Validation of Thin-Layer Chromatographic Methods for Pesticide Residue Analysis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Bmps) for Wildland Stewardship
Best Management Practices (BMPs) for Wildland Stewardship Protecting Wildlife When Using Herbicides for Invasive Plant Management California Invasive Plant Council & Pesticide Research Institute ontrolling invasive plants is often a high priority when protecting wildlife habitat, and those working to protect Cwildlife from invasive plants want to be sure their approach is safe for wildlife. This manual of Best Management Practices focuses on how land managers can best protect wildlife when using herbicides to control invasive plants. While any invasive plant control method can potentially impact wildlife, chemical control methods are the focus of this report. The toxicology information presented shows data on herbicides most commonly used for invasive plant management in California natural areas. The Best Management Practices are drawn from methods used by experienced land managers. Along with providing guidance for land managers, this document is designed to inform the interested public about how herbicides are used to control invasive plants in natural areas. ©2015 California Invasive Plant Council Available at www.cal-ipc.org Cite this report as: Cal-IPC. 2015. Best Management Practices for Wildland Stewardship: Protecting Wildlife When Using Herbicides for Invasive Plant Management. Cal-IPC Publication 2015-1. California Invasive Plant Council, Berkeley, CA. Available: www.cal-ipc.org Cover photos: Large photo: American goldfinch by Gary Kramer, USFWS Top small photo: Herbicide applicator by Jim Dempsey, California State Parks Bottom small photo: Pacific tree frog by Sandy DeSimone, Audubon Starr Ranch Contents 1. Introduction . 1 Wildland Stewardship, Invasive Plant Management and Wildlife . 1 The Importance of Best Management Practices . 3 2. Invasive Plant Management and Wildlife . -

Experiment 2 — Distillation and Gas Chromatography
Chem 21 Fall 2009 Experiment 2 — Distillation and Gas Chromatography _____________________________________________________________________________ Pre-lab preparation (1) Read the supplemental material on distillation theory and techniques from Zubrick, The Organic Chem Lab Survival Manual, and the section on Gas Chromatography from Fessenden, Fessenden, and Feist, Organic Laboratory Techniques, then read this handout carefully. (2) In your notebook, write a short paragraph summarizing what you will be doing in this experiment and what you hope to learn about the efficiencies of the distillation techniques. (3) Sketch the apparatus for the simple and fractional distillations. Your set-up will look much like that shown on p 198 of Zubrick, except that yours will have a simple drip tip in place of the more standard vacuum adaptor. (4) Look up the structures and relevant physical data for the two compounds you will be using. What data are relevant? Read the procedure, think about the data analysis, and decide what you need. (5) Since you have the necessary data, calculate the log of the volatility factor (log α) that you will need for the theoretical plate calculation. Distillation has been used since antiquity to separate the components of mixtures. In one form or another, distillation is used in the manufacture of perfumes, flavorings, liquors, and a variety of other organic chemicals. One of its most important modern applications is in refining crude oil to make fuels, lubricants, and other petrochemicals. The first step in the refining process is separation of crude petroleum into various hydrocarbon fractions by distillation through huge fractionating columns, called distillation towers, that are hundreds of feet high. -

EFFICIENCY of a ROTARY DISTILLATION COLUMN 12 by Beveridge J
U. S. DEPARTMENT OP COMMERCB NATIONAL BUREAU OF STANDARDS RESEARCH PAPER RPl201 Part of Journa.l of Research of the National Bureau of Standards, Volume 22, Ma.y 1939 EFFICIENCY OF A ROTARY DISTILLATION COLUMN 12 By Beveridge J. Mair and Charles B. Willingham a ABSTRACT Determinations of the height equivalent to a theorfltical plate (H.E.T.P.); hold-up, and through-put were made on a rotary distillation column of the Pegram-Urey-Huffman type having a rectifying section 56 em long and 5.08 cm in diameter. Using 8S rotatinl!; members (1) baskets with fine holes, (2) flat plates, and (3) cones. test s of separating efficiency were made at speeds of rota tion ranging from 250 to 1,500 rpm and through-puts ranging from 1 to 10 ml of liquid per minute. The binary 8 y ~ tem methylcyclohexane-n-heptane was used as the test mixture. The lowest H.E T.P. was obtained with the conical rotating members spaced 0.64 cm apart, with which the following average results were obtained: H.E.T.P., 1.04 cm; hold-up, 1.3 to 1.6 ml of liqnid per theoretical plate; maximum through-put, 10 ml 0f liquid per minute. Within the reproducibility of these experiments, there was no variation in H.E.T.P. for values of through-. put from 2 to 10 ml of liquid per minute and speeds of rotation from 250 to 1,500 rpm. CONTENTS Page I. Introduction __________________ _______________________________ __ _ 519 II. Column assembly _______________________ ___________ ______ ___ ___ __ 520 1. -

WO 2010/128115 Al
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date 11 November 2010 (11.11.2010) WO 2010/128115 Al (51) International Patent Classification: (81) Designated States (unless otherwise indicated, for every C12N 9/16 (2006.01) kind of national protection available): AE, AG, AL, AM, AO, AT, AU, AZ, BA, BB, BG, BH, BR, BW, BY, BZ, (21) International Application Number: CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, PCT/EP20 10/056204 DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, (22) International Filing Date: HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, 6 May 2010 (06.05.2010) KR, KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, (25) Filing Language: English NO, NZ, OM, PE, PG, PH, PL, PT, RO, RS, RU, SC, SD, (26) Publication Language: English SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (30) Priority Data: 09159520.7 6 May 2009 (06.05.2009) EP (84) Designated States (unless otherwise indicated, for every kind of regional protection available): ARIPO (BW, GH, (71) Applicant (for all designated States except US): GM, KE, LR, LS, MW, MZ, NA, SD, SL, SZ, TZ, UG, NOVOZYMES A/S [DK/DK]; Krogshoejvej 36, ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, MD, RU, TJ, DK-2880 Bagsvaerd (DK). -

Impact of Pesticide Use on Health in Developing Countries
Impact of pesticide use on health in developing countries Proceedings of a symposium held in Ottawa, Canada, 1 7-20 September 1990 IDRC CRDI International Development Research Centre Centre de recherches pour le devetoppement international 1 March 1993 Dear Reader/Librarian, IDRC is a public corporation created by the Canadian parliament in 1970 to help developing countries find viable solutions to their problems through research. At the 1992 Earth Summit, IDRC's mandate was broadened to emphasize sustainable development issues. As part of IDRC's strengthened commitment to global action and harüony, we are pleased to send you a complimentary copy of our most recent publication: The impact of pesticide use on health in developing countries (March 1993, 352 pages, 0-88936-560-1, $17.95). The first part of this book presents a brief survey of the global situation and the results of twelve epidemiological studies carried out by researchers from Africa, Latin America, Asia and the Middle East. These focus on poisonings resulting from organophosphates, herbicides, and pyrethroids. The second part illustrates the role of the process of development, production, spraying techniques and legislation in protecting the health of workers. A discussion of the benefits and modalities of access to pertinent information for the prevention of pesticide poisonings is provided in the third section. Finally, in the fourth section, consideration is given to the advantages and disadvantages of certain alternatives to the use of synthetic pesticides in agriculture and public health, such as botanical pesticides and integrated pest management strategies. We hope this book is a valuable addition to your collection. -

Consortium for International Crop Protection Pest Management & Related Environmental Protection Project*
CONSORTIUM FOR INTERNATIONAL CROP PROTECTION PEST MANAGEMENT & RELATED ENVIRONMENTAL PROTECTION PROJECT* ANNUAL REPORT TO AGENCY FOR INTERNATIONAL DEVELOPMENT OCTOBER 1983 - SEPIEMBER 1984 Ray F. Smith, University of California, Executive Director Member Institutions: Cornell University North Carolina State University Oregon State University Texas A&M University University of California University of Florida University of Hawaii University of Illinois University of Maryland University of Miami, Florida University of Minnesota Purdue University University of Puerto Rico U. S. Department of Agriculture * Contract No. AID/DSAN-C-0252 Project No. 931-0930 TABLE OF CONTENTS Page Introduction .......................................................... 1 Response to USAID Mission Requests for Technical Assistance ....................................... 6 Sudan ............................................... 7 Niger .......... ............................ ..... ..... 20 Thailand ........ ............................ .... ..... 23 Bolivia......................... ................. 28 Ecuador .............................................. 31 Belize ......................................... 35 Caribbean................. ....................... 40 Kenya........................................... 51 Grenada ..... ..................................... 54 Mexico ........... ....... .................... .. 58 Program Evaluation or Design .......................................... 62 Cameroon .................................................... 63 Central -

Distillation Handbook
I distillation terminology To provide a better understanding of the distillation process, the following briefly explains the terminology most often encountered. SOLVENT RECOVERY The term "solvent recovery" often has been a somewhat vague label applied to the many and very different ways in which solvents can be reclaimed by industry. One approach employed in the printing and coatings industries is merely to take impure solvents containing both soluble and insoluble particles and evapo- rate the solvent from the solids. For a duty of this type, APV offers the Paraflash evaporator, a compact unit which combines a Paraflow plate heat exchanger and a small separator. As the solvent ladened liquid is recirculated through the heat exchanger, it is evaporated and the vapor and liquid separated. This will recover a solvent but it will not separate solvents if two or more are present. Afurther technique is available to handle an air stream that carries solvents. By chilling the air by means of vent condensers or refrigeration equipment, the solvents can be removed from the condenser. Solvents also can be recovered by using extraction, adsorption, absorption, and distillation methods. S 0 LV E NT EXT R ACT I0 N Essentially a liquid/liquid process where one liquid is used to extract another from a secondary stream, solvent extraction generally is performed in a column somewhat similar to a normal distillation column. The primary difference is that the process involves two liquids instead of liquid and vapor. During the process, the lighter (i.e., less dense) liquid is charged to the base of the column and rises through packing or trays while the more dense liquid descends. -

Chemical Name Federal P Code CAS Registry Number Acutely
Acutely / Extremely Hazardous Waste List Federal P CAS Registry Acutely / Extremely Chemical Name Code Number Hazardous 4,7-Methano-1H-indene, 1,4,5,6,7,8,8-heptachloro-3a,4,7,7a-tetrahydro- P059 76-44-8 Acutely Hazardous 6,9-Methano-2,4,3-benzodioxathiepin, 6,7,8,9,10,10- hexachloro-1,5,5a,6,9,9a-hexahydro-, 3-oxide P050 115-29-7 Acutely Hazardous Methanimidamide, N,N-dimethyl-N'-[2-methyl-4-[[(methylamino)carbonyl]oxy]phenyl]- P197 17702-57-7 Acutely Hazardous 1-(o-Chlorophenyl)thiourea P026 5344-82-1 Acutely Hazardous 1-(o-Chlorophenyl)thiourea 5344-82-1 Extremely Hazardous 1,1,1-Trichloro-2, -bis(p-methoxyphenyl)ethane Extremely Hazardous 1,1a,2,2,3,3a,4,5,5,5a,5b,6-Dodecachlorooctahydro-1,3,4-metheno-1H-cyclobuta (cd) pentalene, Dechlorane Extremely Hazardous 1,1a,3,3a,4,5,5,5a,5b,6-Decachloro--octahydro-1,2,4-metheno-2H-cyclobuta (cd) pentalen-2- one, chlorecone Extremely Hazardous 1,1-Dimethylhydrazine 57-14-7 Extremely Hazardous 1,2,3,4,10,10-Hexachloro-6,7-epoxy-1,4,4,4a,5,6,7,8,8a-octahydro-1,4-endo-endo-5,8- dimethanonaph-thalene Extremely Hazardous 1,2,3-Propanetriol, trinitrate P081 55-63-0 Acutely Hazardous 1,2,3-Propanetriol, trinitrate 55-63-0 Extremely Hazardous 1,2,4,5,6,7,8,8-Octachloro-4,7-methano-3a,4,7,7a-tetra- hydro- indane Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]- 51-43-4 Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]-, P042 51-43-4 Acutely Hazardous 1,2-Dibromo-3-chloropropane 96-12-8 Extremely Hazardous 1,2-Propylenimine P067 75-55-8 Acutely Hazardous 1,2-Propylenimine 75-55-8 Extremely Hazardous 1,3,4,5,6,7,8,8-Octachloro-1,3,3a,4,7,7a-hexahydro-4,7-methanoisobenzofuran Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime 26419-73-8 Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime. -
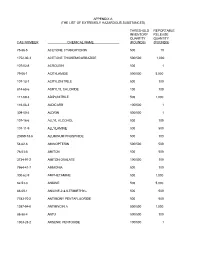
The List of Extremely Hazardous Substances)
APPENDIX A (THE LIST OF EXTREMELY HAZARDOUS SUBSTANCES) THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 75-86-5 ACETONE CYANOHYDRIN 500 10 1752-30-3 ACETONE THIOSEMICARBAZIDE 500/500 1,000 107-02-8 ACROLEIN 500 1 79-06-1 ACRYLAMIDE 500/500 5,000 107-13-1 ACRYLONITRILE 500 100 814-68-6 ACRYLYL CHLORIDE 100 100 111-69-3 ADIPONITRILE 500 1,000 116-06-3 ALDICARB 100/500 1 309-00-2 ALDRIN 500/500 1 107-18-6 ALLYL ALCOHOL 500 100 107-11-9 ALLYLAMINE 500 500 20859-73-8 ALUMINUM PHOSPHIDE 500 100 54-62-6 AMINOPTERIN 500/500 500 78-53-5 AMITON 500 500 3734-97-2 AMITON OXALATE 100/500 100 7664-41-7 AMMONIA 500 100 300-62-9 AMPHETAMINE 500 1,000 62-53-3 ANILINE 500 5,000 88-05-1 ANILINE,2,4,6-TRIMETHYL- 500 500 7783-70-2 ANTIMONY PENTAFLUORIDE 500 500 1397-94-0 ANTIMYCIN A 500/500 1,000 86-88-4 ANTU 500/500 100 1303-28-2 ARSENIC PENTOXIDE 100/500 1 THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 1327-53-3 ARSENOUS OXIDE 100/500 1 7784-34-1 ARSENOUS TRICHLORIDE 500 1 7784-42-1 ARSINE 100 100 2642-71-9 AZINPHOS-ETHYL 100/500 100 86-50-0 AZINPHOS-METHYL 10/500 1 98-87-3 BENZAL CHLORIDE 500 5,000 98-16-8 BENZENAMINE, 3-(TRIFLUOROMETHYL)- 500 500 100-14-1 BENZENE, 1-(CHLOROMETHYL)-4-NITRO- 500/500 500 98-05-5 BENZENEARSONIC ACID 10/500 10 3615-21-2 BENZIMIDAZOLE, 4,5-DICHLORO-2-(TRI- 500/500 500 FLUOROMETHYL)- 98-07-7 BENZOTRICHLORIDE 100 10 100-44-7 BENZYL CHLORIDE 500 100 140-29-4 BENZYL CYANIDE 500 500 15271-41-7 BICYCLO[2.2.1]HEPTANE-2-CARBONITRILE,5- -

Controlling Protein-Surface Interactions in Chromatography
CONTROLLING PROTEIN-SURFACE INTERACTIONS IN CHROMATOGRAPHY USING MIXED SELF-ASSEMBLED MONOLAYERS By Chrysanty Tedjo Dissertation Submitted to the Faculty of the Graduate School of Vanderbilt University in partial fulfillment of the requirements for the degree of DOCTOR OF PHILOSOPHY in Chemical Engineering May, 2011 Nashville, Tennessee Approved: Professor Paul E. Laibinis Professor David E. Cliffel Professor Scott A. Guelcher Professor G. Kane Jennings Professor M. Douglas LeVan Copyright © 2011 by Chrysanty Tedjo All Rights Reserved To my parents, for their continuous support and endless love iii ACKNOWLEDGEMENTS I would first like to acknowledge my advisor Professor Paul Laibinis for his guidance during my research at Vanderbilt University. I am grateful for all the valuable lessons that I have learnt from him. His critical advice and analytical skills have helped me in solving many research problems. I would like to thank the members of my thesis committee Professor Doug LeVan, Kane Jennings, Scott Guelcher, and David Cliffel, who have assisted me since the beginning of my research. Their critical advice from their own research background has provided me with different perspectives on how to approach my research. I would also like to thank Professor Bridget Rogers for her assistance with the x-ray photoelectron spectroscopy (XPS) in her laboratory. The staff of the Department of Chemical and Biomolecular Engineering also deserves to be recognized for their contribution to my research. I would like to thank Mark Holmes for his assistance in setting the equipments in the laboratory and for various technical advice. I am grateful to know Mary Gilleran and am thankful for her assistance and enjoyable discussions about life in general. -

Acutely / Extremely Hazardous Waste List
Acutely / Extremely Hazardous Waste List Federal P CAS Registry Acutely / Extremely Chemical Name Code Number Hazardous 4,7-Methano-1H-indene, 1,4,5,6,7,8,8-heptachloro-3a,4,7,7a-tetrahydro- P059 76-44-8 Acutely Hazardous 6,9-Methano-2,4,3-benzodioxathiepin, 6,7,8,9,10,10- hexachloro-1,5,5a,6,9,9a-hexahydro-, 3-oxide P050 115-29-7 Acutely Hazardous Methanimidamide, N,N-dimethyl-N'-[2-methyl-4-[[(methylamino)carbonyl]oxy]phenyl]- P197 17702-57-7 Acutely Hazardous 1-(o-Chlorophenyl)thiourea P026 5344-82-1 Acutely Hazardous 1-(o-Chlorophenyl)thiourea 5344-82-1 Extemely Hazardous 1,1,1-Trichloro-2, -bis(p-methoxyphenyl)ethane Extemely Hazardous 1,1a,2,2,3,3a,4,5,5,5a,5b,6-Dodecachlorooctahydro-1,3,4-metheno-1H-cyclobuta (cd) pentalene, Dechlorane Extemely Hazardous 1,1a,3,3a,4,5,5,5a,5b,6-Decachloro--octahydro-1,2,4-metheno-2H-cyclobuta (cd) pentalen-2- one, chlorecone Extemely Hazardous 1,1-Dimethylhydrazine 57-14-7 Extemely Hazardous 1,2,3,4,10,10-Hexachloro-6,7-epoxy-1,4,4,4a,5,6,7,8,8a-octahydro-1,4-endo-endo-5,8- dimethanonaph-thalene Extemely Hazardous 1,2,3-Propanetriol, trinitrate P081 55-63-0 Acutely Hazardous 1,2,3-Propanetriol, trinitrate 55-63-0 Extemely Hazardous 1,2,4,5,6,7,8,8-Octachloro-4,7-methano-3a,4,7,7a-tetra- hydro- indane Extemely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]- 51-43-4 Extemely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]-, P042 51-43-4 Acutely Hazardous 1,2-Dibromo-3-chloropropane 96-12-8 Extemely Hazardous 1,2-Propylenimine P067 75-55-8 Acutely Hazardous 1,2-Propylenimine 75-55-8 Extemely Hazardous 1,3,4,5,6,7,8,8-Octachloro-1,3,3a,4,7,7a-hexahydro-4,7-methanoisobenzofuran Extemely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime 26419-73-8 Extemely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime. -

WO 2017/137734 Al 17 August 2017 (17.08.2017) P O P C T
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2017/137734 Al 17 August 2017 (17.08.2017) P O P C T (51) International Patent Classification: (81) Designated States (unless otherwise indicated, for every C09D 5/02 (2006.01) C08K 5/11 (2006.01) kind of national protection available): AE, AG, AL, AM, C09D 7/00 (2006.01) AO, AT, AU, AZ, BA, BB, BG, BH, BN, BR, BW, BY, BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DJ, DK, DM, (21) Number: International Application DO, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, PCT/GB20 17/050302 HN, HR, HU, ID, IL, IN, IR, IS, JP, KE, KG, KH, KN, (22) International Filing Date: KP, KR, KW, KZ, LA, LC, LK, LR, LS, LU, LY, MA, 8 February 2017 (08.02.2017) MD, ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, OM, PA, PE, PG, PH, PL, PT, QA, RO, RS, (25) Filing Language: English RU, RW, SA, SC, SD, SE, SG, SK, SL, SM, ST, SV, SY, (26) Publication Language: English TH, TJ, TM, TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (30) Priority Data: 1602 188.3 8 February 2016 (08.02.2016) GB (84) Designated States (unless otherwise indicated, for every kind of regional protection available): ARIPO (BW, GH, (71) Applicant: CHEMOXY INTERNATIONAL LIMITED GM, KE, LR, LS, MW, MZ, NA, RW, SD, SL, ST, SZ, [GB/GB]; All Saints Refinery, Cargo Fleet Road, Middles TZ, UG, ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, RU, brough TS3 6AF (GB).