Differences Between Intrinsic and Acquired Nucleoside Analogue
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
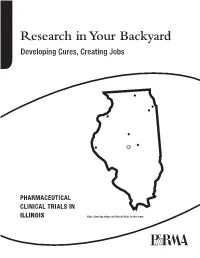
Research in Your Backyard Developing Cures, Creating Jobs
Research in Your Backyard Developing Cures, Creating Jobs PHARMACEUTICAL CLINICAL TRIALS IN ILLINOIS Dots show locations of clinical trials in the state. Executive Summary This report shows that biopharmaceutical research com- Quite often, biopharmaceutical companies hire local panies continue to be vitally important to the economy research institutions to conduct the tests and in Illinois, and patient health in Illinois, despite the recession. they help to bolster local economies in communities all over the state, including Chicago, Decatur, Joliet, Peoria, At a time when the state still faces significant economic Quincy, Rock Island, Rockford and Springfield. challenges, biopharmaceutical research companies are conducting or have conducted more than 4,300 clinical For patients, the trials offer another potential therapeutic trials of new medicines in collaboration with the state’s option. Clinical tests may provide a new avenue of care for clinical research centers, university medical schools and some chronic disease sufferers who are still searching for hospitals. Of the more than 4,300 clinical trials, 2,334 the medicines that are best for them. More than 470 of the target or have targeted the nation’s six most debilitating trials underway in Illinois are still recruiting patients. chronic diseases—asthma, cancer, diabetes, heart dis- ease, mental illnesses and stroke. Participants in clinical trials can: What are Clinical Trials? • Play an active role in their health care. • Gain access to new research treatments before they In the development of new medicines, clinical trials are are widely available. conducted to prove therapeutic safety and effectiveness and compile the evidence needed for the Food and Drug • Obtain expert medical care at leading health care Administration to approve treatments. -

WO 2012/063085 A3 18 May 2012 (18.05.2012) WIPOIPCT
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2012/063085 A3 18 May 2012 (18.05.2012) WIPOIPCT International Patent Classification: House, Wigan Lane, Wigan WN1 2TB (GB). PALIN, C07D 205/08 (2006.01) C07D 281/06 (2006.01) Ronald [GB/GB]; c/o Redx Pharma Limited, Douglas C07D 209/18 (2006.01) C07D 295/02 (2006.01) Bank House, Wigan Lane, Wigan WN1 2TB (GB). MUR C07D 213/60 (2006.01) C07D 307/00 (2006.01) RAY, Neil [GB/GB]; Redx Pharma Limited, Douglas Bank C07D 233/54 (2006.01) C07D 309/32 (2006.01) House, Wigan Lane, Wigan WN1 2TB (GB). LINDSAY, C07D 235/16 (2006.01) C07D 313/04 (2006.01) Derek [GB/GB]; c/o Redx Pharma Limited, Douglas Bank C07D 241/04 (2006.01) C07D 401/04 (2006.01) House, Wigan Lane, Wigan WN 1 2TB (GB). C07D 257/04 (2006.01) C07D 401/12 (2006.01) (74) Agent: HARRISON GODDARD FOOTE; C07D 277/40 (2006.01) Belgrave Hall, Belgrave Street, Leeds, Yorkshire LS2 8DD (GB). (21) International Application Number: PCT/GB2011/052211 (81) Designated States (unless otherwise indicated, for every kind of national protection available)·. AE, AG, AL, AM, (22) International Filing Date: AO, AT, AU, AZ, BA, BB, BG, BH, BR, BW, BY, BZ, 11 November 2011 (11.11.2011) CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, HN, (25) Filing Language: English HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, KR, (26) Publication Language: English KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, (30) Priority Data: OM, PE, PG, PH, PL, PT, QA, RO, RS, RU, RW, SC, SD, 1019078.3 11 November2010 (11.11.2010) GB SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, TR, 1019527.9 18 November 2010 (18.11.2010) GB TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. -

Protoporphyrin IX Is a Dual Inhibitor of P53/MDM2 and P53/MDM4
Jiang et al. Cell Death Discovery (2019) 5:77 https://doi.org/10.1038/s41420-019-0157-7 Cell Death Discovery ARTICLE Open Access Protoporphyrin IX is a dual inhibitor of p53/ MDM2 and p53/MDM4 interactions and induces apoptosis in B-cell chronic lymphocytic leukemia cells Liren Jiang 1,2,3, Natasha Malik 1, Pilar Acedo 1 and Joanna Zawacka-Pankau1 Abstract p53 is a tumor suppressor, which belongs to the p53 family of proteins. The family consists of p53, p63 and p73 proteins, which share similar structure and function. Activation of wild-type p53 or TAp73 in tumors leads to tumor regression, and small molecules restoring the p53 pathway are in clinical development. Protoporphyrin IX (PpIX), a metabolite of aminolevulinic acid, is a clinically approved drug applied in photodynamic diagnosis and therapy. PpIX induces p53-dependent and TAp73-dependent apoptosis and inhibits TAp73/MDM2 and TAp73/MDM4 interactions. Here we demonstrate that PpIX is a dual inhibitor of p53/MDM2 and p53/MDM4 interactions and activates apoptosis in B-cell chronic lymphocytic leukemia cells without illumination and without affecting normal cells. PpIX stabilizes p53 and TAp73 proteins, induces p53-downstream apoptotic targets and provokes cancer cell death at doses non-toxic to normal cells. Our findings open up new opportunities for repurposing PpIX for treating lymphoblastic leukemia with wild-type TP53. 1234567890():,; 1234567890():,; 1234567890():,; 1234567890():,; Introduction than women and leukemia incidence in both genders B-cell chronic lymphocytic leukemia (CLL) is one of the increases above the age of 554. most common forms of blood cancers1,2. The incidence of Common chromosomal aberrations in CLL include CLL in the western world is 4.2/100 000 per year. -

Chemotherapeutic Treatment of Acute Myeloid Leukemia
Clinical Trial Outcomes Mahdi, Mahdi & Knapper Chemotherapeutic treatment of acute myeloid leukemia 5 Clinical Trial Outcomes Chemotherapeutic treatment of acute myeloid leukemia: incorporating the results of recent clinical trials Clin. Invest. (Lond.) Better outcomes for patients with acute myeloid leukemia over the last 30 years have Ali J Mahdi*,1, Eamon J been largely achieved by improvements in supportive care measures rather than Mahdi1 & Steven Knapper1 therapeutic advances. The combination of daunorubicin and cytarabine has remained 1Cardiff University School of Medicine, Hematology Department, Heath Park, the standard of care for patients undergoing intensive induction–consolidation Cardiff CF14 4XN, UK treatment. In less fit older patients, low-dose cytarabine is the equivalent, although *Author for correspondence: the hypomethylating agent azacitidine may be challenging current practice. [email protected] Enhanced understanding of disease pathogenesis and therapy resistance has enabled the entry of novel chemotherapeutic and nonchemotherapeutic agents into clinical development with varied levels of activity. This article examines the evidence behind established chemotherapy practices for intensive and nonintensive acute myeloid leukemia treatments with an emphasis on emerging clinical trial data from novel chemotherapeutic and nonchemotherapeutic agents. Keywords: acute myeloid leukemia • aminopeptidase inhibitors • farnesyl transferase inhibitors • FLT3 inhibitors • gemtuzumab ozogamicin • histone deacytylase inhibitors • hypomethylating • Polo-like kinase 1 inhibitors Methodology AML cases [7]. Older patients over the age of 10.4155/CLI.14.112 An electronic database search (EMBASE, 60 years thus represent an important popula- MEDLINE and PubMed) was undertaken tion from both demographic and therapeutic to identify and review clinical studies in perspectives. Older patients frequently have acute myeloid leukemia (AML) undertaken multiple comorbidities, unfavorable cyto- between 1 January 2009 and 1 June 2014. -

Draft COMP Agenda 16-18 January 2018
12 January 2018 EMA/COMP/818236/2017 Inspections, Human Medicines Pharmacovigilance and Committees Committee for Orphan Medicinal Products (COMP) Draft agenda for the meeting on 16-18 January 2018 Chair: Bruno Sepodes – Vice-Chair: Lesley Greene 16 January 2018, 09:00-19:30, room 2F 17 January 2018, 08:30-19:30, room 2F 18 January 2018, 08:30-18:30, room 2F Health and safety information In accordance with the Agency’s health and safety policy, delegates are to be briefed on health, safety and emergency information and procedures prior to the start of the meeting. Disclaimers Some of the information contained in this agenda is considered commercially confidential or sensitive and therefore not disclosed. With regard to intended therapeutic indications or procedure scopes listed against products, it must be noted that these may not reflect the full wording proposed by applicants and may also vary during the course of the review. Additional details on some of these procedures will be published in the COMP meeting reports once the procedures are finalised. Of note, this agenda is a working document primarily designed for COMP members and the work the Committee undertakes. Note on access to documents Some documents mentioned in the agenda cannot be released at present following a request for access to documents within the framework of Regulation (EC) No 1049/2001 as they are subject to on- going procedures for which a final decision has not yet been adopted. They will become public when adopted or considered public according to the principles stated in the Agency policy on access to documents (EMA/127362/2006). -

WO 2010/135468 Al
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date 25 November 2010 (25.11.2010) WO 2010/135468 Al (51) International Patent Classification: [US/ES]; C/ Rio EsIa 77 PO2 A Villamayor, E-37007 GOlN 33/50 (2006.01) Salamanca (ES). LAGO, Santiago [GB/ES]; C/ Fernando Camino 15 3-E, E-29016 Malaga (ES). MATOSES, (21) International Application Number: Maria [ES/ES]; C/ Los Cristos 1 1 C, E-29008 Malaga PCT/US2010/035474 (ES). SUAREZ, Lilia [ES/ES]; C/ Ancla, 27 Casa 13, (22) International Filing Date: E-29720 CaIa Del Moral (ES). SAPIA, Sandra [IT/ES]; 19 May 2010 (19.05.2010) C/ Zapateros, 2 2 C, E-29005 Malaga (ES). BOSAN- QUET, Andrew [GB/GB]; 4 The Brow, Church Road, English (25) Filing Language: Combe Down, BA2 5JL, Bath (GB). GOR- (26) Publication Language: English ROCHATEGUI, Julian [ES/ES]; C/Puerto de los Leones 5, Majadahonda, E-28220 Madrid (ES). (30) Priority Data: TUDELA, Consuelo [ES/ES]; Av. Juan Carlos I 23, M a 61/179,685 19 May 2009 (19.05.2009) US jadahonda, E-28220 Madrid (ES). HERNANDEZ, Pilar (71) Applicant (for all designated States except US): VIVIA [ES/ES]; C/Uruguay 16, E-37OO3 Salamanca (ES). BIOTECH S.L. [ES/ES]; C/ Menendez Pelayo 12, 3d, CAVEDA, Luis Ignacio [ES/ES]; Av. Valladolid 17, E-47001 Valladolid (ES). E-28008 Madrid (ES). (72) Inventors; and (74) Agent: MALLON, Joseph, J.; Knobbe Martens Olson & (75) Inventors/Applicants (for US only): BALLESTEROS, Bear LLP, 2040 Main Street, 14th Floor, Irvine, CA Juan [ES/ES]; C/ Espalter 8 - 7 B, E-28014 Madrid (ES). -

20 13 Rep Or T
2013 REPORT More Than 240 Medicines in Development for Leukemia, Lymphoma and Other Blood Cancers Every 4 minutes a person is diagnosed with leukemia, Medicines in Development lymphoma or For Leading Blood Cancers myeloma; Accounting for Application Submitted 9% of all cancers Phase III diagnosed each year Phase II Biopharmaceutical research companies • 15 each for myeloproliferative neo- Phase I are developing 241 medicines for blood plasms, such as myelofibrosis, poly- cancers—leukemia, lymphoma and cythemia vera and essential throm- 97 98 myeloma. This report lists medicines in bocythemia; and for myelodysplastic human clinical trials or under review by syndromes, which are diseases affect- the U.S. Food and Drug Administration ing the blood and bone marrow. (FDA). These medicines in development offer The medicines in development include: hope for greater survival for the thou- sands of Americans who are affected by • 98 for lymphoma, including Hodgkin these cancers of the blood. and non-Hodgkin lymphoma, which 52 affect nearly 80,000 Americans each Definitions for the cancers listed in this year. report and other terms can be found on page 27. Links to sponsor company web • 97 for leukemia, including the four sites provide more information on the major types, which affect nearly potential products. 50,000 people in the United States 24 each year. For information on the value of medi- cines, an in-depth look at current in- • 52 for myeloma, a cancer of the novation and key medical breakthroughs plasma cells, which impacts more benefiting blood cancer patients, please than 22,000 people each year in the see Medicines in Development for Leu- United States. -

Acute Lymphoblastic Leukemia
European Working Group for adult Acute Lymphoblastic Leukemia Steering Committee Renato Bassan: [email protected] Hervé Dombret: [email protected] Roberto Foà: Manual of information for adult patients with acute [email protected] Nicola Gökbuget: lymphoblastic leukemia (ALL) [email protected] Dieter Hoelzer: [email protected] Table of contents Jose-Maria Ribera: [email protected] Roelof Willemze: 1. Introduction and objective of this manual [email protected] 2. What is acute lymphoblastic leukemia ? 3. Causes of acute lymphoblastic leukemia 4. Types of acute lymphoblastic leukemia 5. Symptoms of acute lymphoblastic leukemia Constitutional symptoms Symptoms derived from the infiltration of blasts in the bone marrow Symptoms derived from tissue and organ infiltration Other symptoms 6. Diagnosis of acute lymphoblastic leukemia 7. Treatment of acute lymphoblastic leukemia What does the treatment consist in? What complications and secondary effects does the treatment have? What results does treatment provide? Treatment of two special forms of acute lymphoblastic leukemia Treatment of relapse Controls after treatment, long-term effects of acute lymphoblastic leukemia and quality of life 8. Coping with acute lymphoblastic leukemia 9. Glossary 10. Sources of information Authors: European LeukemiaNet, Project 6, Acute Lymphoblastic Leukemia (January 2007) JM Ribera, Department Head, Department of Clinical Hematology, Institut Català d’Oncologia-Hospital Germans Trias i Pujol. JM Sancho, Medical Adjunct, Department of Clinical Hematology, Institut Català d’Oncologia-Hospital Germans Trias i Pujol Steering Committee: Rüdiger Hehlmann (Coordinator), Ute Berger (Scientific Network Manager), Marie C. Béné, Alan Burnett, Guido Finazzi, Christa Fonatsch, Eliane Gluckman, Nicola Gökbuget, David Grimwade, Torsten Haferlach, Michael Hallek, Jörg Hasford, Dieter Hoelzer, Per Ljungmann, Thomas Müller, Dietger Niederwieser, Steven O’Brien, Hubert Serve, Bengt Simonsson, Theo J. -

Purine-Metabolising Enzymes and Apoptosis in Cancer
cancers Review Purine-Metabolising Enzymes and Apoptosis in Cancer 1, , 2, 1 1 Marcella Camici * y , Mercedes Garcia-Gil y , Rossana Pesi , Simone Allegrini and Maria Grazia Tozzi 1 1 Dipartimento di Biologia, Unità di Biochimica, Via S. Zeno 51, 56127 Pisa, Italy 2 Dipartimento di Biologia, Unità di Fisiologia Generale, Via S. Zeno 31, 56127 Pisa, Italy * Correspondence: [email protected]; Tel.: +39-050 2211458 These authors equally contributed to the work. y Received: 24 July 2019; Accepted: 7 September 2019; Published: 12 September 2019 Abstract: The enzymes of both de novo and salvage pathways for purine nucleotide synthesis are regulated to meet the demand of nucleic acid precursors during proliferation. Among them, the salvage pathway enzymes seem to play the key role in replenishing the purine pool in dividing and tumour cells that require a greater amount of nucleotides. An imbalance in the purine pools is fundamental not only for preventing cell proliferation, but also, in many cases, to promote apoptosis. It is known that tumour cells harbour several mutations that might lead to defective apoptosis-inducing pathways, and this is probably at the basis of the initial expansion of the population of neoplastic cells. Therefore, knowledge of the molecular mechanisms that lead to apoptosis of tumoural cells is key to predicting the possible success of a drug treatment and planning more effective and focused therapies. In this review, we describe how the modulation of enzymes involved in purine metabolism in tumour cells may affect the apoptotic programme. The enzymes discussed are: ectosolic and cytosolic 50-nucleotidases, purine nucleoside phosphorylase, adenosine deaminase, hypoxanthine-guanine phosphoribosyltransferase, and inosine-50-monophosphate dehydrogenase, as well as recently described enzymes particularly expressed in tumour cells, such as deoxynucleoside triphosphate triphosphohydrolase and 7,8-dihydro-8-oxoguanine triphosphatase. -

Leukemia I N S I G H T S VOL
Leukemia INSIGHTS VOL. 17 • NO. 1 SPRING 2012 Slow Motion Breakthroughs in Adult ALL Therapy With a recent wave of new, highly active monoclonal for ALL is poor, however, with complete response antibodies and of a new BCR-ABL tyrosine kinase rates of 20 percent to 30 percent, depending on inhibitor (ponatinib), we are at the brink of prior therapy and duration of first remission. therapeutic breakthroughs which will significantly Median disease-free survival ranges from 2 to improve survival of adult acute lymphocytic 7.5 months. Long-term survival after ALL salvage leukemia (ALL). therapy is less than 10 percent. In this issue of Adult ALL encompasses a heterogeneous group Leukemia Insights, we focus on newer investigational of lymphoid malignancies. The two predominant strategies in adult ALL. subtypes are B-ALL and T-ALL, based on expression of B-lineage or T-lineage markers. Prognosis is New Monoclonal Antibodies in Pre-B ALL related to age, karyotype, molecular profile, immu- nophenotype, and other disease features. Prognosis The targeting of CD20 in ALL with combinations for pediatric ALL has improved significantly in the of rituximab and chemotherapy has improved past several decades; the current long-term survival survival in Burkitt leukemia and in CD20+ ALL. rate is greater than 80 percent. Long-term survival In the same light, several conjugated and unconjugated in adult ALL is 35 percent to 40 percent. The most monoclonal antibodies targeting CD22 and CD19 common reason for failure is disease recurrence. are under study as almost all ALL leukemic cells The hyper-CVAD regimen (hyperfractionated also express these markers. -

A Phase II Study of Omacetaxine (OM) in Patients with Intermediate-1 and Higher Risk Myelodysplastic Syndrome (MDS) Post Hypomethylating Agent (HMA) Failure 2013-0870
Protocol Page A Phase II Study of Omacetaxine (OM) in Patients with Intermediate-1 and Higher Risk Myelodysplastic Syndrome (MDS) post Hypomethylating Agent (HMA) Failure 2013-0870 Core Protocol Information Short Title Omacetaxine in Patients with Intermediate-1 and Higher Risk MDS post HMA Failure Study Chair: Elias Jabbour Additional Contact: Jhinelle L. Graham Vicky H. Zoeller Leukemia Protocol Review Group Additional Memo Recipients: Recipients List OPR Recipients (for OPR use only) None Study Staff Recipients None Department: Leukemia Phone: 713-792-4764 Unit: 0428 Full Title: A Phase II Study of Omacetaxine (OM) in Patients with Intermediate-1 and Higher Risk Myelodysplastic Syndrome (MDS) post Hypomethylating Agent (HMA) Failure Protocol Type: Standard Protocol Protocol Phase: Phase II Version Status: Activated -- Closed to new patient entry as of 08/05/2018 Version: 08 Document Status: Saved as "Final" Submitted by: Vicky H. Zoeller--9/11/2017 12:24:33 PM OPR Action: Accepted by: Margaret Okoloise -- 9/14/2017 12:09:50 PM Which Committee will review this protocol? The Clinical Research Committee - (CRC) Protocol Body 2013-0870 March 6, 2017 1 A Phase II Study of Omacetaxine (OM) in Patients with Intermediate-1 and Higher Risk Myelodysplastic Syndrome (MDS) post Hypomethylating Agent (HMA) Failure 2013-0870 March 6, 2017 2 Table of Contents 1.0 Objectives .................................................................................................. 3 2.0 Background .............................................................................................. -

WHO Drug Information Vol. 20, No. 3, 2006
WHO DRUG INFORMATION VOLUME 20• NUMBER 3 • 2006 RECOMMENDED INN LIST 56 INTERNATIONAL NONPROPRIETARY NAMES FOR PHARMACEUTICAL SUBSTANCES WORLD HEALTH ORGANIZATION • GENEVA WHO Drug Information Vol 20, No. 3, 2006 World Health Organization WHO Drug Information Contents Biomedicines and Vaccines Counterfeit taskforce launched 192 Fake artesunate warning sheet 193 Monoclonal antibodies: a special Resolution on counterfeiting 193 regulatory challenge? 165 Improving world health through regulation of biologicals 173 Regulatory Action and News Influenza virus vaccines for 2006–2007 Safety and Efficacy Issues northern hemisphere 196 Trastuzumab approved for primary Global progress in monitoring immuniza- breast cancer 196 tion adverse events 180 Emergency contraception over-the- Intracranial haemorrhage in patients counter 197 receiving tipranavir 180 Ocular Fusarium infections: ReNu Infliximab: hepatosplenic T cell MoistureLoc® voluntary withdrawal 197 lymphoma 181 Saquinavir: withdrawal of soft gel Lamotrigine: increased risk of non- capsule Fortovase® 197 syndromic oral clefts 181 Latest list of prequalified products and Biphosphonates: osteonecrosis of manufacturers 198 the jaw 181 Clopidogrel: new medical use 199 Enoxaparin dosage in chronic kidney Dronedarone: withdrawal of marketing disease 182 authorization 199 Terbinafine and life-threatening blood dyscrasias 183 Adverse reactions in children: why Recent Publications, report? 183 Hepatitis B reactivation and anti-TNF- Information and Events alpha agents 185 MSF issues ninth antiretroviral