UC San Diego UC San Diego Electronic Theses and Dissertations
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Corticotropin-Releasing Hormone Physiology
European Journal of Endocrinology (2006) 155 S71–S76 ISSN 0804-4643 Corticotropin-releasing hormone physiology Joseph A Majzoub Division of Endocrinology, Children’s Hospital Boston, Thomas Morgan Rotch Professor of Pediatrics, Harvard Medical School, 300 Longwood Avenue, Boston, Massachusetts 02115, USA (Correspondence should be addressed to J A Majzoub; Email: [email protected]) Abstract Corticotropin-releasing hormone (CRH), also known as corticotropin-releasing factor, is a highly conserved peptide hormone comprising 41 amino acid residues. Its name derives from its role in the anterior pituitary, where it mediates the release of corticotropin (ACTH) leading to the release of adrenocortical steroids. CRH is the major hypothalamic activator of the hypothalamic–pituitary– adrenal (HPA)axis. Major functions of the HPAinclude: (i) influencing fetal development of major organ systems including lung, liver, and gut, (ii) metabolic functions, including the maintenance of normal blood glucose levels during the fasting state via glycogenolysis and gluconeogenesis, (iii) modulation of immune function, and (iv) maintenance of cardiovascular tone. In addition, CRH, acting both directly and via the HPA, has a role in regulating several neuroendocrine functions including behavior, food intake, reproduction, growth, immune function, and autonomic function. CRH has been localized to the paraventricular nucleus (PVN) of the hypothalamus, which projects to the median eminence and other hypothalamic and midbrain targets. The CRH gene is composed of two exons. The CRH promoter contains a cAMP-response element, and the intron contains a restrictive element-1/neuron restrictive silencing element (RE-1/NRSE) sequence. Recently, a family of CRH-related peptides, termed the urocortins, has been identified. -

Expression of Urocortin and Corticotropin-Releasing Hormone Receptors in the Horse Thyroid Gland
Cell Tissue Res DOI 10.1007/s00441-012-1450-4 REGULAR ARTICLE Expression of urocortin and corticotropin-releasing hormone receptors in the horse thyroid gland Caterina Squillacioti & Adriana De Luca & Sabrina Alì & Salvatore Paino & Giovanna Liguori & Nicola Mirabella Received: 11 January 2012 /Accepted: 3 May 2012 # Springer-Verlag 2012 Abstract Urocortin (UCN) is a 40-amino-acid peptide and UCN, CRHR1 and CRHR2 and that UCN plays a role in a member of the corticotropin-releasing hormone (CRH) the regulation of thyroid physiological functions through a family, which includes CRH, urotensin I, sauvagine, paracrine mechanism. UCN2 and UCN3. The biological actions of CRH family peptides are mediated via two types of G-protein-coupled Keywords Follicular cells . C-cells . RT-PCR . receptors, namely CRH type 1 receptor (CRHR1) and CRH Immunohistochemistry . Horse type 2 receptor (CRHR2). The biological effects of these peptides are mediated and modulated not only by CRH receptors but also via a highly conserved CRH-binding Introduction protein (CRHBP). Our aim was to investigate the expression of UCN, CRHR1, CRHR2 and CRHBP by immunohisto- Urocortin (UCN) is a peptide of 40 amino acids and is a chemistry, Western blot and reverse transcription with the member of the corticotropin-releasing hormone (CRH) fam- polymerase chain reaction (RT-PCR) in the horse thyroid ily, which includes CRH, urotensin I, sauvagine, UCN2 and gland. The results showed that UCN, CRHR1 and CRHR2 UCN3. Vaughan et al. (1995) were the first to identify UCN, were expressed in the thyroid gland, whereas CRHBP was which exhibits 45% homology to CRH (Latchman 2002; not expressed. -

Searching for Novel Peptide Hormones in the Human Genome Olivier Mirabeau
Searching for novel peptide hormones in the human genome Olivier Mirabeau To cite this version: Olivier Mirabeau. Searching for novel peptide hormones in the human genome. Life Sciences [q-bio]. Université Montpellier II - Sciences et Techniques du Languedoc, 2008. English. tel-00340710 HAL Id: tel-00340710 https://tel.archives-ouvertes.fr/tel-00340710 Submitted on 21 Nov 2008 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. UNIVERSITE MONTPELLIER II SCIENCES ET TECHNIQUES DU LANGUEDOC THESE pour obtenir le grade de DOCTEUR DE L'UNIVERSITE MONTPELLIER II Discipline : Biologie Informatique Ecole Doctorale : Sciences chimiques et biologiques pour la santé Formation doctorale : Biologie-Santé Recherche de nouvelles hormones peptidiques codées par le génome humain par Olivier Mirabeau présentée et soutenue publiquement le 30 janvier 2008 JURY M. Hubert Vaudry Rapporteur M. Jean-Philippe Vert Rapporteur Mme Nadia Rosenthal Examinatrice M. Jean Martinez Président M. Olivier Gascuel Directeur M. Cornelius Gross Examinateur Résumé Résumé Cette thèse porte sur la découverte de gènes humains non caractérisés codant pour des précurseurs à hormones peptidiques. Les hormones peptidiques (PH) ont un rôle important dans la plupart des processus physiologiques du corps humain. -
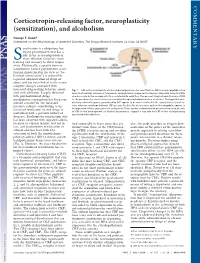
Corticotropin-Releasing Factor, Neuroplasticity (Sensitization), and Alcoholism
COMMENTARY Corticotropin-releasing factor, neuroplasticity (sensitization), and alcoholism George F. Koob* Committee on the Neurobiology of Addictive Disorders, The Scripps Research Institute, La Jolla, CA 92037 ensitization is a ubiquitous bio- logical phenomenon that has a role in the neuroadaptation of many different functions, from Slearning and memory to stress respon- sivity. Historically, a specific form of sensitization termed psychomotor sensi- tization (mistermed in my view as ‘‘be- havioral sensitization’’) is induced by repeated administration of drugs of abuse and has been linked to the neuro- adaptive changes associated with increased drug-seeking behavior associ- Fig. 1. CNS actions relevant to alcohol-induced psychomotor sensitization. CRF is a neuropeptide in the ated with addiction. Largely observed brain that controls autonomic, hormonal, and behavioral responses to stressors. New data show that CRF with psychostimulant drugs, also has a role in the neuroplasticity associated with addiction. The present study extends the role of CRF psychomotor sensitization has been con- to the psychomotor sensitization associated with repeated administration of alcohol. The hypothalamic- sidered a model for the increased pituitary-adrenal responses produced by CRF appear to be more involved in the acquisition of sensitiza- incentive salience contributing to the tion, whereas extrahypothalamic CRF systems, likely to be in structures such as the amygdala, appear to increased motivation to seek drugs in be important for the expression of sensitization. These results, combined with previous studies on the role of CRF in the development of alcohol dependence, suggest a key role for CRF in the neuroplasticity individuals with a previous history of associated with addiction. -

Urocortin 3 Overexpression Reduces ER Stress and Heat Shock Response in 3T3‑L1 Adipocytes Sina Kavalakatt1, Abdelkrim Khadir1, Dhanya Madhu1, Heikki A
www.nature.com/scientificreports OPEN Urocortin 3 overexpression reduces ER stress and heat shock response in 3T3‑L1 adipocytes Sina Kavalakatt1, Abdelkrim Khadir1, Dhanya Madhu1, Heikki A. Koistinen2,3,4, Fahd Al‑Mulla5, Jaakko Tuomilehto4,6, Jehad Abubaker1 & Ali Tiss 1* The neuropeptide urocortin 3 (UCN3) has a benefcial efect on metabolic disorders, such as obesity, diabetes, and cardiovascular disease. It has been reported that UCN3 regulates insulin secretion and is dysregulated with increasing severity of obesity and diabetes. However, its function in the adipose tissue is unclear. We investigated the overexpression of UCN3 in 3T3‑L1 preadipocytes and diferentiated adipocytes and its efects on heat shock response, ER stress, infammatory markers, and glucose uptake in the presence of stress‑inducing concentrations of palmitic acid (PA). UCN3 overexpression signifcantly downregulated heat shock proteins (HSP60, HSP72 and HSP90) and ER stress response markers (GRP78, PERK, ATF6, and IRE1α) and attenuated infammation (TNFα) and apoptosis (CHOP). Moreover, enhanced glucose uptake was observed in both preadipocytes and mature adipocytes, which is associated with upregulated phosphorylation of AKT and ERK but reduced p‑JNK. Moderate efects of UCN3 overexpression were also observed in the presence of 400 μM of PA, and macrophage conditioned medium dramatically decreased the UCN3 mRNA levels in diferentiated 3T3‑L1 cells. In conclusion, the benefcial efects of UCN3 in adipocytes are refected, at least partially, by the improvement in cellular -

Endocrine Cell Type Sorting and Mature Architecture in the Islets Of
www.nature.com/scientificreports OPEN Endocrine cell type sorting and mature architecture in the islets of Langerhans require expression of Received: 16 April 2018 Accepted: 4 July 2018 Roundabout receptors in β cells Published: xx xx xxxx Melissa T. Adams, Jennifer M. Gilbert, Jesus Hinojosa Paiz, Faith M. Bowman & Barak Blum Pancreatic islets of Langerhans display characteristic spatial architecture of their endocrine cell types. This architecture is critical for cell-cell communication and coordinated hormone secretion. Islet architecture is disrupted in type-2 diabetes. Moreover, the generation of architecturally correct islets in vitro remains a challenge in regenerative approaches to type-1 diabetes. Although the characteristic islet architecture is well documented, the mechanisms controlling its formation remain obscure. Here, we report that correct endocrine cell type sorting and the formation of mature islet architecture require the expression of Roundabout (Robo) receptors in β cells. Mice with whole-body deletion of Robo1 and conditional deletion of Robo2 either in all endocrine cells or selectively in β cells show complete loss of endocrine cell type sorting, highlighting the importance of β cells as the primary organizer of islet architecture. Conditional deletion of Robo in mature β cells subsequent to islet formation results in a similar phenotype. Finally, we provide evidence to suggest that the loss of islet architecture in Robo KO mice is not due to β cell transdiferentiation, cell death or loss of β cell diferentiation or maturation. Te islets of Langerhans display typical, species-specifc architecture, with distinct spatial organization of their various endocrine cell types1–5. In the mouse, the core of the islet is composed mostly of insulin-secreting β cells, while glucagon-secreting α cells, somatostatin-secreting δ cells and pancreatic polypeptide-secreting PP cells are located at the islet periphery3. -

Urocortin 2 Modulates Glucose Utilization and Insulin Sensitivity in Skeletal Muscle
Urocortin 2 modulates glucose utilization and insulin sensitivity in skeletal muscle Alon Chen*†, Bhawanjit Brar*, Cheol Soo Choi‡, David Rousso*, Joan Vaughan*, Yael Kuperman†, Shee Ne Kim‡, Cindy Donaldson*, Sean M. Smith*, Pauline Jamieson*, Chien Li*, Tim R. Nagy§, Gerald I. Shulman‡, Kuo-Fen Lee*, and Wylie Vale*¶ *Clayton Foundation Laboratories for Peptide Biology, The Salk Institute for Biological Studies, La Jolla, CA 92037; †Department of Neurobiology, The Weizmann Institute of Science, Rehovot 76100, Israel; ‡Department of Internal Medicine and Cellular and Molecular Physiology and Howard Hughes Medical Institute, Yale University School of Medicine, New Haven, CT 06536; and §Department of Nutrition Sciences, University of Alabama at Birmingham, Birmingham, AL 35294 Contributed by Wylie Vale, August 28, 2006 Skeletal muscle is the principal tissue responsible for insulin- Results and Discussion stimulated glucose disposal and is a major site of peripheral insulin Generation of Ucn 2-Deficient Mice. To explore the physiological resistance. Urocortin 2 (Ucn 2), a member of the corticotropin- role of Ucn 2, we generated mice that are deficient in this releasing factor (CRF) family, and its cognate type 2 CRF receptor peptide. A genomic DNA clone containing Ucn 2 was isolated, (CRFR2) are highly expressed in skeletal muscle. To determine the and a targeting construct in which the full Ucn 2 coding sequence physiological role of Ucn 2, we generated mice that are deficient in was replaced with a neomycin-resistant gene cassette was gen- this peptide. Using glucose-tolerance tests (GTTs), insulin-tolerance erated (Fig. 1a). J1 ES cells were electroporated with the tests (ITTs), and hyperinsulinemic euglycemic glucose clamp stud- targeting construct and were selected as described in ref. -

UC Davis UC Davis Previously Published Works
UC Davis UC Davis Previously Published Works Title Cardiac CRFR1 Expression Is Elevated in Human Heart Failure and Modulated by Genetic Variation and Alternative Splicing. Permalink https://escholarship.org/uc/item/8r73t67n Journal Endocrinology, 157(12) ISSN 0013-7227 Authors Pilbrow, Anna P Lewis, Kathy A Perrin, Marilyn H et al. Publication Date 2016-12-01 DOI 10.1210/en.2016-1448 License https://creativecommons.org/licenses/by-nc-nd/4.0/ 4.0 Peer reviewed eScholarship.org Powered by the California Digital Library University of California Manuscript (MUST INCLUDE TITLE PAGE AND ABSTRACT) Click here to download Manuscript (MUST INCLUDE TITLE PAGE AND ABSTRACT) Endocrinology CRFR1 ms.docx 1 Myocardial expression of Corticotropin-Releasing Factor Receptor 1 (CRFR1) is elevated in human 2 heart failure and is modulated by genetic variation and a novel CRFR1 splice variant. 3 4 Anna P Pilbrow1,2,* PhD, Kathy A Lewis1 BS, Marilyn H Perrin1 PhD, Wendy E Sweet3 MS, Christine S 5 Moravec3 PhD, WH Wilson Tang3 MD, Mark O Huising1 PhD, Richard W Troughton2 MD PhD and 6 Vicky A Cameron2 PhD. 7 8 1. Peptide Biology Laboratories, The Salk Institute for Biological Studies, 10010 North Torrey Pines 9 Road, La Jolla, CA 92037, USA. 10 2. Christchurch Heart Institute, Department of Medicine, University of Otago, Christchurch, 2 11 Riccarton Avenue, PO Box 4345, Christchurch 8011, New Zealand. 12 3. Kaufman Center for Heart Failure, Department of Cardiovascular Medicine, Cleveland Clinic, 9500 13 Euclid Avenue, Cleveland, OH 44195, USA. 14 15 Abbreviated title: CRFR1 in human heart failure 16 Keywords: heart failure, CRFR1, CRHR1, alternative splicing, splice variant, polymorphism, human. -

Genetic Drivers of Pancreatic Islet Function
| INVESTIGATION Genetic Drivers of Pancreatic Islet Function Mark P. Keller,*,1 Daniel M. Gatti,†,1 Kathryn L. Schueler,* Mary E. Rabaglia,* Donnie S. Stapleton,* Petr Simecek,† Matthew Vincent,† Sadie Allen,‡ Aimee Teo Broman,§ Rhonda Bacher,§ Christina Kendziorski,§ Karl W. Broman,§ Brian S. Yandell,** Gary A. Churchill,†,2 and Alan D. Attie*,2 *Department of Biochemistry, §Department of Biostatistics and Medical Informatics, and **Department of Horticulture, University of Wisconsin–Madison, Wisconsin 53706-1544, †The Jackson Laboratory, Bar Harbor, Maine 06409, and ‡Maine School of Science and Mathematics, Limestone, Maine 06409, ORCID IDs: 0000-0002-7405-5552 (M.P.K.); 0000-0002-4914-6671 (K.W.B.); 0000-0001-9190-9284 (G.A.C.); 0000-0002-0568-2261 (A.D.A.) ABSTRACT The majority of gene loci that have been associated with type 2 diabetes play a role in pancreatic islet function. To evaluate the role of islet gene expression in the etiology of diabetes, we sensitized a genetically diverse mouse population with a Western diet high in fat (45% kcal) and sucrose (34%) and carried out genome-wide association mapping of diabetes-related phenotypes. We quantified mRNA abundance in the islets and identified 18,820 expression QTL. We applied mediation analysis to identify candidate causal driver genes at loci that affect the abundance of numerous transcripts. These include two genes previously associated with monogenic diabetes (PDX1 and HNF4A), as well as three genes with nominal association with diabetes-related traits in humans (FAM83E, IL6ST, and SAT2). We grouped transcripts into gene modules and mapped regulatory loci for modules enriched with transcripts specific for a-cells, and another specific for d-cells. -

Expression of Urocortin Peptides in Canine Myocardium and Plasma
Edinburgh Research Explorer Expression of urocortin peptides in canine myocardium and plasma Citation for published version: Veloso, GF, Ohad, DG, Francis, AJ, Vaughan, JM, Brownstein, DG, Culshaw, GJ, Vale, WW, French, AT & Jamieson, PM 2011, 'Expression of urocortin peptides in canine myocardium and plasma', The Veterinary Journal, vol. 188, no. 3, pp. 318-324. https://doi.org/10.1016/j.tvjl.2010.05.019 Digital Object Identifier (DOI): 10.1016/j.tvjl.2010.05.019 Link: Link to publication record in Edinburgh Research Explorer Document Version: Peer reviewed version Published In: The Veterinary Journal Publisher Rights Statement: © 2010 Elsevier Ltd General rights Copyright for the publications made accessible via the Edinburgh Research Explorer is retained by the author(s) and / or other copyright owners and it is a condition of accessing these publications that users recognise and abide by the legal requirements associated with these rights. Take down policy The University of Edinburgh has made every reasonable effort to ensure that Edinburgh Research Explorer content complies with UK legislation. If you believe that the public display of this file breaches copyright please contact [email protected] providing details, and we will remove access to the work immediately and investigate your claim. Download date: 06. Oct. 2021 NIH Public Access Author Manuscript Vet J. Author manuscript; available in PMC 2012 June 1. NIH-PA Author ManuscriptPublished NIH-PA Author Manuscript in final edited NIH-PA Author Manuscript form as: Vet J. 2011 June ; 188(3): 318±324. doi:10.1016/j.tvjl.2010.05.019. Expression of urocortin peptides in canine myocardium and plasma Gemma Fraga Velosoa, Dan G. -

Targeted Pharmacological Therapy Restores Β-Cell Function for Diabetes Remission
Targeted pharmacological therapy restores -cell function for diabetes remission Sachs, Stephan; Bastidas-Ponce, Aimée; Tritschler, Sophie; Bakhti, Mostafa; Böttcher, Anika; Sánchez-Garrido, Miguel A; Tarquis-Medina, Marta; Kleinert, Maximilian; Fischer, Katrin; Jall, Sigrid; Harger, Alexandra; Bader, Erik; Roscioni, Sara; Ussar, Siegfried; Feuchtinger, Annette; Yesildag, Burcak; Neelakandhan, Aparna; Jensen, Christine B; Cornu, Marion; Yang, Bin; Finan, Brian; DiMarchi, Richard D; Tschöp, Matthias H; Theis, Fabian J; Hofmann, Susanna M.; Müller, Timo D; Lickert, Heiko Published in: Nature Metabolism DOI: 10.1038/s42255-020-0171-3 Publication date: 2020 Document version Publisher's PDF, also known as Version of record Document license: CC BY Citation for published version (APA): Sachs, S., Bastidas-Ponce, A., Tritschler, S., Bakhti, M., Böttcher, A., Sánchez-Garrido, M. A., Tarquis-Medina, M., Kleinert, M., Fischer, K., Jall, S., Harger, A., Bader, E., Roscioni, S., Ussar, S., Feuchtinger, A., Yesildag, B., Neelakandhan, A., Jensen, C. B., Cornu, M., ... Lickert, H. (2020). Targeted pharmacological therapy restores - cell function for diabetes remission. Nature Metabolism, 2(2), 192-209. https://doi.org/10.1038/s42255-020- 0171-3 Download date: 05. Oct. 2021 ARTICLES https://doi.org/10.1038/s42255-020-0171-3 There are amendments to this paper Targeted pharmacological therapy restores β-cell function for diabetes remission Stephan Sachs1,2,3,4,19, Aimée Bastidas-Ponce1,4,5,6,19, Sophie Tritschler1,4,7,8,19, Mostafa Bakhti 1,4,5, Anika Böttcher1,4,5, Miguel A. Sánchez-Garrido2, Marta Tarquis-Medina1,4,5,6, Maximilian Kleinert2,9, Katrin Fischer2,3, Sigrid Jall2,3, Alexandra Harger2, Erik Bader1, Sara Roscioni1, Siegfried Ussar 4,6,10, Annette Feuchtinger11, Burcak Yesildag12, Aparna Neelakandhan12, Christine B. -

Perifornical Area Urocortin-3 Neurons Promote Infant-Directed Neglect and Aggression
bioRxiv preprint doi: https://doi.org/10.1101/697334; this version posted July 9, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Perifornical Area Urocortin-3 Neurons Promote Infant-directed Neglect and Aggression Anita E Autry1,2, Zheng Wu1,3, Johannes Kohl1,4, Dhananjay Bambah-Mukku1, Nimrod D Rubinstein1,5, Brenda Marin-Rodriguez1, Ilaria Carta2, Victoria Sedwick2, & Catherine Dulac1 1 Howard Hughes Medical Institute, Department of Molecular and Cellular Biology, Center for Brain Science, Harvard University, Cambridge, Massachusetts 02138, USA. 2 Dominick P. Purpura Department of Neuroscience, Department of Psychiatry and Behavioral Sciences, Albert Einstein College of Medicine, Bronx, NY 10461, USA. 3 Current Address: Mortimer B. Zuckerman Mind Brain Behavior Institute and Department of Neuroscience, Columbia University, New York, NY 10027, USA. 4 Current Address: The Francis Crick Institute, London, UK. 5 Current Address: Calico Life Sciences, LLC, 1170 Veterans Blvd, South San Francisco, 94080, USA . 1 bioRxiv preprint doi: https://doi.org/10.1101/697334; this version posted July 9, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. SUMMARY Mammals invest considerable resources in protecting and nurturing young offspring. However, under certain physiological and environmental conditions, animals neglect or attack young conspecifics. Males in some species attack unfamiliar infants to gain reproductive advantage1-3 and females kill or neglect their young during stressful circumstances such as food shortage or threat of predation4-8.