOSSARY OF TERMS USED IN THIS DOCUMENT 9 ACTIVE SUBSTANCES Phenylbutazone (ATC: M01AA01) 11 Mofebutazone (ATC: M01AA02) 17 Oxyphenbutazone (ATC: M01AA03) 18 Clofezone (ATC: M01AA05) 19 Kebuzone (ATC: M01AA06) 20 Indometacin (ATC: M01AB01) 21 Sulindac (ATC: M01AB02) 25 Tolmetin (ATC: M01AB03) 30 Zomepirac (ATC: M01AB04) 33 Diclofenac (ATC: M01AB05) 34 Alclofenac (ATC: M01AB06) 39 Bumadizone (ATC: M01AB07) 40 Etodolac (ATC: M01AB08) 41 Lonazolac (ATC: M01AB09) 45 Fentiazac (ATC: M01AB10) 46 Acemetacin (ATC: M01AB11) 48 Difenpiramide (ATC: M01AB12) 53 Oxametacin (ATC: M01AB13) 54 Proglumetacin (ATC: M01AB14) 55 Ketorolac (ATC: M01AB15) 57 Aceclofenac (ATC: M01AB16) 63 Bufexamac (ATC: M01AB17) 67 2 Indometacin, Combinations (ATC: M01AB51) 68 Diclofenac, Combinations (ATC: M01AB55) 69 Piroxicam (ATC: M01AC01) 73 Tenoxicam (ATC: M01AC02) 77 Droxicam (ATC: M01AC04) 82 Lornoxicam (ATC: M01AC05) 83 Meloxicam (ATC: M01AC06) 87 Meloxicam, Combinations (ATC: M01AC56) 91 Ibuprofen (ATC: M01AE01) 92 Naproxen (ATC: M01AE02) 98 Ketoprofen (ATC: M01AE03) 104 Fenoprofen (ATC: M01AE04) 109 Fenbufen (ATC: M01AE05) 112 Benoxaprofen (ATC: M01AE06) 113 Suprofen (ATC: M01AE07) 114 Pirprofen (ATC: M01AE08) 115 Flurbiprofen (ATC: M01AE09) 116 Indoprofen (ATC: M01AE10) 120 Tiaprofenic Acid (ATC: -

Supervision Registers for Mentally Ill People Medicolegal Issues Seem Likely to Dominate Decisions by Clinicians
LONDON, SATURDAY 3 SEPTEMBER 1994 Supervision registers for mentally ill people Medicolegal issues seem likely to dominate decisions by clinicians The Department of Health and the Royal College of personal liability, but they are time consuming and Psychiatrists do not see eye to eye over the introduction of expensive. Judicial review in relation to child abuse supervision registers for patients in the community who are registers is increasing, and supervision registers seem likely judged to be at risk. In a recent exchange of corres- to follow their example. pondence the college expressed "strong concerns" about The introduction of supervision registers will be an invi- guidelines issued by the department for the introduction of tation to litigation. Nowhere is this clearer than in the case the register on 1 October. 1-3 Further discussions are of a failure to include a person on the register, when in the planned, but the differences will not easily be resolved. event of a subsequent untoward incident this decision may The issue is much more than a little local difficulty in retrospect create an impression of negligence, whatever between psychiatrists and the Department of Health; its the reality. The prediction of dangerousness is far from an resolution will be important for all mental health profes- exact science, and a court might recognise that-but only sionals and for purchasers of psychiatric services. The at trial, after the defendants have undergone considerable college is concerned that the criteria for including patients frustration, professional soul searching, and expense. on supervision registers are too broad and about the sub- Violent incidents and tragic suicides provoke enormous stantial costs of setting up and servicing the registers. -
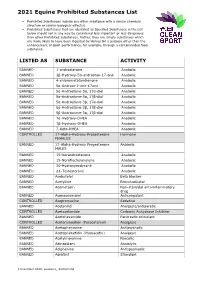
2021 Equine Prohibited Substances List
2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). Prohibited Substances that are identified as Specified Substances in the List below should not in any way be considered less important or less dangerous than other Prohibited Substances. Rather, they are simply substances which are more likely to have been ingested by Horses for a purpose other than the enhancement of sport performance, for example, through a contaminated food substance. LISTED AS SUBSTANCE ACTIVITY BANNED 1-androsterone Anabolic BANNED 3β-Hydroxy-5α-androstan-17-one Anabolic BANNED 4-chlorometatandienone Anabolic BANNED 5α-Androst-2-ene-17one Anabolic BANNED 5α-Androstane-3α, 17α-diol Anabolic BANNED 5α-Androstane-3α, 17β-diol Anabolic BANNED 5α-Androstane-3β, 17α-diol Anabolic BANNED 5α-Androstane-3β, 17β-diol Anabolic BANNED 5β-Androstane-3α, 17β-diol Anabolic BANNED 7α-Hydroxy-DHEA Anabolic BANNED 7β-Hydroxy-DHEA Anabolic BANNED 7-Keto-DHEA Anabolic CONTROLLED 17-Alpha-Hydroxy Progesterone Hormone FEMALES BANNED 17-Alpha-Hydroxy Progesterone Anabolic MALES BANNED 19-Norandrosterone Anabolic BANNED 19-Noretiocholanolone Anabolic BANNED 20-Hydroxyecdysone Anabolic BANNED Δ1-Testosterone Anabolic BANNED Acebutolol Beta blocker BANNED Acefylline Bronchodilator BANNED Acemetacin Non-steroidal anti-inflammatory drug BANNED Acenocoumarol Anticoagulant CONTROLLED Acepromazine Sedative BANNED Acetanilid Analgesic/antipyretic CONTROLLED Acetazolamide Carbonic Anhydrase Inhibitor BANNED Acetohexamide Pancreatic stimulant CONTROLLED Acetominophen (Paracetamol) Analgesic BANNED Acetophenazine Antipsychotic BANNED Acetophenetidin (Phenacetin) Analgesic BANNED Acetylmorphine Narcotic BANNED Adinazolam Anxiolytic BANNED Adiphenine Antispasmodic BANNED Adrafinil Stimulant 1 December 2020, Lausanne, Switzerland 2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). -

Drug/Substance Trade Name(S)
A B C D E F G H I J K 1 Drug/Substance Trade Name(s) Drug Class Existing Penalty Class Special Notation T1:Doping/Endangerment Level T2: Mismanagement Level Comments Methylenedioxypyrovalerone is a stimulant of the cathinone class which acts as a 3,4-methylenedioxypyprovaleroneMDPV, “bath salts” norepinephrine-dopamine reuptake inhibitor. It was first developed in the 1960s by a team at 1 A Yes A A 2 Boehringer Ingelheim. No 3 Alfentanil Alfenta Narcotic used to control pain and keep patients asleep during surgery. 1 A Yes A No A Aminoxafen, Aminorex is a weight loss stimulant drug. It was withdrawn from the market after it was found Aminorex Aminoxaphen, Apiquel, to cause pulmonary hypertension. 1 A Yes A A 4 McN-742, Menocil No Amphetamine is a potent central nervous system stimulant that is used in the treatment of Amphetamine Speed, Upper 1 A Yes A A 5 attention deficit hyperactivity disorder, narcolepsy, and obesity. No Anileridine is a synthetic analgesic drug and is a member of the piperidine class of analgesic Anileridine Leritine 1 A Yes A A 6 agents developed by Merck & Co. in the 1950s. No Dopamine promoter used to treat loss of muscle movement control caused by Parkinson's Apomorphine Apokyn, Ixense 1 A Yes A A 7 disease. No Recreational drug with euphoriant and stimulant properties. The effects produced by BZP are comparable to those produced by amphetamine. It is often claimed that BZP was originally Benzylpiperazine BZP 1 A Yes A A synthesized as a potential antihelminthic (anti-parasitic) agent for use in farm animals.