Emerging Mechanisms and Treatments for Depression Beyond Ssris and Snris
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Corporate Release No 485 12 December 2012 FDA Accepts
H. Lundbeck A/S Ottiliavej 9 Tel +45 36 30 13 11 E-mail [email protected] DK-2500 Valby, Copenhagen Fax +45 36 43 82 62 www.lundbeck.com CVR number: 56759913 Corporate Release No 485 12 December 2012 FDA accepts Takeda and Lundbeck’s filing for review of Brintellix (vortioxetine) for the treatment of major depression FDA has determined that the New Drug Application filed in October 2012 is sufficiently complete to permit a substantive review Upon the acceptance of the filing by the FDA, Lundbeck is to receive a milestone of USD 50 million (approximately DKK 285 million) from Takeda H. Lundbeck A/S (Lundbeck) announced today that the U.S. Food and Drug Administration (FDA) has accepted the submission of a New Drug Application (NDA) for BrintellixTM (vortioxetine) for the treatment of major depressive disorder (MDD) in adult patients. Brintellix (pronounced “brin′-tel-ix”) is the proposed global trade name for vortioxetine. According to the timelines established by the Prescription Drug User Fee Act (PDUFA), the review of the NDA is targeted for completion by 2 October, 2013. The NDA includes positive data from six short-term studies and one long-term maintenance study. The vortioxetine global clinical development program included more than 7,500 individuals aged 18 to 88 years old exposed to the drug. Major depression, often referred to as depression, is a common, debilitating illness affecting around 15 million Americans and 121 million people worldwide. Depression was the third leading contributor to the global burden of disease in 2004 and is projected to be the leading contributor to the worldwide burden of disease by 2030. -

Download Resume
Curriculum Vitae Full Name: David M. Marks, M.D. Contact: [email protected] Mobile (619) 822-7117 Credentials: Diplomate, American Board of Psychiatry and Neurology (Psychiatry) Subspecialty Certification in Psychosomatic Medicine Diplomate, American Board of Pain Medicine Position Title: Associate Professor Department of Psychiatry and Behavioral Sciences Department of Community and Family Medicine Duke University Medical Center Duke Clinical Research Institute Duke Pain and Palliative Care Clinic Education: Institution & Location Degree Year Conferred Field of Study University of California at San Diego Fellowship 1999 - 2000 Consultation and San Diego, CA Liaison Psychiatry Medical College of Pennsylvania / Senior 1998 - 1999 Psychiatry Clinical Neuroscience Research Unit Resident Philadelphia, PA University of California at San Diego Resident 1995 - 1998 Psychiatry San Diego, CA University of Texas Medical Branch M.D. 1995 Galveston, TX Rice University B.A. 1991 Psychology Houston, TX Research and Professional Experience: Position Institution/Employer & Location Dates of Employment Attending Faculty Physician Duke Pain and Palliative Care Clinic 09/08-present (Chronic Pain Management) Attending Faculty Physician Duke University Medical Center, 07/06-present Inpatient Psychiatric Service, Emergency Service, Consultation/Liaison Service Attending Faculty Physician Durham Regional Hospital, 09/08-07/11 Consultation/Liaison Service Medical Director, Inpatient and Duke University Medical Center 07/06 – 02/07 Emergency Psychiatry Services Medical Director, CNS Division EStudy Site 05/05 -- 07/06 La Mesa, Oceanside, National City CA Chief Executive Officer/Medical Optimum Health Services 01/02 – 05/05 Director La Mesa, Oceanside CA Chief of Staff Alvarado Parkway Institute 01/04 – 01/05 1 La Mesa, CA Page _____________________________________________________________________ David M. -

Metabotropic Glutamate Receptors
mGluR Metabotropic glutamate receptors mGluR (metabotropic glutamate receptor) is a type of glutamate receptor that are active through an indirect metabotropic process. They are members of thegroup C family of G-protein-coupled receptors, or GPCRs. Like all glutamate receptors, mGluRs bind with glutamate, an amino acid that functions as an excitatoryneurotransmitter. The mGluRs perform a variety of functions in the central and peripheral nervous systems: mGluRs are involved in learning, memory, anxiety, and the perception of pain. mGluRs are found in pre- and postsynaptic neurons in synapses of the hippocampus, cerebellum, and the cerebral cortex, as well as other parts of the brain and in peripheral tissues. Eight different types of mGluRs, labeled mGluR1 to mGluR8, are divided into groups I, II, and III. Receptor types are grouped based on receptor structure and physiological activity. www.MedChemExpress.com 1 mGluR Agonists, Antagonists, Inhibitors, Modulators & Activators (-)-Camphoric acid (1R,2S)-VU0155041 Cat. No.: HY-122808 Cat. No.: HY-14417A (-)-Camphoric acid is the less active enantiomer (1R,2S)-VU0155041, Cis regioisomer of VU0155041, is of Camphoric acid. Camphoric acid stimulates a partial mGluR4 agonist with an EC50 of 2.35 osteoblast differentiation and induces μM. glutamate receptor expression. Camphoric acid also significantly induced the activation of NF-κB and AP-1. Purity: ≥98.0% Purity: ≥98.0% Clinical Data: No Development Reported Clinical Data: No Development Reported Size: 10 mM × 1 mL, 100 mg Size: 10 mM × 1 mL, 5 mg, 10 mg, 25 mg (2R,4R)-APDC (R)-ADX-47273 Cat. No.: HY-102091 Cat. No.: HY-13058B (2R,4R)-APDC is a selective group II metabotropic (R)-ADX-47273 is a potent mGluR5 positive glutamate receptors (mGluRs) agonist. -

New Drug Evaluation Monograph Template
© Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 | Fax 503-947-1119 Class Update: Second Generation Antidepressant Medications Month/Year of Review: May 2014 Last Oregon Review: April 2012 PDL Classes: Psychiatric: Antidepressants Source Document: OSU College of Pharmacy New drug(s): vortioxetine (Brintellix®) Manufacturer: Takeda & Lundbeck/Forest levomilnacipran extended-release (Fetzima®) Dossier Received: Yes/Pending Current Status of Voluntary PDL Class: Preferred Agents: BUPROPION HCL TABLET/TABLET ER, CITALOPRAM TABLET/SOLUTION, FLUOXETINE CAPSULE/SOLUTION/TABLET, FLUVOXAMINE, MIRTAZEPINE TAB RAPDIS/TABLET, PAROXETINE TABLET, SERTRALINE ORAL CONC/TABLET, VENLAFAXINE TABLET, VENLAFAXINE ER Non-Preferred Agents: BUPROPRION XL, DESVENLAFAXINE (PRISTIQ ER), DULOXETINE (CYMBALTA®), ESCITALOPRAM, FLUOXETINE DF (PROZAC® WEEKLY), NEFAZODONE, PAROXETINE HCL (PAXIL CR®), SELEGILINE PATCH (ENSAM®), VILAZODONE (VIIBRYD®), OLANZAPINE/FLUOXETINE (SYMBYAX®) Status of the Voluntary Mental Health Preferred Drug List Currently, all antidepressants are available without prior authorization for non-preferred placement. Oregon law prohibits traditional methods of PDL enforcement on mental health drugs. Second generation antidepressants have been reviewed for clinical efficacy and safety and specific agents were chosen as clinically preferred; this eliminates a copay. Oregon’s Medicaid program currently -

Compositions for Treating Centrally Mediated
(19) TZZ Z_T (11) EP 2 722 045 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: A61K 31/4178 (2006.01) A61K 31/473 (2006.01) 06.07.2016 Bulletin 2016/27 A61K 31/496 (2006.01) A61K 31/573 (2006.01) A61K 45/06 (2006.01) A61K 9/00 (2006.01) (2006.01) (2006.01) (21) Application number: 14151683.1 A61K 9/20 A61K 9/48 A61P 1/08 (2006.01) (22) Date of filing: 18.11.2010 (54) Compositions for treating centrally mediated nausea and vomiting Zusammensetzungen zur Behandlung von zentral vermitteltem Unwohlsein und Erbrechen Compositions pour traiter les nausées et vomissements à médiation centrale (84) Designated Contracting States: (56) References cited: AL AT BE BG CH CY CZ DE DK EE ES FI FR GB WO-A1-2008/049552 WO-A2-2007/096763 GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR • REDDY G KESAVA ET AL: "Novel neurokinin-1 antagonists as antiemetics for the treatment of (30) Priority: 18.11.2009 US 262470 P chemotherapy-induced emesis.", SUPPORTIVE 14.09.2010 US 382709 P CANCER THERAPY 1 APR 2006 LNKD- PUBMED:18632487, vol. 3, no. 3, 1 April 2006 (43) Date of publication of application: (2006-04-01), pages140-142, XP002626039, ISSN: 23.04.2014 Bulletin 2014/17 1543-2912 • DIEMUNSCH P ET AL: "Neurokinin-1 receptor (62) Document number(s) of the earlier application(s) in antagonists in the prevention of postoperative accordance with Art. -

Effects of Serotonin in the Hippocampus: How Ssris and Multimodal Antidepressants Might Regulate Pyramidal Cell Function
CNS Spectrums (2016), 21, 143–161. © Cambridge University Press 2015. The online version of this article is published within an Open Access environment subject to the conditions of the Creative Commons Attribution-NonCommercial-ShareAlike licence <http://creativecommons.org/licenses/by-nc-sa/3.0/>. The written permission of Cambridge University Press must be obtained for commercial re-use. doi:10.1017/S1092852915000425 REVIEW ARTICLE Effects of serotonin in the hippocampus: how SSRIs and multimodal antidepressants might regulate pyramidal cell function Elena Dale,1* Alan L. Pehrson,1 Theepica Jeyarajah,1 Yan Li,1 Steven C. Leiser,1 Gennady Smagin,1 Christina K. Olsen,2 and Connie Sanchez1 1 Lundbeck Research USA, Paramus, New Jersey, USA 2 Lundbeck DK, Copenhagen-Valby, Denmark The hippocampus plays an important role in emotional and cognitive processing, and both of these domains are affected in patients with major depressive disorder (MDD). Extensive preclinical research and the notion that modulation of serotonin (5-HT) neurotransmission plays a key role in the therapeutic efficacy of selective serotonin reuptake inhibitors (SSRIs) support the view that 5-HT is important for hippocampal function in normal and disease- like conditions. The hippocampus is densely innervated by serotonergic fibers, and the majority of 5-HT receptor subtypes are expressed there. Furthermore, hippocampal cells often co-express multiple 5-HT receptor subtypes that can have either complementary or opposing effects on cell function, adding to the complexity of 5-HT neurotransmission. Here we review the current knowledge of how 5-HT, through its various receptor subtypes, modulates hippocampal output and the activity of hippocampal pyramidal cells in rodents. -

Stems for Nonproprietary Drug Names
USAN STEM LIST STEM DEFINITION EXAMPLES -abine (see -arabine, -citabine) -ac anti-inflammatory agents (acetic acid derivatives) bromfenac dexpemedolac -acetam (see -racetam) -adol or analgesics (mixed opiate receptor agonists/ tazadolene -adol- antagonists) spiradolene levonantradol -adox antibacterials (quinoline dioxide derivatives) carbadox -afenone antiarrhythmics (propafenone derivatives) alprafenone diprafenonex -afil PDE5 inhibitors tadalafil -aj- antiarrhythmics (ajmaline derivatives) lorajmine -aldrate antacid aluminum salts magaldrate -algron alpha1 - and alpha2 - adrenoreceptor agonists dabuzalgron -alol combined alpha and beta blockers labetalol medroxalol -amidis antimyloidotics tafamidis -amivir (see -vir) -ampa ionotropic non-NMDA glutamate receptors (AMPA and/or KA receptors) subgroup: -ampanel antagonists becampanel -ampator modulators forampator -anib angiogenesis inhibitors pegaptanib cediranib 1 subgroup: -siranib siRNA bevasiranib -andr- androgens nandrolone -anserin serotonin 5-HT2 receptor antagonists altanserin tropanserin adatanserin -antel anthelmintics (undefined group) carbantel subgroup: -quantel 2-deoxoparaherquamide A derivatives derquantel -antrone antineoplastics; anthraquinone derivatives pixantrone -apsel P-selectin antagonists torapsel -arabine antineoplastics (arabinofuranosyl derivatives) fazarabine fludarabine aril-, -aril, -aril- antiviral (arildone derivatives) pleconaril arildone fosarilate -arit antirheumatics (lobenzarit type) lobenzarit clobuzarit -arol anticoagulants (dicumarol type) dicumarol -

( 12 ) United States Patent
US009974742B2 (12 ) United States Patent (10 ) Patent No. : US 9 , 974 , 742 B2 Ottoboni et al. (45 ) Date of Patent: * May 22, 2018 ( 54 ) EMULSION FORMULATIONS OF AN NK - 1 2013 /0236501 A1 * 9 /2013 Booth . .. .. .. A61K 9 /0019 424 / 400 RECEPTOR ANTAGONIST AND USES 2013 /0317016 AL 11 /2013 Hingorani et al. THEREOF 2016 / 0024092 Al 1 / 2016 Wan et al. 2016 / 0082013 Al 3 / 2016 Ottoboni et al . @(71 ) Applicant : Heron Therapeutics , Inc. , Redwood 2016 /0206622 A1 3 / 2016 Ottoboni et al . City , CA (US ) 2017 / 0112847 AL 4 /2017 Ottoboni et al. @(72 ) Inventors : Thomas B . Ottoboni, Belmont, CA (US ) ; Han Han , Mountain View , CA FOREIGN PATENT DOCUMENTS (US ) CN 102379 * 3 / 2012 CN 102379845 A 3 / 2012 @(73 ) Assignee : Heron Therapeutics, Inc. , San Diego , WO WO 2005 /016308 AL 2 / 2005 WO WO 2009 / 124756 AL 10 / 2009 CA (US ) WO WO 2011 / 158053 AL 12 / 2011 WO WO 2013 / 177501 A2 11 / 2013 @( * ) Notice : Subject to any disclaimer , the term of this WO WO 2014 /0209962 AL 12 /2014 patent is extended or adjusted under 35 WO WO 2014 /005606 AL 3 / 2016 U . S . C . 154 ( b ) by 0 days . days . WO WO 2016 /044784 Al 3 / 2016 This patent is subject to a terminal dis claimer . OTHER PUBLICATIONS (21 ) Appl. No. : 15 /012 , 532 Cassileth et al. in Arch . Intern Med . 1983 ; 143 ( 7 ) : 1347 - 1349 ( Abstract ) . * Dexamethasone Hydrogen Phosphate at web .archive . org/ web / ( 22 ) Filed : Feb . 1 , 2016 20141224130045 /http :/ /www . drugs . com / pro / dexamethasone -so dium -phosphate .html ( retrieved on the internet Mar. -

GPCR/G Protein
Inhibitors, Agonists, Screening Libraries www.MedChemExpress.com GPCR/G Protein G Protein Coupled Receptors (GPCRs) perceive many extracellular signals and transduce them to heterotrimeric G proteins, which further transduce these signals intracellular to appropriate downstream effectors and thereby play an important role in various signaling pathways. G proteins are specialized proteins with the ability to bind the nucleotides guanosine triphosphate (GTP) and guanosine diphosphate (GDP). In unstimulated cells, the state of G alpha is defined by its interaction with GDP, G beta-gamma, and a GPCR. Upon receptor stimulation by a ligand, G alpha dissociates from the receptor and G beta-gamma, and GTP is exchanged for the bound GDP, which leads to G alpha activation. G alpha then goes on to activate other molecules in the cell. These effects include activating the MAPK and PI3K pathways, as well as inhibition of the Na+/H+ exchanger in the plasma membrane, and the lowering of intracellular Ca2+ levels. Most human GPCRs can be grouped into five main families named; Glutamate, Rhodopsin, Adhesion, Frizzled/Taste2, and Secretin, forming the GRAFS classification system. A series of studies showed that aberrant GPCR Signaling including those for GPCR-PCa, PSGR2, CaSR, GPR30, and GPR39 are associated with tumorigenesis or metastasis, thus interfering with these receptors and their downstream targets might provide an opportunity for the development of new strategies for cancer diagnosis, prevention and treatment. At present, modulators of GPCRs form a key area for the pharmaceutical industry, representing approximately 27% of all FDA-approved drugs. References: [1] Moreira IS. Biochim Biophys Acta. 2014 Jan;1840(1):16-33. -

Current Phase III Clinical Trials
Toolbox: Current phase III clinical trials Tiffany-Jade Kreys, PharmD1 1Assistant Professor of Pharmacy Practice University of the Incarnate Word Feik School of Pharmacy, San Antonio, Texas KEYWORDS clinical trials, depression, bipolar, schizophrenia, medications Table 1. Novel Agents for Major Depression, Bipolar Disorder, and/or Schizophrenia Product Name Sponsor Indication Mechanism of Action (previous name) Downloaded from http://meridian.allenpress.com/mhc/article-pdf/2/6/138/2094102/mhc_n129046.pdf by guest on 26 September 2021 Amitifadine Euthymics MDD Triple Reuptake Inhibitor (EB-1010) Bioscience (1:2:8 ratio of serotonin: norepinephrine: dopamine inhibition) Bitopertin Roche Schizophrenia Glycine reuptake inhibitor (RG1678) Enhances NMDA receptor activity Brexpiprazole Lundbeck MDD, Claimed to have "broad activity across multiple monoamine (OPC-34712) Otsuka America Schizophrenia systems and exhibits reduced partial agonist activity at D2 Pharmaceuticals receptors and enhanced affinity for specific serotonin receptors" Cariprazine Forest Laboratories BPAD, MDD adjunct D3-preferring/D2 receptor partial agonist (RGH-188) therapy, Schizophrenia Citalopram/ PharmaNeuro- MDD 5HT2A/D4 antagonist Pipamperone Boost (PNB01) Edivoxetine Eli Lilly MDD adjunct therapy Norepinephrine reuptake inhibitor (LY2216684) Vortioxetine Lundbeck MDD In vitro studies: 5HT3 and 7 receptor antagonist, 5HT1B partial (LUAA21004) Takeda agonist, 5HT1A agonist, serotonin transporter inhibitor Pharmaceuticals In vivo: Increases serotonin, norepinephrine, dopamine, -

Patent Application Publication ( 10 ) Pub . No . : US 2019 / 0192440 A1
US 20190192440A1 (19 ) United States (12 ) Patent Application Publication ( 10) Pub . No. : US 2019 /0192440 A1 LI (43 ) Pub . Date : Jun . 27 , 2019 ( 54 ) ORAL DRUG DOSAGE FORM COMPRISING Publication Classification DRUG IN THE FORM OF NANOPARTICLES (51 ) Int . CI. A61K 9 / 20 (2006 .01 ) ( 71 ) Applicant: Triastek , Inc. , Nanjing ( CN ) A61K 9 /00 ( 2006 . 01) A61K 31/ 192 ( 2006 .01 ) (72 ) Inventor : Xiaoling LI , Dublin , CA (US ) A61K 9 / 24 ( 2006 .01 ) ( 52 ) U . S . CI. ( 21 ) Appl. No. : 16 /289 ,499 CPC . .. .. A61K 9 /2031 (2013 . 01 ) ; A61K 9 /0065 ( 22 ) Filed : Feb . 28 , 2019 (2013 .01 ) ; A61K 9 / 209 ( 2013 .01 ) ; A61K 9 /2027 ( 2013 .01 ) ; A61K 31/ 192 ( 2013. 01 ) ; Related U . S . Application Data A61K 9 /2072 ( 2013 .01 ) (63 ) Continuation of application No. 16 /028 ,305 , filed on Jul. 5 , 2018 , now Pat . No . 10 , 258 ,575 , which is a (57 ) ABSTRACT continuation of application No . 15 / 173 ,596 , filed on The present disclosure provides a stable solid pharmaceuti Jun . 3 , 2016 . cal dosage form for oral administration . The dosage form (60 ) Provisional application No . 62 /313 ,092 , filed on Mar. includes a substrate that forms at least one compartment and 24 , 2016 , provisional application No . 62 / 296 , 087 , a drug content loaded into the compartment. The dosage filed on Feb . 17 , 2016 , provisional application No . form is so designed that the active pharmaceutical ingredient 62 / 170, 645 , filed on Jun . 3 , 2015 . of the drug content is released in a controlled manner. Patent Application Publication Jun . 27 , 2019 Sheet 1 of 20 US 2019 /0192440 A1 FIG . -
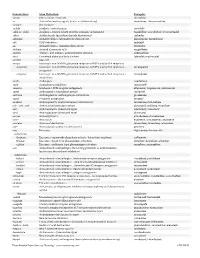
Downloaded from Survive Nursing | Survivenursing.Com V20110426
Generic Stem Stem Definition Examples -abine (see -arabine, -citabine) decitabine -ac Anti-inflammatory agents (acetic acid derivatives) bromfenac; dexpemedolac -acetam See -racetam -actide Synthetic corticotropins seractide -adol or -aldol- Analgesics (mixed opiate receptor agonists/ antagonists) tazadolene; spiradolene; levonantradol -adox Antibacterials (quinoline dioxide derivatives) carbadox -afenone Antiarrhythmics (propafenone derivatives) alprafenone; diprafenone -afil PDE5 inhibitors tadalafil -aj- Antiarrhythmics (ajmaline derivatives) lorajmine -aldrate Antacid aluminum salts magaldrate -algron Alpha1 - and alpha2 - adrenoreceptor agonists dabuzalgron -alol Combined alpha and beta blockers labetalol; medroxalol -amivir (see -vir) -ampa Ionotropic non-NMDA glutamate receptors (AMPA and/or KA receptors) -ampanel Ionotropic non-NMDA glutamate receptors (AMPA and/or KA receptors) ; becampanel antagonists -ampator Ionotropic non-NMDA glutamate receptors (AMPA and/or KA receptors) ; forampator modulators -andr- Androgens nandrolone -anib Angiogenesis inhibitors semaxanib -anserin Serotonin 5-HT2 receptor antagonists altanserin; tropanserin; adatanserin -antel Anthelmintics (undefined group) carbantel -antrone Antineoplastics; anthraquinone derivatives pixantrone -apsel P-selectin antagonists torapsel -arabine Antineoplastics (arabinofuranosyl derivatives) fazarabine; fludarabine aril-, -aril, -aril- Antiviral (arildone derivatives) pleconaril; arildone; fosarilate -arit Antirheumatics (lobenzarit type) lobenzarit; clobuzarit -arol