Embryo and Larval Biology of the Deep- Sea Octocoral Dentomuricea Aff
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Coelenterata: Anthozoa), with Diagnoses of New Taxa
PROC. BIOL. SOC. WASH. 94(3), 1981, pp. 902-947 KEY TO THE GENERA OF OCTOCORALLIA EXCLUSIVE OF PENNATULACEA (COELENTERATA: ANTHOZOA), WITH DIAGNOSES OF NEW TAXA Frederick M. Bayer Abstract.—A serial key to the genera of Octocorallia exclusive of the Pennatulacea is presented. New taxa introduced are Olindagorgia, new genus for Pseudopterogorgia marcgravii Bayer; Nicaule, new genus for N. crucifera, new species; and Lytreia, new genus for Thesea plana Deich- mann. Ideogorgia is proposed as a replacement ñame for Dendrogorgia Simpson, 1910, not Duchassaing, 1870, and Helicogorgia for Hicksonella Simpson, December 1910, not Nutting, May 1910. A revised classification is provided. Introduction The key presented here was an essential outgrowth of work on a general revisión of the octocoral fauna of the western part of the Atlantic Ocean. The far-reaching zoogeographical affinities of this fauna made it impossible in the course of this study to ignore genera from any part of the world, and it soon became clear that many of them require redefinition according to modern taxonomic standards. Therefore, the type-species of as many genera as possible have been examined, often on the basis of original type material, and a fully illustrated generic revisión is in course of preparation as an essential first stage in the redescription of western Atlantic species. The key prepared to accompany this generic review has now reached a stage that would benefit from a broader and more objective testing under practical conditions than is possible in one laboratory. For this reason, and in order to make the results of this long-term study available, even in provisional form, not only to specialists but also to the growing number of ecologists, biochemists, and physiologists interested in octocorals, the key is now pre- sented in condensed form with minimal illustration. -

Zoologische Verhandelingen
Corals of the South-west Indian Ocean: VI. The Alcyonacea (Octocorallia) of Mozambique, with a discussion on soft coral distribution on south equatorial East African reefs Y. Benayahu, A. Shlagman & M.H. Schleyer Benayahu, Y., A. Shlagman & M.H. Schleyer. Corals of the South-west Indian Ocean: VI. The Alcyo- nacea (Octocorallia) of Mozambique, with a discussion on soft coral distribution on south equatorial East African reefs. Zool. Verh. Leiden 345, 31.x.2003: 49-57, fig. 1.— ISSN 0024-1652/ISBN 90-73239-89-3. Y. Benayahu & A. Shlagman. Department of Zoology, George S. Wise Faculty of Life Sciences, Tel Aviv University, Ramat Aviv 69978, Israel (e-mail: [email protected]). M.H. Schleyer. Oceanographic Research Institute, P.O. Box 10712, Marine Parade 4056, Durban, South Africa. Key words: Mozambique; East African reefs; Octocorallia; Alcyonacea. A list of 46 species of Alcyonacea is presented for the coral reefs of the Segundas Archipelago and north- wards in Mozambique, as well as a zoogeographical record for the Bazaruto Archipelago in southern Mozambique. Among the 12 genera listed, Rhytisma, Lemnalia and Briareum were recorded on Mozambi- can reefs for the first time and the study yielded 27 new zoogeographical records. The survey brings the number of soft coral species listed for Mozambique to a total of 53. A latitudinal pattern in soft coral diversity along the south equatorial East African coast is presented, with 46 species recorded in Tanza- nia, 46 along the northern coast of Mozambique, dropping to 29 in the Bazaruto Archipelago in southern Mozambique and rising again to 38 along the KwaZulu-Natal coast in South Africa. -
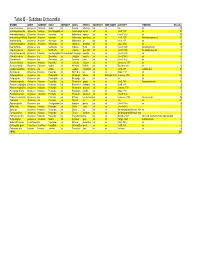
Table B – Subclass Octocorallia
Table B – Subclass Octocorallia BINOMEN ORDER SUBORDER FAMILY SUBFAMILY GENUS SPECIES SUBSPECIES COMN_NAMES AUTHORITY SYNONYMS #Records Acanella arbuscula Alcyonacea Calcaxonia Isididae n/a Acanella arbuscula n/a n/a n/a n/a 59 Acanthogorgia armata Alcyonacea Holaxonia Acanthogorgiidae n/a Acanthogorgia armata n/a n/a Verrill, 1878 n/a 95 Anthomastus agassizii Alcyonacea Alcyoniina Alcyoniidae n/a Anthomastus agassizii n/a n/a (Verrill, 1922) n/a 35 Anthomastus grandiflorus Alcyonacea Alcyoniina Alcyoniidae n/a Anthomastus grandiflorus n/a n/a Verrill, 1878 Anthomastus purpureus 37 Anthomastus sp. Alcyonacea Alcyoniina Alcyoniidae n/a Anthomastus sp. n/a n/a Verrill, 1878 n/a 1 Anthothela grandiflora Alcyonacea Scleraxonia Anthothelidae n/a Anthothela grandiflora n/a n/a (Sars, 1856) n/a 24 Capnella florida Alcyonacea n/a Nephtheidae n/a Capnella florida n/a n/a (Verrill, 1869) Eunephthya florida 44 Capnella glomerata Alcyonacea n/a Nephtheidae n/a Capnella glomerata n/a n/a (Verrill, 1869) Eunephthya glomerata 4 Chrysogorgia agassizii Alcyonacea Holaxonia Acanthogorgiidae Chrysogorgiidae Chrysogorgia agassizii n/a n/a (Verrill, 1883) n/a 2 Clavularia modesta Alcyonacea n/a Clavulariidae n/a Clavularia modesta n/a n/a (Verrill, 1987) n/a 6 Clavularia rudis Alcyonacea n/a Clavulariidae n/a Clavularia rudis n/a n/a (Verrill, 1922) n/a 1 Gersemia fruticosa Alcyonacea Alcyoniina Alcyoniidae n/a Gersemia fruticosa n/a n/a Marenzeller, 1877 n/a 3 Keratoisis flexibilis Alcyonacea Calcaxonia Isididae n/a Keratoisis flexibilis n/a n/a Pourtales, 1868 n/a 1 Lepidisis caryophyllia Alcyonacea n/a Isididae n/a Lepidisis caryophyllia n/a n/a Verrill, 1883 Lepidisis vitrea 13 Muriceides sp. -

Planula Release, Settlement, Metamorphosis and Growth in Two Deep-Sea Soft Corals
REPRODUCTIVE BIOLOGY OF DEEP-SEA SOFT CORALS IN THE NEWFOUNDLAND AND LABRADOR REGION by ©Zhao Sun A thesis submitted to the School of Graduate Studies in partial fulfillment of the requirements for the degree of Master of Science Ocean Sciences Centre and Department of Biology, Memorial University, St. John's (Newfoundland and Labrador) Canada 28 April2009 ABSTRACT This research integrates processing of pre erved samples and, for the first time, long-term monitoring of live colonies and the study of planula behaviour and settlement preferences in four deep-sea brooding octocorals (Alcyonacea: Nephtheidae). Results indicate that reproduction can be correlated to bottom temperature, photoperiod, wind speed and fluctuations in phytoplankton abundance. Large planula larvae are polymorphic, exhibit ubstratum selectivity and can fuse together or with a parent colony. Planulae of two Drifa species are also able to metamorphose in the water column before ettlement. Thi research thus brings evidence of both the resilience (i.e., extended breeding period, demersal larvae with a long competency period) and vulnerability (i.e., substratum selectivity, slow growth) of deep-water corals; and open up new perspectives on experimental tudies of deep-sea organisms. II ACKNOWLEDGEMENTS I would like to thank my supervisor Annie Mercier, as well a Jean-Fran~oi Hamel, for their continuou guidance, support and encouragement. With great patience and pas ion, they helped me adapt to graduate studies. I would also like to thank my co- upervisor Evan Edinger, committee member Paul Snelgrove and examiners Catherine McFadden and Robert Hooper for providing valuable input and for comment on the manuscripts and thesis. -

Symbiont Identity Influences Patterns of Symbiosis Establishment, Host
Reference: Biol. Bull. 234: 1–10. (February 2018) © 2018 The University of Chicago Symbiont Identity Influences Patterns of Symbiosis Establishment, Host Growth, and Asexual Reproduction in a Model Cnidarian- Dinoflagellate Symbiosis YASMIN GABAY1, VIRGINIA M. WEIS2, AND SIMON K. DAVY1,* 1School of Biological Sciences, Victoria University of Wellington, Kelburn Parade, Wellington 6140, New Zealand; and 2Department of Integrative Biology, Oregon State University, Corvallis, Oregon 97331 Abstract. The genus Symbiodinium is physiologically di- study enhances our understanding of the link between symbi- verse and so may differentially influence symbiosis establish- ont identity and the performance of the overall symbiosis, ment and function. To explore this, we inoculated aposymbiotic which is important for understanding the potential establish- individuals of the sea anemone Exaiptasia pallida (commonly ment and persistence of novel host-symbiont pairings. Impor- referred to as “Aiptasia”), a model for coral symbiosis, with one tantly, we also provide a baseline for further studies on this of five Symbiodinium species or types (S. microadriaticum, topic with the globally adopted “Aiptasia” model system. S. minutum, phylotype C3, S. trenchii,orS. voratum). The spatial pattern of colonization was monitored over time via Introduction confocal microscopy, and various physiological parameters were Among the most significant marine mutualisms are those measured to assess symbiosis functionality. Anemones rapidly between cnidarians and their photosynthetic dinoflagellate formed a symbiosis with the homologous symbiont, S. minu- symbionts (Roth, 2014). These interactions, in particular, be- tum, but struggled or failed to form a long-lasting symbiosis tween anthozoan cnidarians (e.g., corals and sea anemones) with Symbiodinium C3 or S. voratum, respectively. -

Pattern of Distribution and Adaptation to Different Lrradiance Levels of Zooxanthellae in the Soft Coral Litophyton Arboreum (Octocorallia, Alcyonacea)
Symbiosis, 3 (1987) 23-40 23 Balaban Publishers, Philadelphia/Rehovot Pattern of Distribution and Adaptation to Different lrradiance Levels of Zooxanthellae in the Soft Coral Litophyton arboreum (Octocorallia, Alcyonacea) TAMAR BERNER, YAIR ACHITUV, ZVY DUBINSKY and YEHUDA BENAYAHU1 Department of Life Sciences, Bar-flan University, Ramat-Gan 52100, 1 Department of Zoology, Tel-Aviv University, Ramat-Aoio, Israel Tel. 03-718283 Telex 361311 BARIL IL Received 27 August, 1986; Accepted 2 December 1986 Abstract Photoadaptation responses of endosymbiotic zooxanthellae exposed to differ• ent light intensity within the same colony of the soft coral Litophyton arboreum were studied. Differences in the ultrastructure, including number and total length of sections of thylakoids as compared to chloroplast and cell mem• brane perimeter were measured. Chlorophyll a is 2.5-fold higher in cells from low light intensity as compared with cells taken from high light intensity. Chlorophyll a to c ratio of algae under lower light intensity increases in com• parison to high irradiance. All the results show higher light harvesting capac• ity in terms of ultrastructure and pigment content in the same shade-adapted algae as compared to the light-adapted ones. Keywords: Litophyton, Octocorallia, zooxanthellae, light-shade adaptation, syrn• biosls, ultrastructure, Red Sea 1. Introduction The existence of symbiotic algae within Coelenterata has been recognized as early as 1882 (Brandt, 1882; Entz, 1882) and various aspect of this as• sociation have been extensively studied since then. These studies have been reviewed, among others, by Droop (1963), McLaughlin and Zahl (1966), Trench (1971), Taylor (1973), Muscatine (1974), and Glider and Pardy (1982). -

Best Practice Guidelines in the Development and Maintenance of Regional Marine Species Checklists Version 1.0
www.gbif.org Best Practice Guidelines in the Development and Maintenance of Regional Marine Species Checklists Version 1.0 August 2012 Suggested citation: Nozères, C., Vandepitte, L., Appeltans, W., Kennedy, M. (2012). Best Practice Guidelines in the Development and Maintenance of Regional Marine Species Checklists, version 1.0, released on August 2012. Copenhagen: Global Biodiversity Information Facility, 32 pp, ISBN: 87-92020-46-1, accessible online at http://www.gbif.org/orc/?doc_id=4712 . ISBN: 87-92020-46-1 (10 digits), 978-87-92020-46-8 (13 digits). P e rs is te n t URI: http://www.gbif.org/orc/7doc_idM712 . Language: English. Copyright © Nozères, C., Vandepitte, L., Appeltans, W., Kennedy, M. & Global Biodiversity Information Facility, 2012. This document was produced in collaboration with OBIS Canada/Fisheries and Oceans Canada, EurOBIS/VLIZ and the UNESCO/IOC project office for IODE, Ostend, Belgium. Fisheries and Oceans Pêches et Océans 1 * 1 Canada Canada Flanders Marine Institute L icense: This document is licensed under a Creative Commons Attribution 3.0 Unported License Document Control: Version Description Date of release Author(s) 0.1 First complete draft, released for April 2012 Nozères, Vandepitte, public review. Appeltans & Kennedy 1.0 First public final version of the August 2012 Nozères, Vandepitte, docum ent. Appeltans & Kennedy Cover Art Credit: GBIF Secretariat, 2012. Image by Gytizzz (Lithuania), obtained by stock.xchnghttp://www.sxc.hu/photo/1379825 ( ). ISBN 978879202Q468 9 788792 020468 11 Development and Maintenance of Regional Marine Species Checklists Version 1.0 About GBIF The Global Biodiversity Information Facility (GBIF) was established as a global mega science initiative to address one of the great challenges of the 21st century - harnessing knowledge of the Earth’s biological diversity. -

Title a DISTRIBUTION STUDY of the OCTOCORALLIA OF
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Kyoto University Research Information Repository A DISTRIBUTION STUDY OF THE OCTOCORALLIA OF Title OREGON Author(s) Belcik, Francis P. PUBLICATIONS OF THE SETO MARINE BIOLOGICAL Citation LABORATORY (1977), 24(1-3): 49-52 Issue Date 1977-11-30 URL http://hdl.handle.net/2433/175960 Right Type Departmental Bulletin Paper Textversion publisher Kyoto University A DISTRIBUTION STUDY OF THE OCTOCORALLIA OF OREGON FRANCIS P. BELCIK Department of Biology, East Carolina University, Greenville, North Carolina 27834, U.S.A. With Text-figure 1 and Tables 1-2 Introduction: The purpose of this report was to identify the species of octocorals, note their occurrence or distribution and also their numbers. The Octocorals of this report were collected :rhainly from the Oregonian Region. The majority of specimens were collected by the Oceanography Department of Oregon State University at depths below 86 meters. A few inshore species were collected at various sites along the Oregon Coast (see Fig. 1). Only two species were found in the Intertidal Zone; the bulk of the Octocoral fauna occur offshore in deeper water. Most of the deep water specimens are now deposited in the Oceanography Department of Oregon State University in Corvallis, Oregon. The inshore speci mens have remained in my personal collection. Identification Methods: No references have been published for the soft corals of Oregon; although col lections have possibly been made in the past. Helpful sources for identification, after the standard methods of corrosion, and spicule measurements have been made are: Bayer, 1961; Hickson, 1915; Kiikenthal, 1907, and 1913; Nutting, 1909 and 1912; Utinomi, 1960, 1961, and 1966 and Verrill, 1922. -

CNIDARIA Corals, Medusae, Hydroids, Myxozoans
FOUR Phylum CNIDARIA corals, medusae, hydroids, myxozoans STEPHEN D. CAIRNS, LISA-ANN GERSHWIN, FRED J. BROOK, PHILIP PUGH, ELLIOT W. Dawson, OscaR OcaÑA V., WILLEM VERvooRT, GARY WILLIAMS, JEANETTE E. Watson, DENNIS M. OPREsko, PETER SCHUCHERT, P. MICHAEL HINE, DENNIS P. GORDON, HAMISH J. CAMPBELL, ANTHONY J. WRIGHT, JUAN A. SÁNCHEZ, DAPHNE G. FAUTIN his ancient phylum of mostly marine organisms is best known for its contribution to geomorphological features, forming thousands of square Tkilometres of coral reefs in warm tropical waters. Their fossil remains contribute to some limestones. Cnidarians are also significant components of the plankton, where large medusae – popularly called jellyfish – and colonial forms like Portuguese man-of-war and stringy siphonophores prey on other organisms including small fish. Some of these species are justly feared by humans for their stings, which in some cases can be fatal. Certainly, most New Zealanders will have encountered cnidarians when rambling along beaches and fossicking in rock pools where sea anemones and diminutive bushy hydroids abound. In New Zealand’s fiords and in deeper water on seamounts, black corals and branching gorgonians can form veritable trees five metres high or more. In contrast, inland inhabitants of continental landmasses who have never, or rarely, seen an ocean or visited a seashore can hardly be impressed with the Cnidaria as a phylum – freshwater cnidarians are relatively few, restricted to tiny hydras, the branching hydroid Cordylophora, and rare medusae. Worldwide, there are about 10,000 described species, with perhaps half as many again undescribed. All cnidarians have nettle cells known as nematocysts (or cnidae – from the Greek, knide, a nettle), extraordinarily complex structures that are effectively invaginated coiled tubes within a cell. -

Cytotoxic and HIV-1 Enzyme Inhibitory Activities of Red Sea Marine Organisms Mona S Ellithey1, Namrita Lall2, Ahmed a Hussein3 and Debra Meyer1*
Ellithey et al. BMC Complementary and Alternative Medicine 2014, 14:77 http://www.biomedcentral.com/1472-6882/14/77 RESEARCH ARTICLE Open Access Cytotoxic and HIV-1 enzyme inhibitory activities of Red Sea marine organisms Mona S Ellithey1, Namrita Lall2, Ahmed A Hussein3 and Debra Meyer1* Abstract Background: Cancer and HIV/AIDS are two of the greatest public health and humanitarian challenges facing the world today. Infection with HIV not only weakens the immune system leading to AIDS and increasing the risk of opportunistic infections, but also increases the risk of several types of cancer. The enormous biodiversity of marine habitats is mirrored by the molecular diversity of secondary metabolites found in marine animals, plants and microbes which is why this work was designed to assess the anti-HIV and cytotoxic activities of some marine organisms of the Red Sea. Methods: The lipophilic fractions of methanolic extracts of thirteen marine organisms collected from the Red Sea (Egypt) were screened for cytotoxicity against two human cancer cell lines; leukaemia (U937) and cervical cancer (HeLa) cells. African green monkey kidney cells (Vero) were used as normal non-malignant control cells. The extracts were also tested for their inhibitory activity against HIV-1 enzymes, reverse transcriptase (RT) and protease (PR). Results: Cytotoxicity results showed strong activity of the Cnidarian Litophyton arboreum against U-937 (IC50; 6.5 μg/ml ±2.3) with a selectivity index (SI) of 6.45, while the Cnidarian Sarcophyton trochliophorum showed strong activity against HeLa cells (IC50; 5.2 μg/ml ±1.2) with an SI of 2.09. -

Deep-Sea Coral Taxa in the U.S. Northeast Region: Depth and Geographical Distribution (V
Deep-Sea Coral Taxa in the U.S. Northeast Region: Depth and Geographical Distribution (v. 2020) by David B. Packer1, Martha S. Nizinski2, Stephen D. Cairns3, 4 and Thomas F. Hourigan 1. NOAA Habitat Ecology Branch, Northeast Fisheries Science Center, Sandy Hook, NJ 2. NOAA National Systematics Laboratory Smithsonian Institution, Washington, DC 3. National Museum of Natural History, Smithsonian Institution, Washington, DC 4. NOAA Deep Sea Coral Research and Technology Program, Office of Habitat Conservation, Silver Spring, MD This annex to the U.S. Northeast chapter in “The State of Deep-Sea Coral and Sponge Ecosystems of the United States” provides a revised and updated list of deep-sea coral taxa in the Phylum Cnidaria, Class Anthozoa, known to occur in U.S. waters from Maine to Cape Hatteras (Figure 1). Deep-sea corals are defined as azooxanthellate, heterotrophic coral species occurring in waters 50 meters deep or more. Details are provided on the vertical and geographic extent of each species (Table 1). This list is adapted from Packer et al. (2017) with the addition of new species and range extensions into Northeast U.S. waters reported through 2020, along with a number of species previously not included. No new species have been described from this region since 2017. Taxonomic names are generally those currently accepted in the World Register of Marine Species (WoRMS), and are arranged by order, then alphabetically by family, genus, and species. Data sources (references) listed are those principally used to establish geographic and depth distributions. The total number of distinct deep-sea corals documented for the U.S. -

Deep-Sea Coral Taxa in the U.S. Southeast Region: Depth and Geographic Distribution (V
Deep-Sea Coral Taxa in the U.S. Southeast Region: Depth and Geographic Distribution (v. 2020) by Thomas F. Hourigan1, Stephen D. Cairns2, John K. Reed3, and Steve W. Ross4 1. NOAA Deep Sea Coral Research and Technology Program, Office of Habitat Conservation, Silver Spring, MD 2. National Museum of Natural History, Smithsonian Institution, Washington, DC 3. Cooperative Institute of Ocean Exploration, Research, and Technology, Harbor Branch Oceanographic Institute, Florida Atlantic University, Fort Pierce, FL 4. Center for Marine Science, University of North Carolina, Wilmington This annex to the U.S. Southeast chapter in “The State of Deep-Sea Coral and Sponge Ecosystems in the United States” provides a list of deep-sea coral taxa in the Phylum Cnidaria, Classes Anthozoa and Hydrozoa, known to occur in U.S. waters from Cape Hatteras to the Florida Keys (Figure 1). Deep-sea corals are defined as azooxanthellate, heterotrophic coral species occurring in waters 50 meters deep or more. Details are provided on the vertical and geographic extent of each species (Table 1). This list is an update of the peer-reviewed 2017 list (Hourigan et al. 2017) and includes taxa recognized through 2019, including one newly described species. Taxonomic names are generally those currently accepted in the World Register of Marine Species (WoRMS), and are arranged by order, and alphabetically within order by family, genus, and species. Data sources (references) listed are those principally used to establish geographic and depth distribution. Figure 1. U.S. Southeast region delimiting the geographic boundaries considered in this work. The region extends from Cape Hatteras to the Florida Keys and includes the Jacksonville Lithoherms (JL), Blake Plateau (BP), Oculina Coral Mounds (OC), Miami Terrace (MT), Pourtalès Terrace (PT), Florida Straits (FS), and Agassiz/Tortugas Valleys (AT).