Riverscape-Mediated Effects of Introduced Trout on Non-Diadromous
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Kowai River Gravel Extraction
Screenworks Ltd www.rmps.work FORM 9: APPLICATION FOR RESOURCE CONSENT UNDER SECTION 124 OF THE RESOURCE MANAGEMENT ACT 1991 Kowai River Gravel Extraction To: Environment Canterbury Southern Screenworks Ltd, applies for the resource consent described below: 1. The names and addresses of the owner and occupier (other than the applicant) of any land to which the application relates are as follows: The owner of the land is The Crown that is managed through LINZ – Private Bag 4721 Christchurch Central 8140. 1. The land to which the application relates is: The application relates to the Kowai River. The site is identified as LINZ Primary Parcel 6911718. 2 The type of resource consent sought is as follows: Land Use Consent 3. A description of the activity to which the application relates is: The applicant seeks resource consent for the following activities: ● Extract gravel from the bed of the Kowai River. The subject sites are shown on the plan attached marked Attachment A and within the individual Gravel Availability Advice. 4. The following additional resource consents are required in relation to this proposal and have or have not been applied for: Not applicable. 5. I attach an assessment of any effects that the proposed activity may have on the environment in accordance with section 88 of, and the Fourth Schedule to the Act: N/A 1 Screenworks Ltd www.rmps.work 6. Attach other information (if any) required to be included in the application by the district plan or regional plan or regulations. All information is contained in the application and the plans provided. -
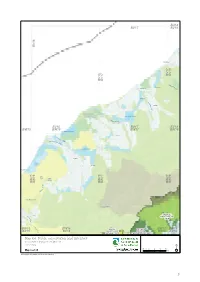
Draft Canterbury CMS 2013 Vol II: Maps
BU18 BV17 BV18 BV16 Donoghues BV17 BV18 BV16 BV17 M ik onu Fergusons i R iv Kakapotahi er Pukekura W a i ta h Waitaha a a R iv e r Lake Ianthe/Matahi W an g anui Rive r BV16 BV17 BV18 BW15 BW16 BW17 BW18 Saltwater Lagoon Herepo W ha ta ro a Ri aitangi ver W taon a R ive r Lake Rotokino Rotokino Ōkārito Lagoon Te Taho Ōkārito The Forks Lake Wahapo BW15 BW16 BW16 BW17 BW17 BW18 r e v i R to ri kā Ō Lake Mapourika Perth River Tatare HAKATERE W ai CONSERVATION h o R PARK i v e r C a l le r y BW15 R BW16 AORAKI TE KAHUI BW17 BW18 iv BX15 e BX16 MOUNT COOK KAUPEKA BX17 BX18 r NATIONAL PARK CONSERVATION PARK Map 6.6 Public conservation land inventory Conservation Management Strategy Canterbury 01 2 4 6 8 Map 6 of 24 Km Conservation unit data is current as of 21/12/2012 51 Public conservation land inventory Canterbury Map table 6.7 Conservation Conservation Unit Name Legal Status Conservation Legal Description Description Unit number Unit Area I35028 Adams Wilderness Area CAWL 7143.0 Wilderness Area - s.20 Conservation Act 1987 - J35001 Rangitata/Rakaia Head Waters Conservation Area CAST 53959.6 Stewardship Area - s.25 Conservation Act 1987 Priority ecosystem J35002 Rakaia Forest Conservation Area CAST 4891.6 Stewardship Area - s.25 Conservation Act 1987 Priority ecosystem J35007 Marginal Strip - Double Hill CAMSM 19.8 Moveable Marginal Strip - s.24(1) & (2) Conservation Act 1987 - J35009 Local Purpose Reserve Public Utility Lake Stream RALP 0.5 Local Purpose Reserve - s.23 Reserves Act 1977 - K34001 Central Southern Alps Wilberforce Conservation -

Late Pleistocene Geology of the Kowai River Valley, Mid-Canterbury
LATE PLEISTOCENE GEOLOGY OF THE KOWAl RIVER VALLEY MID-CANTERBURY A thesis submitted. in partial fulfilment of the requirements for the degree of Master of Science in Geology iD; the University of Canterbury by Michael Marden 5 University of Canterbury 1976 THl:SlS WcJt, 5 ABSTRACT ; The Kowai River Valley, a formerly glaciated -tributary of the Waimakariri River, drains the eastern side of the Torlesse Range in the vicinity of Porter's Pass. -. A study of the glacial deposits and stratigraphy was made, investigating three specific but complimentary aspects:· - · ( 1) Mapping of the glacial stratigraphic succession for the purpose of correlation.with that established for the Waimakariri River System (Gage, 1958) ; (2) a detailed comparative textural analysis of outwash gravels from :two glacial advances to determine whether an·y significant differences exist, that can be directly related to environmental processes operating during transportation and deposition of these materials~ - (3) a clay mineral study of weathering products as a potential tool for age determination-and correlation. From logitudinal terrace profiles, a _distinct ·surface and moraine system was found for each of the Woodstock, Otarama, Blackwater I and II and Poulter Glacial Advances, from which their magnitude and maximum extension were interpreted. The stratigraphy and glacial history of the Kawai . River reflects that of the Waimakariri River very closely, but, several. anomalous topographical features indicate that-the Kawai Valley traverses a zone of active tectonism. The principal sites of movement were identified and something of the tectonic history was deduced. Parameters measured for the gravel analysis included size distribution, composition, ·sphericity and shape factor. -

Canterbury Railways: Full Steam Ahead the Provincial Railways of Canterbury, 1863-76
Canterbury Railways: Full Steam Ahead The Provincial Railways of Canterbury, 1863-76 A thesis submitted in partial fulfilment of the requirements for the Degree of Master of Arts in History in the University of Canterbury by Alastair Adrian Cross University of Canterbury 2017 Abstract The broad-gauge Canterbury Railways are considered unanimously by New Zealand historians as the origins of the modern-day railway network in New Zealand. Built by the Canterbury Provincial Government in 1863 to relieve transport issues between Christchurch and Lyttelton, the broad-gauge railway later expanded to reach Amberley in the north and Rakaia in the south, opening up the Canterbury Plains and stimulating trade and immigration. Brought under the control of the Public Works Department in 1876 along with several narrow-gauge lines built by the Provincial Government, the broad-gauge was converted to the New Zealand standard narrow-gauge in 1878 and the locomotives and rolling-stock were sold to the South Australian Railways. Unfortunately, there has been little engagement with the history of the Canterbury Railways in the last fifty years and in particular with the primary sources from the period since the publication in 1964 of W. A. Pierre’s book Canterbury Provincial Railways: Genesis of the NZR. The majority of what has been written in this timeframe has been for the railway enthusiast market, and therefore has contributed to the marginalisation of the part played by the Canterbury Railways in the context of the wider New Zealand history. By engaging with period primary sources held by Archives New Zealand and suitably supported with selected secondary sources, this thesis aims to recover this history within an academic framework considering, among other themes, the prehistory of the railway before 1863, the operation of the CR network and comparisons with other Provincial-era railway operations within this period. -

The Christchurch Tramper
TTHEHE CCHRISTCHURCHHRISTCHURCH TTRAMPERRAMPER Published by CHRISTCHURCH TRAMPING CLUB INC PO Box 527, Christchurch. www.ctc.org.nz Affiliated with the Federated Mountain Clubs of NZ Inc. Any similarity between the opinions expressed in this newsletter and Club policy is purely coincidental. Vol. 83 August 2013 No. 4 The CHRISTCHURCH TRAMPING CLUB has members of all ages, and runs tramping trips every weekend, ranging from easy (minimal experience required) to hard (high fitness and experience required). We also organise instructional courses and hold weekly social meetings. We have a club hut in Arthurs Pass and have gear available for hire to members. Membership rates per year are $45 member, $65 couple, $25 junior or associate, with a $5 discount for members Queuing up the summit ridge who opt to obtain this newsletter electronically. (from Barrosa trip) For more about how the club operates, see the last two pages. News New Members Welcome: We welcome Eleven new members to the club this month; Winsome Brown, Mathew Tracy, Gareth Pritchard, Conrad Dekker, Chris McGimpsey, Pablo Soares De Oliveira, David Rowley, Nikky Kear, Joshua Johnson, Charlie Lane and Guillaume Clin. Please accept the club's warm welcome to you all. Annual Mid Winter Breakfast Report: We had 30 odd (and a couple of not so odd) people at the annual Mid Winter Breakfast and subsequent LooLoo of the year award. Which is to vote for the person who has done the silliest thing during the year. The nominations were. 1, Bryce Williamson for leaving his raincoat behind on a cold wet and windy winter day trip. -

Proposed Plan Change 2 to the Waimakariri River Regional Plan
Proposed Plan Change 2 to the Waimakariri River Regional Plan R19/26-1 Environment Canterbury Contents Section Page How to Read Plan Change 7 to the Canterbury Land and Water Regional Plan 1 Section 1 Introduction 3 Section 3 Resource Overview 7 Section 5 Water Quantity Introduction 16 Section 6 Water Quality 25 Section 7 River and Lake Beds 30 Section 9 Monitoring and Review 32 Appendix 1 – Definition of Terms 37 Appendix 3 – Overview of the Main Waimakariri River Catchment Aquatic Values 38 How to read Proposed Plan Change 2 to the Waimakariri River Regional Plan Introduction This document is included for information purposes only and does not form part of proposed Plan Change 2 (PC2) to the Waimakariri River Regional Plan (WRRP). The following sections contain: Information for the Reader This section contains general information about proposed PC2 to the WRRP. It provides an overview of key sections in the WRRP that are proposed to be amended. Readers should refer to the plan change itself, for a full description of all changes proposed by PC2. Legal effect of rules in Plan Change 2 This section describes the legal status of rules in the proposed plan change. How proposed amendments to the WRRP are shown This section sets out how changes (ie additions, deletions and amendments) proposed by part of PC2 are indicated in the document. Relationship between proposed Plan Change 2 to the Waimakariri River Regional Plan and proposed Plan Change 7 to the Canterbury Land and Water Regional Plan This section sets out the relationship between proposed PC2 to the WRRP and proposed PC7 to the LWRP. -

Hydrology and Stream Sediments in a Mountain Catchment
Lincoln University Digital Thesis Copyright Statement The digital copy of this thesis is protected by the Copyright Act 1994 (New Zealand). This thesis may be consulted by you, provided you comply with the provisions of the Act and the following conditions of use: you will use the copy only for the purposes of research or private study you will recognise the author's right to be identified as the author of the thesis and due acknowledgement will be made to the author where appropriate you will obtain the author's permission before publishing any material from the thesis. HYDROLOGY AND STREAM SEDIMENTS IN A MOUNTAIN CATCHMENT VOLUME I (of three volumes) A SUMMARY A dissertation presented to the University of Canterbury in fulfilment of the requirements for the degree Doctor of Philosophy. JOHN A. HAYWARD 1978 HYDROLOGY AND STREAM SEDIMENTS IN A MOUNTAIN CATCHMENT VOLUME I (being Volume I of three volumes) A SUMMARY page nos. ACKNOWLEDGMENTS CHAPTER 1: 1970. The status of knowledge about hydrology, erosion and sediments in New Zealand mountain 1ands. 6 CHAPTER 2: A summary account of studies in the Torlesse stream catchment 1972 - 1977. 15 CHAPTER 3: The floods of April 1378: An example to illustrate the findings of this study. 25 CHAPTER 4: The implications for future management. 42 REFERENCES: 54 LIST OF FIGURES, VOLUME I Fo llows page No. 1. Extracts from the Christchurch Press and Christchurch Star, 1940 - 1943. 8 2. Extracts from Campbell (undated). 11 ll 3. Extract from "The Weekly News • 29 May 1963. 11 4. The Torlesse stream catchment. -

Proposed Additions to Korowai/Torlesse Tussocklands Park
Proposal to Add Land to Korowai/Torlesse Tussocklands Park Purpose This document provides information enabling the public to comment on a Department proposal to add approximately 3282 hectares of stewardship land (held under section 25 of the Conservation Act 1987) to Korowai/Torlesse Tussocklands (Conservation) Park. The land proposed for addition to the Park is numbered on the map below and described in the table on page 2. Context The Minister of Conservation invites any person or organisation to provide to the Department of Conservation’s Director-General written objections or submissions on the proposal. The Department shall take these objections or submissions into account when providing a department briefing to the Minister seeking a decision. Relevant land Location Map Areas proposed for inclusion into the park are coloured red and are numbered to correspond with Table 1 on page 2. 1 DOC-5439111 Table 1: Relevant Land Proposed to become Korowai/Torlesse Tussocklands Park Location Important Conservation Values Identifying Legal Area Map Name Description (hectares) Identifier • Landscapes of the western Torlesse Range (refer Photos 1-6). (Part) • Rock outcrops and bluffs on upper Section 7 Conservation slopes, with inaka (Dracophyllum) SO 387006 Area - Castle shrubland and mountain beech 1 3250.8000 Hill forest together below • Mid- higher altitudes contain: Slim Snow-Tussock Grassland; Montane rockland; Montane scree and alpine plant communities. • A small part of a regionally Conservation significant natural the landscape on Section 12 Area - Ben 2 29.6550 the eastern Big Ben Range with SO 378671 More mountain beech and tussocklands (Photo 7). • A very small part of a regionally Conservation significant natural landscape being RS 41336; Area - Little 3 1.4280 the eastern Torlesse Range. -

List of Rivers of New Zealand
Sl. No River Name 1 Aan River 2 Acheron River (Canterbury) 3 Acheron River (Marlborough) 4 Ada River 5 Adams River 6 Ahaura River 7 Ahuriri River 8 Ahuroa River 9 Akatarawa River 10 Akitio River 11 Alexander River 12 Alfred River 13 Allen River 14 Alma River 15 Alph River (Ross Dependency) 16 Anatoki River 17 Anatori River 18 Anaweka River 19 Anne River 20 Anti Crow River 21 Aongatete River 22 Aorangiwai River 23 Aorere River 24 Aparima River 25 Arahura River 26 Arapaoa River 27 Araparera River 28 Arawhata River 29 Arnold River 30 Arnst River 31 Aropaoanui River 32 Arrow River 33 Arthur River 34 Ashburton River / Hakatere 35 Ashley River / Rakahuri 36 Avoca River (Canterbury) 37 Avoca River (Hawke's Bay) 38 Avon River (Canterbury) 39 Avon River (Marlborough) 40 Awakari River 41 Awakino River 42 Awanui River 43 Awarau River 44 Awaroa River 45 Awarua River (Northland) 46 Awarua River (Southland) 47 Awatere River 48 Awatere River (Gisborne) 49 Awhea River 50 Balfour River www.downloadexcelfiles.com 51 Barlow River 52 Barn River 53 Barrier River 54 Baton River 55 Bealey River 56 Beaumont River 57 Beautiful River 58 Bettne River 59 Big Hohonu River 60 Big River (Southland) 61 Big River (Tasman) 62 Big River (West Coast, New Zealand) 63 Big Wainihinihi River 64 Blackwater River 65 Blairich River 66 Blind River 67 Blind River 68 Blue Duck River 69 Blue Grey River 70 Blue River 71 Bluff River 72 Blythe River 73 Bonar River 74 Boulder River 75 Bowen River 76 Boyle River 77 Branch River 78 Broken River 79 Brown Grey River 80 Brown River 81 Buller -

New Zealand Gazet1,E
jumb. 56. 2237 SUPPLEMENT TO THE NEW ZEALAND GAZET1,E OF THUBSDAY, JULY 19, 1934. WELLINGTON, THURSDAY, JULY 19, 1934. Tenders for Inland Mail-service Contracts. -South Island, 12. Havelock, Brightlands, Elaine Ray, Fitzroy Bay, Te 193.5~~7. Towaka, Waitata Bay, Bulwer, Port Ligar, Forsyth Island, Wakatahuri, Te Puru, Okoha, Anakoha, General Post Office, Titirangi, Pohuenui, Waimaru, Clova Bay, Manaroa, Wellington, 19th July, 1934. Hopai, Eli Bay, Crail Ray, Taranui, Homewood, and EALED tenders will be received at the several Chief Havelock (part private-bag delivery), twice weekly S Post-offices in the South Island until noon on Wednes (except Te Towaka, and Fitzroy Bay, weekly). day, the 29th August, 1934, for the conveyance of mails (Alternative to No. 13.) between the undermentioned places, for a period of THREE 13. Havelock, Brightlands, Elaine Bay, Fitzroy Bay, Te YEARS, from the 1st January, 1935 :- Towaka, Waitata Bay, Bulwer, Port Ligar, Forsyth Island, Whakatahuri, Te Puru, Okoha, Anakoha, PoSTAL DISTRICT OF BLENHEIM. Titirangi, Pohuenui, Beatrix Bay, Waimaru, Clova Bay, Manaroa, Hopai, Eli Bay, Crail Bay, Taranui, l. (a) Blenheim, Jordan, Upcot, and Molesworth, weekly. Homewood, and Havelock (part private-bag delivery), (b) Blenheim, Jordan, and Upcot (part private-bag de twice weekly (except Te Towaka, and Fitzroy Bay, livery), weekly. weekly). (Alternative to No. 12.) 2. (a) Blenheim, Netherwood, and Te Arowhenua, twice 14. Havelock, Mahakipawa, Grove, and Grove Wharf, thrice weekly. weekly. (Alternative to No. 10.) 15. Havelock, Whatanihi, Palmer's, :Maungarimu, Pinohia, (b) Rlenheim,"Bcnhopai, Power-station, and Malvern (pri· :1-laori Bay, J. H. Wells, Nydia Bay, Roadley's, Hamil vate-bag delivery), weekly. -
Paleoseismic Analysis of the Springfield Fault, Central Canterbury
PALEOSEISMIC ANALYSIS OF THE SPRINGFIELD FAULT, CENTRAL CANTERBURY A thesis submitted in partial fulfillment of the requirements for the degree of Bachelor of Science with Honors at the University of Canterbury by STEWART THOMAS EVANS University of Canterbury, JULY2000 ABSTRACT The Springfield Fault is a northeast striking thrust fault bisecting the Russell Range at the northern end of the Malvern Hills near Springfield. The trace of the fault is best expressed by offset river terraces, as it cuts across the flood plane of the Hawkins River in the Upper Hawkins Basin. The Russell Range is composed chiefly of the basement Triassic rocks of the Torlesse tenane with Cretaceous to Cenozoic rocks occurring in a fault bounded slice and around the hinge of the hanging wall anticline of the fault. Quaternary glacial sediments occur on the plains adjacent to the Russell Range with Holocene deposits occurring in the Upper Hawkins Basin. Folding in the highly sheared basement is along NW-SE trending fold axes. The current direction of principal shortening was determined from slickenside data to be NW-SE to NE-SW. Geomorphology of the area in the Upper Hawkins Basin provided a chronology of the activity on the Springfield Fault. Offset of terrace surfaces provide evidence for the number of fault ruptures, which is at least two. An attempt to date the age of fault and river tenace scarps was made using their morphology and a diffusion model of scarp degradation. Analysis requires the diffusivity rate operating on a scarp to be known to produce an absolute age for the scarp. -

Angler Usage of Lake and River Fisheries Managed by Fish & Game
Angler usage of lake and river fisheries managed by Fish & Game New Zealand: results from the 2001/02 National Angling Survey NIWA Client Report: CHC2003-114 December 2003 NIWA Project: NZF03501 Angler usage of lake and river fisheries managed by Fish & Game New Zealand: results from the 2001/02 National Angling Survey Martin Unwin Katie Image Prepared for Fish & Game New Zealand NIWA Client Report: CHC2003- 114 December 2003 NIWA Project: NZF03501 National Institute of Water & Atmospheric Research Ltd 10 Kyle Street, Riccarton, Christchurch P O Box 8602, Christchurch, New Zealand Phone +64-3-348 8987, Fax +64-3-348 5548 www.niwa.co.nz All rights reserved. This publication may not be reproduced or copied in any form, without the permission of the client. Such permission is to be given only in accordance with the terms of the client’s contract with NIWA. This copyright extends to all forms of copying and any storage of material in any kind of information retrieval system. Contents Summary i 1. Introduction 3 1.1. Freshwater angling in New Zealand 3 2. Survey design and implementation 5 2.1. Scope, format, and objectives 5 2.2. Sampling design 7 2.2.1. Licence types and strata 7 2.2.2. Survey population 9 2.2.3. Sample sizes 9 2.2.4. Interview procedures 13 2.3. Data analysis 16 3. Results 17 3.1. Licence database 17 3.2. The replies 22 3.3. Usage estimates 22 3.3.1. Cross-boundary usage 24 3.3.2. Multi-reach rivers 26 3.3.3.