Rocky Seashore Activities
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Risk Analysis: Vessel Biofouling
Risk Analysis: Vessel Biofouling ISBN 978-0-478-37548-0 (print) ISBN 978-0-478-37549-7 (online) 15 February 2011 Risk Analysis: Vessel Biofouling 15 February 2011 Approved for general release Christine Reed Manager, Risk Analysis Ministry of Agriculture and Forestry Requests for further copies should be directed to: Publication Adviser MAF Information Bureau P O Box 2526 WELLINGTON Telephone: 0800 00 83 33 Facsimile: 04-894 0300 This publication is also available on the MAF website at http://www.biosecurity.govt.nz/regs/imports/ihs/risk © Crown Copyright - Ministry of Agriculture and Forestry i Contributors to this risk analysis 1. Primary author/s Dr Andrew Bell Senior Adviser MAF Biosecurity New Zealand Risk Analysis, Marine Wellington Simon Phillips Adviser MAF Biosecurity New Zealand Risk Analysis, Marine Wellington Dr Eugene Georgiades Senior Adviser MAF Biosecurity New Zealand Risk Analysis, Marine Wellington Dr Daniel Kluza Senior Adviser MAF Biosecurity New Zealand Risk Analysis, Marine Wellington 2. Secondary contributors Dr Christopher Denny Adviser MAF Biosecurity New Zealand Border Standards Wellington 3. External peer review John Lewis Principal Marine Consultant ES Link Services Pty Ltd Melbourne, Victoria, Australia Richard Piola Senior Scientist Cawthron Institute Nelson, New Zealand The draft risk analysis has also been internally reviewed by: Liz Jones (Border Standards); Justin McDonald (Post-Clearance); Melanie Newfield (Risk Analysis); Howard Pharo (Risk Analysis); Sandy Toy (Risk Analysis). The contribution of all the reviewers is gratefully acknowledged. ii Contents Page Executive summary 1 Definitions 7 1. Introduction 8 1.1. Background 8 1.2. Scope 13 1.3. References 14 2. Methodology 19 2.1. -
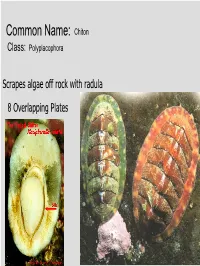
Common Name: Chiton Class: Polyplacophora
Common Name: Chiton Class: Polyplacophora Scrapes algae off rock with radula 8 Overlapping Plates Phylum? Mollusca Class? Gastropoda Common name? Brown sea hare Class? Scaphopoda Common name? Tooth shell or tusk shell Mud Tentacle Foot Class? Gastropoda Common name? Limpet Phylum? Mollusca Class? Bivalvia Class? Gastropoda Common name? Brown sea hare Phylum? Mollusca Class? Gastropoda Common name? Nudibranch Class? Cephalopoda Cuttlefish Octopus Squid Nautilus Phylum? Mollusca Class? Gastropoda Most Bivalves are Filter Feeders A B E D C • A: Mantle • B: Gill • C: Mantle • D: Foot • E: Posterior adductor muscle I.D. Green: Foot I.D. Red Gills Three Body Regions 1. Head – Foot 2. Visceral Mass 3. Mantle A B C D • A: Radula • B: Mantle • C: Mouth • D: Foot What are these? Snail Radulas Dorsal HingeA Growth line UmboB (Anterior) Ventral ByssalC threads Mussel – View of Outer Shell • A: Hinge • B: Umbo • C: Byssal threads Internal Anatomy of the Bay Mussel A B C D • A: Labial palps • B: Mantle • C: Foot • D: Byssal threads NacreousB layer Posterior adductorC PeriostracumA muscle SiphonD Mantle Byssal threads E Internal Anatomy of the Bay Mussel • A: Periostracum • B: Nacreous layer • C: Posterior adductor muscle • D: Siphon • E: Mantle Byssal gland Mantle Gill Foot Labial palp Mantle Byssal threads Gill Byssal gland Mantle Foot Incurrent siphon Byssal Labial palp threads C D B A E • A: Foot • B: Gills • C: Posterior adductor muscle • D: Excurrent siphon • E: Incurrent siphon Heart G F H E D A B C • A: Foot • B: Gills • C: Mantle • D: Excurrent siphon • E: Incurrent siphon • F: Posterior adductor muscle • G: Labial palps • H: Anterior adductor muscle Siphon or 1. -

Crepidula Fornicata
NOBANIS - Marine invasive species in Nordic waters - Fact Sheet Crepidula fornicata Author of this species fact sheet: Kathe R. Jensen, Zoological Museum, Natural History Museum of Denmark, Universiteteparken 15, 2100 København Ø, Denmark. Phone: +45 353-21083, E-mail: [email protected] Bibliographical reference – how to cite this fact sheet: Jensen, Kathe R. (2010): NOBANIS – Invasive Alien Species Fact Sheet – Crepidula fornicata – From: Identification key to marine invasive species in Nordic waters – NOBANIS www.nobanis.org, Date of access x/x/201x. Species description Species name Crepidula fornicata, Linnaeus, 1758 – Slipper limpet Synonyms Patella fornicata Linnaeus, 1758; Crepidula densata Conrad, 1871; Crepidula virginica Conrad, 1871; Crepidula maculata Rigacci, 1866; Crepidula mexicana Rigacci, 1866; Crepidula violacea Rigacci, 1866; Crepidula roseae Petuch, 1991; Crypta nautarum Mörch, 1877; Crepidula nautiloides auct. NON Lesson, 1834 (ISSG Database; Minchin, 2008). Common names Tøffelsnegl (DK, NO), Slipper limpet, American slipper limpet, Common slipper snail (UK, USA), Østerspest (NO), Ostronpest (SE), Amerikanische Pantoffelschnecke (DE), La crépidule (FR), Muiltje (NL), Seba (E). Identification Crepidula fornicata usually sit in stacks on a hard substrate, e.g., a shell of other molluscs, boulders or rocky outcroppings. Up to 13 animals have been reported in one stack, but usually 4-6 animals are seen in a stack. Individual shells are up to about 60 mm in length, cap-shaped, distinctly longer than wide. The “spire” is almost invisible, slightly skewed to the right posterior end of the shell. Shell height is highly variable. Inside the body whorl is a characteristic calcareous shell-plate (septum) behind which the visceral mass is protected. -

ECOLOGICAL ENERGETICS of TROPICAL LIMPET Cellana Testudinaria (Linnaeus, 1758) LIVING on the ROCKY SHORE of OHOIWAIT, SOUTHEAST MOLUCCAS, INDONESIA
Journal of Coastal Deveolpment ISSN : 1410-5217 Volume 11, Number 2, February 2008 : 89-96 ECOLOGICAL ENERGETICS OF TROPICAL LIMPET Cellana testudinaria (Linnaeus, 1758) LIVING ON THE ROCKY SHORE OF OHOIWAIT, SOUTHEAST MOLUCCAS, INDONESIA Abraham Seumel Khouw Faculty of Fisheries and Marine Sciences, Pattimura University, Ambon Indonesia Received : November, 2, 2007 ; Accepted :January,4, 2008 ABSTRACT Study on ecological energetics of tropical limpet C. testudinaria has been carried out at approximately one year from October 2001 to September 2002. Population energy budgets estimated on the assumption of steady state conditions for C. testudinaria (Linnaeus, 1758) on the rocky shore of Ohoiwait, are presented. Large difference in population structure, and hence energetics, occurred at different localities along the rocky shore. Relatively high proportions (98 %) of the assimilated energy was lost via metabolism. Assimilation efficiency is 39 %, net growth efficiency is 1.8 %, and ecological efficiency 0.3 %. Production (P), energy flow (A) and total energy consumption (C) were expressed as functions of animal size, in order to facilitate gross estimations of the energy component for which data on size frequency and density are available. Key words: ecological energetics, cellana testudinaria, energy components Correspondence: Phone : +6281343044295, e-mail: [email protected] INTRODUCTION Cellana testudinaria is intertidal, grazing Little has been published on the gastropod abundant on medium to very ecology of C. testudinaria. Khouw (2002) exposed rocky shores of Ohoiwait. The discussed their growth pattern and shell species shows marked zonation, with only a shape variation in relation to zonal little overlap between zones. C. testudinaria distribution. Distribution, abundance, and occurs at several spatial and temporal scales biomass were investigated by Khouw from the extreme low water spring tide (2006a) and presented evidence for the (ELWST) to the extreme high water spring effects of drying. -

Marine Invertebrate Field Guide
Marine Invertebrate Field Guide Contents ANEMONES ....................................................................................................................................................................................... 2 AGGREGATING ANEMONE (ANTHOPLEURA ELEGANTISSIMA) ............................................................................................................................... 2 BROODING ANEMONE (EPIACTIS PROLIFERA) ................................................................................................................................................... 2 CHRISTMAS ANEMONE (URTICINA CRASSICORNIS) ............................................................................................................................................ 3 PLUMOSE ANEMONE (METRIDIUM SENILE) ..................................................................................................................................................... 3 BARNACLES ....................................................................................................................................................................................... 4 ACORN BARNACLE (BALANUS GLANDULA) ....................................................................................................................................................... 4 HAYSTACK BARNACLE (SEMIBALANUS CARIOSUS) .............................................................................................................................................. 4 CHITONS ........................................................................................................................................................................................... -

Animal Adaptations
Animal Adaptations The Animal Adaptations program at Hatfield Marine Science Center is designed to be a 50- minute lab-based program for 3rd-12th grade students that examines marine organisms from three different habitats (sandy beach, rocky shore and estuary) and explores the many ways they are adapted to their particular environment. This lab focuses on the adaptations of several groups of marine animals including Mollusks, Crustaceans, and Echinoderms, and investigates how they differ depending on whether they are found in a sandy beach, rocky shore, or estuary environment. Students will work in small groups with a variety of live animals, studying individual characteristics and how these organisms interact with their environment and one another. Background Information The Oregon Coast is made up of a series of rocky shores, sandy beaches, and estuaries, all of which are greatly affected by fluctuating tides. Many of these areas are intertidal and are alternately inundated by seawater and exposed to air, wind, and dramatic changes in temperature and salinity. High tide floods these areas with cold, nutrient laden seawater, bringing food to organisms that live there in the form of plankton and detritus. Low tides often expose these organisms to the dangers of predation and desiccation. In addition to tidal effects, organisms that inhabit sandy beaches and rocky shores also have to deal with the physical stresses of pounding waves. Because of these harsh conditions, organisms have developed special adaptations that not only help them to survive but thrive in these environments. An adaptation is a physical or behavioral trait that helps a plant or animal survive in a specific environment or habitat. -

Title Biogeography in Cellana (Patellogastropoda, Nacellidae) with Special Emphasis on the Relationships of Southern Hemisphere
Biogeography in Cellana (Patellogastropoda, Nacellidae) with Title Special Emphasis on the Relationships of Southern Hemisphere Oceanic Island Species González-Wevar, Claudio A.; Nakano, Tomoyuki; Palma, Author(s) Alvaro; Poulin, Elie Citation PLOS ONE (2017), 12(1) Issue Date 2017-01-18 URL http://hdl.handle.net/2433/218484 © 2017 González-Wevar et al. This is an open access article distributed under the terms of the Creative Commons Right Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Type Journal Article Textversion publisher Kyoto University RESEARCH ARTICLE Biogeography in Cellana (Patellogastropoda, Nacellidae) with Special Emphasis on the Relationships of Southern Hemisphere Oceanic Island Species Claudio A. GonzaÂlez-Wevar1,2*, Tomoyuki Nakano3, Alvaro Palma4, Elie Poulin1 1 GAIA-AntaÂrtica, Universidad de Magallanes, Punta Arenas, Chile, 2 Instituto de EcologõÂa y Biodiversidad Ä a1111111111 (IEB), Departamento de Ciencias EcoloÂgicas, Facultad de Ciencias, Universidad de Chile, Nuñoa, Santiago, Chile, 3 Seto Marine Biological Laboratory, Field Science Education and Research Centre, Kyoto University, a1111111111 Nishimuro, Wakayama, Japan, 4 Universidad Gabriela Mistral, Facultad de IngenierõÂa y Negocios, a1111111111 Providencia, Santiago, Chile a1111111111 a1111111111 * [email protected] Abstract OPEN ACCESS Oceanic islands lacking connections to other land are extremely isolated from sources of Citation: GonzaÂlez-Wevar CA, Nakano T, Palma A, potential colonists and have acquired their biota mainly through dispersal from geographi- Poulin E (2017) Biogeography in Cellana cally distant areas. Hence, isolated island biota constitutes interesting models to infer bio- (Patellogastropoda, Nacellidae) with Special geographical mechanisms of dispersal, colonization, differentiation, and speciation. Limpets Emphasis on the Relationships of Southern Hemisphere Oceanic Island Species. -

Seashore the SANDYSEASHORE the Soilisinfertile, Anditisoften Windy, Andsalty
SEASHORE DESCRIPTION The seashore is an area fi lled with an interesting mix of unique plants and animals that have adapted to cope with this environment. Living on the edge of the sea is not easy. The soil is infertile, and it is often windy, dry and salty. THE SANDY SEASHORE Along a sandy shore there are no large rocks, algae or tidal pools. The sandy seashore can be divided into four general zones: Intertidal, Pioneer, Fixed Dune and Scrub Woodland. 1. Intertidal Zone: Between the low tide and the high tide mark is the intertidal zone. When the tide goes out the creatures living in this zone are left stranded. They have to endure the heat of the sun and the higher salinity of the water resulting from evaporation. Notice the many small holes on a sandy beach; they are the doorways to the homes of many animals which burrow under the sand where it is cooler. Some of Ecosystems of The Bahamas the creatures living in this zone are sea worms, sand fl eas and sand crabs. 2. Pioneer Zone: So named because it is where the fi rst plants to try to grow over sand. These plants must adapt to loose, shifting sand and poor soil. There is no protection from wind or salt spray. Plants here are usually low growing vines with waxy leaves. Some plants found in the pioneer zone are Purple seaside bean (C. rosea), Saltwort (Batis maritima), Goat's foot (Ipomea pes-caprae), and Sea purslane (Sesuvium portulacastrum). 3. Fixed Dune Zone: The next zone is the fi xed dune, so named because as the plants in the pioneer zone grip the sand around their roots and make the beach more stable, the sand mounds up into small humps or dunes. -

Phylum MOLLUSCA Chitons, Bivalves, Sea Snails, Sea Slugs, Octopus, Squid, Tusk Shell
Phylum MOLLUSCA Chitons, bivalves, sea snails, sea slugs, octopus, squid, tusk shell Bruce Marshall, Steve O’Shea with additional input for squid from Neil Bagley, Peter McMillan, Reyn Naylor, Darren Stevens, Di Tracey Phylum Aplacophora In New Zealand, these are worm-like molluscs found in sandy mud. There is no shell. The tiny MOLLUSCA solenogasters have bristle-like spicules over Chitons, bivalves, sea snails, sea almost the whole body, a groove on the underside of the body, and no gills. The more worm-like slugs, octopus, squid, tusk shells caudofoveates have a groove and fewer spicules but have gills. There are 10 species, 8 undescribed. The mollusca is the second most speciose animal Bivalvia phylum in the sea after Arthropoda. The phylum Clams, mussels, oysters, scallops, etc. The shell is name is taken from the Latin (molluscus, soft), in two halves (valves) connected by a ligament and referring to the soft bodies of these creatures, but hinge and anterior and posterior adductor muscles. most species have some kind of protective shell Gills are well-developed and there is no radula. and hence are called shellfish. Some, like sea There are 680 species, 231 undescribed. slugs, have no shell at all. Most molluscs also have a strap-like ribbon of minute teeth — the Scaphopoda radula — inside the mouth, but this characteristic Tusk shells. The body and head are reduced but Molluscan feature is lacking in clams (bivalves) and there is a foot that is used for burrowing in soft some deep-sea finned octopuses. A significant part sediments. The shell is open at both ends, with of the body is muscular, like the adductor muscles the narrow tip just above the sediment surface for and foot of clams and scallops, the head-foot of respiration. -

The American Slipper Limpet Crepidula Fornicata (L.) in the Northern Wadden Sea 70 Years After Its Introduction
Helgol Mar Res (2003) 57:27–33 DOI 10.1007/s10152-002-0119-x ORIGINAL ARTICLE D. W. Thieltges · M. Strasser · K. Reise The American slipper limpet Crepidula fornicata (L.) in the northern Wadden Sea 70 years after its introduction Received: 14 December 2001 / Accepted: 15 August 2001 / Published online: 25 September 2002 © Springer-Verlag and AWI 2002 Abstract In 1934 the American slipper limpet 1997). In the centre of its European distributional range Crepidula fornicata (L.) was first recorded in the north- a population explosion has been observed on the Atlantic ern Wadden Sea in the Sylt-Rømø basin, presumably im- coast of France, southern England and the southern ported with Dutch oysters in the preceding years. The Netherlands. This is well documented (reviewed by present account is the first investigation of the Crepidula Blanchard 1997) and sparked a variety of studies on the population since its early spread on the former oyster ecological and economic impacts of Crepidula. The eco- beds was studied in 1948. A field survey in 2000 re- logical impacts of Crepidula are manifold, and include vealed the greatest abundance of Crepidula in the inter- the following: tidal/subtidal transition zone on mussel (Mytilus edulis) (1) Accumulation of pseudofaeces and of fine sediment beds. Here, average abundance and biomass was 141 m–2 through the filtration activity of Crepidula and indi- and 30 g organic dry weight per square metre, respec- viduals protruding in stacks into the water column. tively. On tidal flats with regular and extended periods of This was reported to cause changes in sediments and emersion as well as in the subtidal with swift currents in near-bottom currents (Ehrhold et al. -

PROTISTS Shore and the Waves Are Large, Often the Largest of a Storm Event, and with a Long Period
(seas), and these waves can mobilize boulders. During this phase of the storm the rapid changes in current direction caused by these large, short-period waves generate high accelerative forces, and it is these forces that ultimately can move even large boulders. Traditionally, most rocky-intertidal ecological stud- ies have been conducted on rocky platforms where the substrate is composed of stable basement rock. Projec- tiles tend to be uncommon in these types of habitats, and damage from projectiles is usually light. Perhaps for this reason the role of projectiles in intertidal ecology has received little attention. Boulder-fi eld intertidal zones are as common as, if not more common than, rock plat- forms. In boulder fi elds, projectiles are abundant, and the evidence of damage due to projectiles is obvious. Here projectiles may be one of the most important defi ning physical forces in the habitat. SEE ALSO THE FOLLOWING ARTICLES Geology, Coastal / Habitat Alteration / Hydrodynamic Forces / Wave Exposure FURTHER READING Carstens. T. 1968. Wave forces on boundaries and submerged bodies. Sarsia FIGURE 6 The intertidal zone on the north side of Cape Blanco, 34: 37–60. Oregon. The large, smooth boulders are made of serpentine, while Dayton, P. K. 1971. Competition, disturbance, and community organi- the surrounding rock from which the intertidal platform is formed zation: the provision and subsequent utilization of space in a rocky is sandstone. The smooth boulders are from a source outside the intertidal community. Ecological Monographs 45: 137–159. intertidal zone and were carried into the intertidal zone by waves. Levin, S. A., and R. -

Chitons (Mollusca: Polyplacophora) Known from Benthic Monitoring Programs in the Southern California Bight
ISSN 0738-9388 THE FESTIVUS A publication of the San Diego Shell Club Volume XLI Special Issue June 11, 2009 Chitons (Mollusca: Polyplacophora) Known from Benthic Monitoring Programs in the Southern California Bight Timothy D. Stebbins and Douglas J. Eernisse COVER PHOTO Live specimen of Lepidozona sp. C occurring on a piece of metal debris collected off San Diego, southern California at a depth of 90 m. Photo provided courtesy of R. Rowe. Vol. XLI(6): 2009 THE FESTIVUS Page 53 CHITONS (MOLLUSCA: POLYPLACOPHORA) KNOWN FROM BENTHIC MONITORING PROGRAMS IN THE SOUTHERN CALIFORNIA BIGHT TIMOTHY D. STEBBINS 1,* and DOUGLAS J. EERNISSE 2 1 City of San Diego Marine Biology Laboratory, Metropolitan Wastewater Department, San Diego, CA, USA 2 Department of Biological Science, California State University, Fullerton, CA, USA Abstract: About 36 species of chitons possibly occur at depths greater than 30 m along the continental shelf and slope of the Southern California Bight (SCB), although little is known about their distribution or ecology. Nineteen species are reported here based on chitons collected as part of long-term, local benthic monitoring programs or less frequent region-wide surveys of the entire SCB, and these show little overlap with species that occur at depths typically encountered by scuba divers. Most chitons were collected between 30-305 m depths, although records are included for a few from slightly shallower waters. Of the two extant chiton lineages, Lepidopleurida is represented by Leptochitonidae (2 genera, 3 species), while Chitonida is represented by Ischnochitonidae (2 genera, 6-9 species) and Mopaliidae (4 genera, 7 species).