Chapter 6: Elasticity (Pdf)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
![Arxiv:1910.01953V1 [Cond-Mat.Soft] 4 Oct 2019 Tion of the Load](https://docslib.b-cdn.net/cover/4322/arxiv-1910-01953v1-cond-mat-soft-4-oct-2019-tion-of-the-load-104322.webp)
Arxiv:1910.01953V1 [Cond-Mat.Soft] 4 Oct 2019 Tion of the Load
Geometric charges and nonlinear elasticity of soft metamaterials Yohai Bar-Sinai,1 Gabriele Librandi,1 Katia Bertoldi,1 and Michael Moshe2, ∗ 1School of Engineering and Applied Sciences, Harvard University, Cambridge MA 02138 2Racah Institute of Physics, The Hebrew University of Jerusalem, Jerusalem, Israel 91904 Problems of flexible mechanical metamaterials, and highly deformable porous solids in general, are rich and complex due to nonlinear mechanics and nontrivial geometrical effects. While numeric approaches are successful, analytic tools and conceptual frameworks are largely lacking. Using an analogy with electrostatics, and building on recent developments in a nonlinear geometric formu- lation of elasticity, we develop a formalism that maps the elastic problem into that of nonlinear interaction of elastic charges. This approach offers an intuitive conceptual framework, qualitatively explaining the linear response, the onset of mechanical instability and aspects of the post-instability state. Apart from intuition, the formalism also quantitatively reproduces full numeric simulations of several prototypical structures. Possible applications of the tools developed in this work for the study of ordered and disordered porous mechanical metamaterials are discussed. I. INTRODUCTION tic metamaterials out of an underlying nonlinear theory of elasticity. The hallmark of condensed matter physics, as de- A theoretical analysis of the elastic problem requires scribed by P.W. Anderson in his paper \More is Dif- solving the nonlinear equations of elasticity while satis- ferent" [1], is the emergence of collective phenomena out fying the multiple free boundary conditions on the holes of well understood simple interactions between material edges - a seemingly hopeless task from an analytic per- elements. Within the ever increasing list of such systems, spective. -

Application of Visco-Hyperelastic Devices in Structural Response Control
Application of visco-hyperelastic devices in structural response control Anantha Narayan Chittur Krishna Murthy Thesis submitted to the faculty of the Virginia Polytechnic Institute and State University in partial fulfillment of the requirements for the degree of Master of Science In Civil Engineering Dr. Finley A. Charney Dr. Raymond H. Plaut Dr. Carin L. Roberts Wollmann 11 May 2005 Blacksburg, Virginia Keywords: Visco-hyperelastic, Viscoelastic, dampers, Passive energy systems, Earthquakes, Seismic, Tires Application of visco-hyperelastic devices in structural response control (Anantha Narayan Chittur Krishna Murthy) Abstract Structural engineering has progressed from design for life safety limit states to performance based engineering, in which energy dissipation systems in structural frameworks assume prime importance. A visco-hyperelastic device is a completely new type of passive energy dissipation system that not only combines the energy dissipation properties of velocity and displacement dependent devices but also provides additional stability to the structure precluding overall collapse. The device consists of a viscoelastic material placed between two steel rings. The energy dissipation in the device is due to a combination of viscoelastic dissipation from rubber and plastic dissipation due to inelastic behavior of the steel elements. The device performs well under various levels of excitation, providing an excellent means of energy dissipation. The device properties are fully controlled through modifiable parameters. An initial study was conducted on motorcycle tires to evaluate the hyperelastic behavior and energy dissipation potential of circular rubber elements, which was preceded by preliminary finite element modeling. The rubber tires provided considerable energy dissipation while displaying a nonlinear stiffening behavior. The proposed device was then developed to provide additional stiffness that was found lacking in rubber tires. -
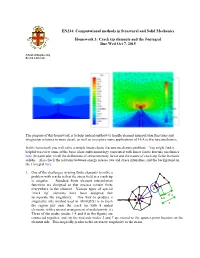
Crack Tip Elements and the J Integral
EN234: Computational methods in Structural and Solid Mechanics Homework 3: Crack tip elements and the J-integral Due Wed Oct 7, 2015 School of Engineering Brown University The purpose of this homework is to help understand how to handle element interpolation functions and integration schemes in more detail, as well as to explore some applications of FEA to fracture mechanics. In this homework you will solve a simple linear elastic fracture mechanics problem. You might find it helpful to review some of the basic ideas and terminology associated with linear elastic fracture mechanics here (in particular, recall the definitions of stress intensity factor and the nature of crack-tip fields in elastic solids). Also check the relations between energy release rate and stress intensities, and the background on the J integral here. 1. One of the challenges in using finite elements to solve a problem with cracks is that the stress field at a crack tip is singular. Standard finite element interpolation functions are designed so that stresses remain finite a everywhere in the element. Various types of special b c ‘crack tip’ elements have been designed that 3L/4 incorporate the singularity. One way to produce a L/4 singularity (the method used in ABAQUS) is to mesh L the region just near the crack tip with 8 noded elements, with a special arrangement of nodal points: (i) Three of the nodes (nodes 1,4 and 8 in the figure) are connected together, and (ii) the mid-side nodes 2 and 7 are moved to the quarter-point location on the element side. -

Linear Elasticity
10 Linear elasticity When you bend a stick the reaction grows noticeably stronger the further you go — until it perhaps breaks with a snap. If you release the bending force before it breaks, the stick straightens out again and you can bend it again and again without it changing its reaction or its shape. That is elasticity. Robert Hooke (1635–1703). In elementary mechanics the elasticity of a spring is expressed by Hooke’s law English physicist. Worked on elasticity, built tele- which says that the force necessary to stretch or compress a spring is propor- scopes, and the discovered tional to how much it is stretched or compressed. In continuous elastic materials diffraction of light. The Hooke’s law implies that stress is proportional to strain. Some materials that we famous law which bears his name is from 1660. usually think of as highly elastic, for example rubber, do not obey Hooke’s law He stated already in 1678 except under very small deformation. When stresses grow large, most materials the inverse square law for gravity, over which he deform more than predicted by Hooke’s law. The proper treatment of non-linear got involved in a bitter elasticity goes far beyond the simple linear elasticity which we shall discuss in controversy with Newton. this book. The elastic properties of continuous materials are determined by the under- lying molecular structure, but the relation between material properties and the molecular structure and arrangement in solids is complicated, to say the least. Luckily, there are broad classes of materials that may be described by a few Thomas Young (1773–1829). -

Method for the Unique Identification of Hyperelastic Material Properties Using Full-Field Measures
UCLA UCLA Previously Published Works Title Method for the unique identification of hyperelastic material properties using full-field measures. Application to the passive myocardium material response. Permalink https://escholarship.org/uc/item/1vs1s3jk Journal International journal for numerical methods in biomedical engineering, 33(11) ISSN 2040-7939 Authors Perotti, Luigi E Ponnaluri, Aditya VS Krishnamoorthi, Shankarjee et al. Publication Date 2017-11-01 DOI 10.1002/cnm.2866 Peer reviewed eScholarship.org Powered by the California Digital Library University of California Method for the unique identification of hyperelastic material properties using full field measures. Application to the passive myocardium material response Luigi E. Perotti1∗, Aditya V. Ponnaluri2, Shankarjee Krishnamoorthi2, Daniel Balzani3, Daniel B. Ennis1, William S. Klug2 1 Department of Radiological Sciences and Department of Bioengineering, University of California, Los Angeles 2 Department of Mechanical and Aerospace Engineering, University of California, Los Angeles 3 Institute of Mechanics and Shell Structures, TU Dresden, Germany, and Dresden Center of Computational Material Science SUMMARY Quantitative measurement of the material properties (e.g., stiffness) of biological tissues is poised to become a powerful diagnostic tool. There are currently several methods in the literature to estimating material stiffness and we extend this work by formulating a framework that leads to uniquely identified material properties. We design an approach to work with full field displacement data — i.e., we assume the displacement field due to the applied forces is known both on the boundaries and also within the interior of the body of interest — and seek stiffness parameters that lead to balanced internal and external forces in a model. -

Learning Corrections for Hyperelastic Models from Data David González, Francisco Chinesta, Elías Cueto
Learning corrections for hyperelastic models from data David González, Francisco Chinesta, Elías Cueto To cite this version: David González, Francisco Chinesta, Elías Cueto. Learning corrections for hyperelastic models from data. Frontiers in Materials. Computational Materials Science section, Frontiers, 2019, 6, 10.3389/fmats.2019.00014. hal-02165295v1 HAL Id: hal-02165295 https://hal.archives-ouvertes.fr/hal-02165295v1 Submitted on 25 Jun 2019 (v1), last revised 3 Jul 2019 (v2) HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Learning Corrections for Hyperelastic Models From Data David González 1, Francisco Chinesta 2 and Elías Cueto 1* 1 Aragon Institute of Engineering Research, Universidad de Zaragoza, Zaragoza, Spain, 2 ESI Group Chair and PIMM Lab, ENSAM ParisTech, Paris, France Unveiling physical laws from data is seen as the ultimate sign of human intelligence. While there is a growing interest in this sense around the machine learning community, some recent works have attempted to simply substitute physical laws by data. We believe that getting rid of centuries of scientific knowledge is simply nonsense. There are models whose validity and usefulness is out of any doubt, so try to substitute them by data seems to be a waste of knowledge. -

Linear Elastostatics
6.161.8 LINEAR ELASTOSTATICS J.R.Barber Department of Mechanical Engineering, University of Michigan, USA Keywords Linear elasticity, Hooke’s law, stress functions, uniqueness, existence, variational methods, boundary- value problems, singularities, dislocations, asymptotic fields, anisotropic materials. Contents 1. Introduction 1.1. Notation for position, displacement and strain 1.2. Rigid-body displacement 1.3. Strain, rotation and dilatation 1.4. Compatibility of strain 2. Traction and stress 2.1. Equilibrium of stresses 3. Transformation of coordinates 4. Hooke’s law 4.1. Equilibrium equations in terms of displacements 5. Loading and boundary conditions 5.1. Saint-Venant’s principle 5.1.1. Weak boundary conditions 5.2. Body force 5.3. Thermal expansion, transformation strains and initial stress 6. Strain energy and variational methods 6.1. Potential energy of the external forces 6.2. Theorem of minimum total potential energy 6.2.1. Rayleigh-Ritz approximations and the finite element method 6.3. Castigliano’s second theorem 6.4. Betti’s reciprocal theorem 6.4.1. Applications of Betti’s theorem 6.5. Uniqueness and existence of solution 6.5.1. Singularities 7. Two-dimensional problems 7.1. Plane stress 7.2. Airy stress function 7.2.1. Airy function in polar coordinates 7.3. Complex variable formulation 7.3.1. Boundary tractions 7.3.2. Laurant series and conformal mapping 7.4. Antiplane problems 8. Solution of boundary-value problems 1 8.1. The corrective problem 8.2. The Saint-Venant problem 9. The prismatic bar under shear and torsion 9.1. Torsion 9.1.1. Multiply-connected bodies 9.2. -

Gent Models for the Inflation of Spherical Balloons
International Journal of Non-Linear Mechanics 68 (2015) 52–58 Contents lists available at ScienceDirect International Journal of Non-Linear Mechanics journal homepage: www.elsevier.com/locate/nlm Gent models for the inflation of spherical balloons$ Robert Mangan a,n, Michel Destrade a,b a School of Mathematics, Statistics and Applied Mathematics, National University of Ireland Galway, University Road, Galway, Ireland b School of Mechanical and Materials Engineering, University College Dublin, Belfield, Dublin 4, Ireland article info abstract Article history: We revisit an iconic deformation of non-linear elasticity: the inflation of a rubber spherical thin shell. We Received 19 April 2014 use the 3-parameter Mooney and Gent-Gent (GG) phenomenological models to explain the stretch– Received in revised form strain curve of a typical inflation, as these two models cover a wide spectrum of known models for 26 May 2014 rubber, including the Varga, Mooney–Rivlin, one-term Ogden, Gent-Thomas and Gent models. We find Accepted 26 May 2014 that the basic physics of inflation exclude the Varga, one-term Ogden and Gent-Thomas models. We find Available online 25 June 2014 the link between the exact solution of non-linear elasticity and the membrane and Young–Laplace Keywords: theories often used a priori in the literature. We compare the performance of both models on fitting the Spherical shells data for experiments on rubber balloons and animal bladder. We conclude that the GG model is the most Membrane theory accurate and versatile model on offer for the modelling of rubber balloon inflation. Rubber modelling & 2014 Elsevier Ltd. -

20. Rheology & Linear Elasticity
20. Rheology & Linear Elasticity I Main Topics A Rheology: Macroscopic deformation behavior B Linear elasticity for homogeneous isotropic materials 10/29/18 GG303 1 20. Rheology & Linear Elasticity Viscous (fluid) Behavior http://manoa.hawaii.edu/graduate/content/slide-lava 10/29/18 GG303 2 20. Rheology & Linear Elasticity Ductile (plastic) Behavior http://www.hilo.hawaii.edu/~csav/gallery/scientists/LavaHammerL.jpg http://hvo.wr.usgs.gov/kilauea/update/images.html 10/29/18 GG303 3 http://upload.wikimedia.org/wikipedia/commons/8/89/Ropy_pahoehoe.jpg 20. Rheology & Linear Elasticity Elastic Behavior https://thegeosphere.pbworks.com/w/page/24663884/Sumatra http://www.earth.ox.ac.uk/__Data/assets/image/0006/3021/seismic_hammer.jpg 10/29/18 GG303 4 20. Rheology & Linear Elasticity Brittle Behavior (fracture) 10/29/18 GG303 5 http://upload.wikimedia.org/wikipedia/commons/8/89/Ropy_pahoehoe.jpg 20. Rheology & Linear Elasticity II Rheology: Macroscopic deformation behavior A Elasticity 1 Deformation is reversible when load is removed 2 Stress (σ) is related to strain (ε) 3 Deformation is not time dependent if load is constant 4 Examples: Seismic (acoustic) waves, http://www.fordogtrainers.com rubber ball 10/29/18 GG303 6 20. Rheology & Linear Elasticity II Rheology: Macroscopic deformation behavior A Elasticity 1 Deformation is reversible when load is removed 2 Stress (σ) is related to strain (ε) 3 Deformation is not time dependent if load is constant 4 Examples: Seismic (acoustic) waves, rubber ball 10/29/18 GG303 7 20. Rheology & Linear Elasticity II Rheology: Macroscopic deformation behavior B Viscosity 1 Deformation is irreversible when load is removed 2 Stress (σ) is related to strain rate (ε ! ) 3 Deformation is time dependent if load is constant 4 Examples: Lava flows, corn syrup http://wholefoodrecipes.net 10/29/18 GG303 8 20. -

Design of a Meta-Material with Targeted Nonlinear Deformation Response Zachary Satterfield Clemson University, [email protected]
Clemson University TigerPrints All Theses Theses 12-2015 Design of a Meta-Material with Targeted Nonlinear Deformation Response Zachary Satterfield Clemson University, [email protected] Follow this and additional works at: https://tigerprints.clemson.edu/all_theses Part of the Mechanical Engineering Commons Recommended Citation Satterfield, Zachary, "Design of a Meta-Material with Targeted Nonlinear Deformation Response" (2015). All Theses. 2245. https://tigerprints.clemson.edu/all_theses/2245 This Thesis is brought to you for free and open access by the Theses at TigerPrints. It has been accepted for inclusion in All Theses by an authorized administrator of TigerPrints. For more information, please contact [email protected]. DESIGN OF A META-MATERIAL WITH TARGETED NONLINEAR DEFORMATION RESPONSE A Thesis Presented to the Graduate School of Clemson University In Partial Fulfillment of the Requirements for the Degree Master of Science Mechanical Engineering by Zachary Tyler Satterfield December 2015 Accepted by: Dr. Georges Fadel, Committee Chair Dr. Nicole Coutris, Committee Member Dr. Gang Li, Committee Member ABSTRACT The M1 Abrams tank contains track pads consist of a high density rubber. This rubber fails prematurely due to heat buildup caused by the hysteretic nature of elastomers. It is therefore desired to replace this elastomer by a meta-material that has equivalent nonlinear deformation characteristics without this primary failure mode. A meta-material is an artificial material in the form of a periodic structure that exhibits behavior that differs from its constitutive material. After a thorough literature review, topology optimization was found as the only method used to design meta-materials. Further investigation determined topology optimization as an infeasible method to design meta-materials with the targeted nonlinear deformation characteristics. -

Geometric Charges and Nonlinear Elasticity of Two-Dimensional Elastic Metamaterials
Geometric charges and nonlinear elasticity of two-dimensional elastic metamaterials Yohai Bar-Sinai ( )a , Gabriele Librandia , Katia Bertoldia, and Michael Moshe ( )b,1 aSchool of Engineering and Applied Sciences, Harvard University, Cambridge, MA 02138; and bRacah Institute of Physics, The Hebrew University of Jerusalem, Jerusalem, Israel 91904 Edited by John A. Rogers, Northwestern University, Evanston, IL, and approved March 23, 2020 (received for review November 17, 2019) Problems of flexible mechanical metamaterials, and highly A theoretical analysis of the elastic problem requires solv- deformable porous solids in general, are rich and complex due ing the nonlinear equations of elasticity while satisfying the to their nonlinear mechanics and the presence of nontrivial geo- multiple free boundary conditions on the holes’ edges—a seem- metrical effects. While numeric approaches are successful, ana- ingly hopeless task from an analytic perspective. However, direct lytic tools and conceptual frameworks are largely lacking. Using solutions of the fully nonlinear elastic equations are accessi- an analogy with electrostatics, and building on recent devel- ble using finite-element models, which accurately reproduce the opments in a nonlinear geometric formulation of elasticity, we deformation fields, the critical strain, and the effective elastic develop a formalism that maps the two-dimensional (2D) elas- coefficients, etc. (6). The success of finite-element (FE) simula- tic problem into that of nonlinear interaction of elastic charges. tions in predicting the mechanics of perforated elastic materials This approach offers an intuitive conceptual framework, qual- confirms that nonlinear elasticity theory is a valid description, itatively explaining the linear response, the onset of mechan- but emphasizes the lack of insightful analytical solutions to ical instability, and aspects of the postinstability state. -

On the Path-Dependence of the J-Integral Near a Stationary Crack in an Elastic-Plastic Material
On the Path-Dependence of the J-integral Near a Stationary Crack in an Elastic-Plastic Material Dorinamaria Carka and Chad M. Landis∗ The University of Texas at Austin, Department of Aerospace Engineering and Engineering Mechanics, 210 East 24th Street, C0600, Austin, TX 78712-0235 Abstract The path-dependence of the J-integral is investigated numerically, via the finite element method, for a range of loadings, Poisson's ratios, and hardening exponents within the context of J2-flow plasticity. Small-scale yielding assumptions are employed using Dirichlet-to-Neumann map boundary conditions on a circular boundary that encloses the plastic zone. This construct allows for a dense finite element mesh within the plastic zone and accurate far-field boundary conditions. Details of the crack tip field that have been computed previously by others, including the existence of an elastic sector in Mode I loading, are confirmed. The somewhat unexpected result is that J for a contour approaching zero radius around the crack tip is approximately 18% lower than the far-field value for Mode I loading for Poisson’s ratios characteristic of metals. In contrast, practically no path-dependence is found for Mode II. The applications of T or S stresses, whether applied proportionally with the K-field or prior to K, have only a modest effect on the path-dependence. Keywords: elasto-plastic fracture mechanics, small scale yielding, path-dependence of the J-integral, finite element methods 1. Introduction The J-integral as introduced by Eshelby [1,2] and Rice [3] is perhaps the most useful quantity for the analysis of the mechanical fields near crack tips in both linear elastic and non-linear elastic materials.