From Alchemy to Atoms-The Making of Plutonium
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Hitler and Heisenberg
The Wall Street Journal December 27-28, 2014 Book Review “Serving the Reich” by Philip Ball, Chicago, $30, 303 pages Hitler and Heisenberg Werner Heisenberg thought that once Germany had won, the ‘good Germans’ would get rid of the Nazis. Fizzing The museum inside the former basement tavern where in early 1945 German scientists built a small nuclear reactor. Associated Press by Jeremy Bernstein The German scientists who remained in Germany throughout the Nazi period can be divided into three main groups. At one end were the anti-Nazis—people who not only despised the regime but tried to do something about it. They were very few. Two that come to mind were Fritz Strassmann and Max von Laue. Strassmann was a radiochemist—a specialist in radioactive materials—who was blacklisted from university jobs by the Society of German Chemists in 1933 for his anti-Nazi views. He was fortunate to get a half-pay job at the Kaiser Wilhelm Institute for Chemistry, which got some of its funding from industry. He and his wife (they had a 3-year-old son) hid a Jewish friend in their apartment for months. With the radiochemist Otto Hahn, he performed the 1938 experiment in which fission was observed for the first time. Von Laue won the Nobel Prize in physics in 1914. Soon after Einstein published his 1905 paper on relativity, von Laue went to Switzerland to see him. They remained friends for life. Under the Nazis, Einstein’s work was denounced as “Jewish science.” When a number of von Laue’s colleagues agreed to use relativity but attribute it to some Aryan, he refused. -

Table 2.Iii.1. Fissionable Isotopes1
FISSIONABLE ISOTOPES Charles P. Blair Last revised: 2012 “While several isotopes are theoretically fissionable, RANNSAD defines fissionable isotopes as either uranium-233 or 235; plutonium 238, 239, 240, 241, or 242, or Americium-241. See, Ackerman, Asal, Bale, Blair and Rethemeyer, Anatomizing Radiological and Nuclear Non-State Adversaries: Identifying the Adversary, p. 99-101, footnote #10, TABLE 2.III.1. FISSIONABLE ISOTOPES1 Isotope Availability Possible Fission Bare Critical Weapon-types mass2 Uranium-233 MEDIUM: DOE reportedly stores Gun-type or implosion-type 15 kg more than one metric ton of U- 233.3 Uranium-235 HIGH: As of 2007, 1700 metric Gun-type or implosion-type 50 kg tons of HEU existed globally, in both civilian and military stocks.4 Plutonium- HIGH: A separated global stock of Implosion 10 kg 238 plutonium, both civilian and military, of over 500 tons.5 Implosion 10 kg Plutonium- Produced in military and civilian 239 reactor fuels. Typically, reactor Plutonium- grade plutonium (RGP) consists Implosion 40 kg 240 of roughly 60 percent plutonium- Plutonium- 239, 25 percent plutonium-240, Implosion 10-13 kg nine percent plutonium-241, five 241 percent plutonium-242 and one Plutonium- percent plutonium-2386 (these Implosion 89 -100 kg 242 percentages are influenced by how long the fuel is irradiated in the reactor).7 1 This table is drawn, in part, from Charles P. Blair, “Jihadists and Nuclear Weapons,” in Gary A. Ackerman and Jeremy Tamsett, ed., Jihadists and Weapons of Mass Destruction: A Growing Threat (New York: Taylor and Francis, 2009), pp. 196-197. See also, David Albright N 2 “Bare critical mass” refers to the absence of an initiator or a reflector. -

James Chadwick: Ahead of His Time
July 15, 2020 James Chadwick: ahead of his time Gerhard Ecker University of Vienna, Faculty of Physics Boltzmanngasse 5, A-1090 Wien, Austria Abstract James Chadwick is known for his discovery of the neutron. Many of his earlier findings and ideas in the context of weak and strong nuclear forces are much less known. This biographical sketch attempts to highlight the achievements of a scientist who paved the way for contemporary subatomic physics. arXiv:2007.06926v1 [physics.hist-ph] 14 Jul 2020 1 Early years James Chadwick was born on Oct. 20, 1891 in Bollington, Cheshire in the northwest of England, as the eldest son of John Joseph Chadwick and his wife Anne Mary. His father was a cotton spinner while his mother worked as a domestic servant. In 1895 the parents left Bollington to seek a better life in Manchester. James was left behind in the care of his grandparents, a parallel with his famous predecessor Isaac Newton who also grew up with his grandmother. It might be an interesting topic for sociologists of science to find out whether there is a correlation between children educated by their grandmothers and future scientific geniuses. James attended Bollington Cross School. He was very attached to his grandmother, much less to his parents. Nevertheless, he joined his parents in Manchester around 1902 but found it difficult to adjust to the new environment. The family felt they could not afford to send James to Manchester Grammar School although he had been offered a scholarship. Instead, he attended the less prestigious Central Grammar School where the teaching was actually very good, as Chadwick later emphasised. -

An Introduction to Isotopic Calculations John M
An Introduction to Isotopic Calculations John M. Hayes ([email protected]) Woods Hole Oceanographic Institution, Woods Hole, MA 02543, USA, 30 September 2004 Abstract. These notes provide an introduction to: termed isotope effects. As a result of such effects, the • Methods for the expression of isotopic abundances, natural abundances of the stable isotopes of practically • Isotopic mass balances, and all elements involved in low-temperature geochemical • Isotope effects and their consequences in open and (< 200°C) and biological processes are not precisely con- closed systems. stant. Taking carbon as an example, the range of interest is roughly 0.00998 ≤ 13F ≤ 0.01121. Within that range, Notation. Absolute abundances of isotopes are com- differences as small as 0.00001 can provide information monly reported in terms of atom percent. For example, about the source of the carbon and about processes in 13 13 12 13 atom percent C = [ C/( C + C)]100 (1) which the carbon has participated. A closely related term is the fractional abundance The delta notation. Because the interesting isotopic 13 13 fractional abundance of C ≡ F differences between natural samples usually occur at and 13F = 13C/(12C + 13C) (2) beyond the third significant figure of the isotope ratio, it has become conventional to express isotopic abundances These variables deserve attention because they provide using a differential notation. To provide a concrete the only basis for perfectly accurate mass balances. example, it is far easier to say – and to remember – that Isotope ratios are also measures of the absolute abun- the isotope ratios of samples A and B differ by one part dance of isotopes; they are usually arranged so that the per thousand than to say that sample A has 0.3663 %15N more abundant isotope appears in the denominator and sample B has 0.3659 %15N. -

Chapter 4. CLASSIFICATION UNDER the ATOMIC ENERGY
Chapter 4 CLASSIFICATION UNDER THE ATOMIC ENERGY ACT INTRODUCTION The Atomic Energy Act of 1946 was the first and, other than its successor, the Atomic Energy Act of 1954, to date the only U.S. statute to establish a program to restrict the dissemination of information. This Act transferred control of all aspects of atomic (nuclear) energy from the Army, which had managed the government’s World War II Manhattan Project to produce atomic bombs, to a five-member civilian Atomic Energy Commission (AEC). These new types of bombs, of awesome power, had been developed under stringent secrecy and security conditions. Congress, in enacting the 1946 Atomic Energy Act, continued the Manhattan Project’s comprehensive, rigid controls on U.S. information about atomic bombs and other aspects of atomic energy. That Atomic Energy Act designated the atomic energy information to be protected as “Restricted Data” and defined that data. Two types of atomic energy information were defined by the Atomic Energy Act of 1954, Restricted Data (RD) and a type that was subsequently termed Formerly Restricted Data (FRD). Before discussing further the Atomic Energy Act of 1946 and its unique requirements for controlling atomic energy information, some of the special information-control activities that accompanied the research, development, and production efforts that led to the first atomic bomb will be mentioned. Realization that an atomic bomb was possible had a profound impact on the scientists who first became aware of that possibility. The implications of such a weapon were so tremendous that the U.S. scientists conducting the initial, basic research related to nuclear fission voluntarily restricted the publication of their scientific work in this area. -

The Development of the Periodic Table and Its Consequences Citation: J
Firenze University Press www.fupress.com/substantia The Development of the Periodic Table and its Consequences Citation: J. Emsley (2019) The Devel- opment of the Periodic Table and its Consequences. Substantia 3(2) Suppl. 5: 15-27. doi: 10.13128/Substantia-297 John Emsley Copyright: © 2019 J. Emsley. This is Alameda Lodge, 23a Alameda Road, Ampthill, MK45 2LA, UK an open access, peer-reviewed article E-mail: [email protected] published by Firenze University Press (http://www.fupress.com/substantia) and distributed under the terms of the Abstract. Chemistry is fortunate among the sciences in having an icon that is instant- Creative Commons Attribution License, ly recognisable around the world: the periodic table. The United Nations has deemed which permits unrestricted use, distri- 2019 to be the International Year of the Periodic Table, in commemoration of the 150th bution, and reproduction in any medi- anniversary of the first paper in which it appeared. That had been written by a Russian um, provided the original author and chemist, Dmitri Mendeleev, and was published in May 1869. Since then, there have source are credited. been many versions of the table, but one format has come to be the most widely used Data Availability Statement: All rel- and is to be seen everywhere. The route to this preferred form of the table makes an evant data are within the paper and its interesting story. Supporting Information files. Keywords. Periodic table, Mendeleev, Newlands, Deming, Seaborg. Competing Interests: The Author(s) declare(s) no conflict of interest. INTRODUCTION There are hundreds of periodic tables but the one that is widely repro- duced has the approval of the International Union of Pure and Applied Chemistry (IUPAC) and is shown in Fig.1. -

Models of the Atomic Nucleus
Models of the Atomic Nucleus Second Edition Norman D. Cook Models of the Atomic Nucleus Unification Through a Lattice of Nucleons Second Edition 123 Prof. Norman D. Cook Kansai University Dept. Informatics 569 Takatsuki, Osaka Japan [email protected] Additional material to this book can be downloaded from http://extras.springer.com. ISBN 978-3-642-14736-4 e-ISBN 978-3-642-14737-1 DOI 10.1007/978-3-642-14737-1 Springer Heidelberg Dordrecht London New York Library of Congress Control Number: 2010936431 © Springer-Verlag Berlin Heidelberg 2006, 2010 This work is subject to copyright. All rights are reserved, whether the whole or part of the material is concerned, specifically the rights of translation, reprinting, reuse of illustrations, recitation, broadcasting, reproduction on microfilm or in any other way, and storage in data banks. Duplication of this publication or parts thereof is permitted only under the provisions of the German Copyright Law of September 9, 1965, in its current version, and permission for use must always be obtained from Springer. Violations are liable to prosecution under the German Copyright Law. The use of general descriptive names, registered names, trademarks, etc. in this publication does not imply, even in the absence of a specific statement, that such names are exempt from the relevant protective laws and regulations and therefore free for general use. Cover design: WMXDesign GmbH, Heidelberg Printed on acid-free paper Springer is part of Springer Science+Business Media (www.springer.com) Preface to the Second Edition Already by the 1970s, some theorists had declared that nuclear structure physics was a “closed chapter” in science, but since then it has repeatedly been found necessary to re-open this closed chapter to address old problems and to explain new phenom- ena. -

Compilation and Evaluation of Fission Yield Nuclear Data Iaea, Vienna, 2000 Iaea-Tecdoc-1168 Issn 1011–4289
IAEA-TECDOC-1168 Compilation and evaluation of fission yield nuclear data Final report of a co-ordinated research project 1991–1996 December 2000 The originating Section of this publication in the IAEA was: Nuclear Data Section International Atomic Energy Agency Wagramer Strasse 5 P.O. Box 100 A-1400 Vienna, Austria COMPILATION AND EVALUATION OF FISSION YIELD NUCLEAR DATA IAEA, VIENNA, 2000 IAEA-TECDOC-1168 ISSN 1011–4289 © IAEA, 2000 Printed by the IAEA in Austria December 2000 FOREWORD Fission product yields are required at several stages of the nuclear fuel cycle and are therefore included in all large international data files for reactor calculations and related applications. Such files are maintained and disseminated by the Nuclear Data Section of the IAEA as a member of an international data centres network. Users of these data are from the fields of reactor design and operation, waste management and nuclear materials safeguards, all of which are essential parts of the IAEA programme. In the 1980s, the number of measured fission yields increased so drastically that the manpower available for evaluating them to meet specific user needs was insufficient. To cope with this task, it was concluded in several meetings on fission product nuclear data, some of them convened by the IAEA, that international co-operation was required, and an IAEA co-ordinated research project (CRP) was recommended. This recommendation was endorsed by the International Nuclear Data Committee, an advisory body for the nuclear data programme of the IAEA. As a consequence, the CRP on the Compilation and Evaluation of Fission Yield Nuclear Data was initiated in 1991, after its scope, objectives and tasks had been defined by a preparatory meeting. -

VII. Nuclear Fission and Fusion Lorant Csige
VII.VII. NuclearNuclear FissionFission andand FusionFusion LorantLorant CsigeCsige LaboratoryLaboratory forfor NuclearNuclear PhysicsPhysics HungarianHungarian AcademyAcademy ofof SciencesSciences NuclearNuclear bindingbinding energy:energy: possibilitypossibility ofof energyenergy productionproduction NuclearNuclear FusionFusion ● Some basic principles: ─ two light nuclei should be very close to defeat the Coulomb repulsion → nuclear attraction: kinetic energy!! ─ not spontenious, difficult to achieve in reality ─ advantage: large amount of hydrogen and helium + solutions without producing any readioactive product NuclearNuclear fusionfusion inin starsstars ● proton-proton cycle: ~ mass of Sun, many branches ─ 2 + ν first step in all branches (Q=1.442 MeV): p+p → H+e +νe ● diproton formation (and immediate decay back to two protons) is the ruling process ● stable diproton is not existing → proton – proton fusion with instant beta decay! ● very slow process since weak interaction plays role → cross section has not yet been measrued experimentally (one proton „waits” 9 billion years to fuse) ─ second step: 2H+1H → 3He + γ + 5.49 MeV ● very fast process: only 4 seconds on the avarage ● 3He than fuse to produce 4He with three (four) differenct reactions (branches) ─ ppI branch: 3He + 3He → 4He + 1H + 1H + 12.86 MeV ─ ppII branch: ● 3He + 4He → 7Be + γ 7 7 ● 7 7 ν Be + e- → Li + νe ● 7Li + 1H → 4He + 4He ─ ppIII branch: only 0.11% energy of Sun, but source of neutrino problem ● 3He + 4He → 7Be + γ 7 1 8 8 + 4 4 ● 7 1 8 γ 8 + ν 4 4 Be + H → -
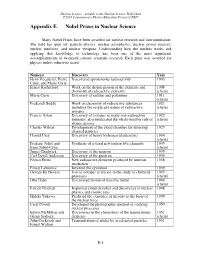
Appendix E Nobel Prizes in Nuclear Science
Nuclear Science—A Guide to the Nuclear Science Wall Chart ©2018 Contemporary Physics Education Project (CPEP) Appendix E Nobel Prizes in Nuclear Science Many Nobel Prizes have been awarded for nuclear research and instrumentation. The field has spun off: particle physics, nuclear astrophysics, nuclear power reactors, nuclear medicine, and nuclear weapons. Understanding how the nucleus works and applying that knowledge to technology has been one of the most significant accomplishments of twentieth century scientific research. Each prize was awarded for physics unless otherwise noted. Name(s) Discovery Year Henri Becquerel, Pierre Discovered spontaneous radioactivity 1903 Curie, and Marie Curie Ernest Rutherford Work on the disintegration of the elements and 1908 chemistry of radioactive elements (chem) Marie Curie Discovery of radium and polonium 1911 (chem) Frederick Soddy Work on chemistry of radioactive substances 1921 including the origin and nature of radioactive (chem) isotopes Francis Aston Discovery of isotopes in many non-radioactive 1922 elements, also enunciated the whole-number rule of (chem) atomic masses Charles Wilson Development of the cloud chamber for detecting 1927 charged particles Harold Urey Discovery of heavy hydrogen (deuterium) 1934 (chem) Frederic Joliot and Synthesis of several new radioactive elements 1935 Irene Joliot-Curie (chem) James Chadwick Discovery of the neutron 1935 Carl David Anderson Discovery of the positron 1936 Enrico Fermi New radioactive elements produced by neutron 1938 irradiation Ernest Lawrence -

Appendix: Key Concepts and Vocabulary for Nuclear Energy
Appendix: Key Concepts and Vocabulary for Nuclear Energy The likelihood of fission depends on, among other Power Plant things, the energy of the incoming neutron. Some In most power plants around the world, heat, usually nuclei can undergo fission even when hit by a low- produced in the form of steam, is converted to energy neutron. Such elements are called fissile. electricity. The heat could come through the burning The most important fissile nuclides are the uranium of coal or natural gas, in the case of fossil-fueled isotopes, uranium-235 and uranium-233, and the power plants, or the fission of uranium or plutonium plutonium isotope, plutonium-239. Isotopes are nuclei. The rate of electrical power production in variants of the same chemical element that have these power plants is usually measured in megawatts the same number of protons and electrons, but or millions of watts, and a typical large coal or nuclear differ in the number of neutrons. Of these, only power plant today produces electricity at a rate of uranium-235 is found in nature, and it is found about 1,000 megawatts. A much smaller physical only in very low concentrations. Uranium in nature unit, the kilowatt, is a thousand watts, and large contains 0.7 percent uranium-235 and 99.3 percent household appliances use electricity at a rate of a few uranium-238. This more abundant variety is an kilowatts when they are running. The reader will have important example of a nucleus that can be split only heard about the “kilowatt-hour,” which is the amount by a high-energy neutron. -

Cockcroft: a Novel This One, the Comprehensive Compilation Lutely Fundamental Discovery
~7~~-------------------------------------SPRINGBCXJKS------------------------N_A_ru_~__ v_o_L._Jo_s_~_AP __ Rr_L_r9M_ same, when it's compiled as successfully as what was in the sheer physics of it an abso Cockcroft: a novel this one, the comprehensive compilation lutely fundamental discovery. The authors perspective undeniably earns its place in the library. of this book take in their stride Cockcroft I was an admirer of Cockcroft, a fine, and Walton's being made to wait so long William Cooper stocky, sensible, cautious Yorkshireman, theirs not to reason why, they say. But what who indulged in a minimum of speech and about ours? Did it take the Nobel Cockcroft and the Atom. in practically no change of facial committee nineteen years to recognize the By Guy Hartcup and T.E. Allibone. expression. (That expression was neverthe experiment as breaking open an entirely Adam Hilger, Bristol!Heyden, Phila- less amiable, thoughtful, smiling.) If one new line of powerful experiments to be delphia: 1984. Pp.320. £18.95, $34. addressed a remark to him, it was only made with particle-accelerators? Or did it when, after the ensuing silence, he either take those members who opposed the took the famous black notebook out of his award all that time to disappear from the SrNCE the title of Hartcup and Allibone's pocket or actually said something, that one scene? Or what? Someone must know. book may sound a bit vague, perhaps I knew for certain that he'd heard. It gave me There's a fascinating detail I should really should begin by saying what Cockcroft and lasting pleasure to think that at a dinner like to be told.