Shapiro Reaction
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Synthetic Strategies to Access Biologically Important Fluorinated Motifs: Fluoroalkenes and Difluoroketones by Ming-Hsiu Yang Su
Synthetic Strategies to Access Biologically Important Fluorinated Motifs: Fluoroalkenes and Difluoroketones By Ming-Hsiu Yang Submitted to the graduate degree program in Medicinal Chemistry and the Graduate Faculty of the University of Kansas in partial fulfillment of the requirements for the degree of Doctor of Philosophy Chairperson Ryan A. Altman Michael D. Clift Apurba Dutta Michael F. Rafferty Jon A. Tunge Date Defended: April 26, 2017 The Dissertation Committee for Ming-Hsiu Yang certifies that this is the approved version of the following dissertation: Synthetic Strategies to Access Biologically Important Fluorinated Motifs: Fluoroalkenes and Difluoroketones Chairperson Ryan A. Altman Date Approved: April 26, 2017 ii Abstract Ming-Hsiu Yang Department of Medicinal Chemistry, April 2017 The University of Kansas Fluorine plays an important role in drug design, because of some unique features imparted by fluorine. The incorporation of fluorine into small molecules can modulate molecular physicochemical properties, metabolic stability, lipophilicity, and binding affinity to the target proteins. However, few fluorinated molecules are biosynthesized by enzymes. This means incorporating fluorine into the molecules relies on synthetic methods. Thus, efficient synthetic strategies to access the molecules bearing a variety of privileged fluorinated moieties are important for drug discovery. Fluoroalkenes are an isopolar and isosteric mimic of an amide bond with distinct biophysical properties, including decreased H-bond donating and accepting abilities, increased lipophilicity, and metabolic stability. Moreover, fluoroalkenes can also serve as probes for conducting conformational analyses of amides. These potential applications require the development of efficient methods to access fluoroalkenes. In chapter 2, a Shapiro fluorination strategy to access peptidomimetic fluoroalkenes is demonstrated. -

Syllabus CHEM 6352 2014
CHEM 6352 Organic Reactions & Synthesis Fall 2014 Jeremy A. May Office: 5025 SERC Office hours: T/Th 10-11 am or by appointment (email me) Email: [email protected] Website: http://mynsm.uh.edu/groups/maygroup/wiki/b24dc/Classes.html Lectures: 154 Fleming Tuesdays and Thursdays 8:30–10:00. August 26–December 6, 2014. Homework Session Saturdays 3:00 pm to 5:30 pm in Fleming 154/160/162. No class November 27–29, 2014 (Thanksgiving recess); Oct. 31st is last day to withdraw Optional Texts (on reserve at MD Anderson Library) Zweifel, G.; Nantz, M. “Modern Organic Synthesis: An Introduction” March, J. “Advanced Organic Chemistry” Corey, E. J.; Cheng, X.-M. “The Logic of Chemical Synthesis” Warren, S. “Designing Organic Syntheses: A Programmed Introduction to the Synthon Approach” Kürti, L.; Czakó, B. “Strategic Applications of Named Reactions in Organic Synthesis” Grossman, R. “The Art of Writing Reasonable Organic Reaction Mechanisms” Model Sets: Students are strongly encouraged to purchase at least one set. HGS biochemistry molecular model sets are recommended and are available at Research Stores in the Old Science Building. Other relevant texts and references: Greene; Wuts. “Protective Groups in Organic Synthesis” Nicolaou, K.C.; Sorensen, E. “Classics in Total Synthesis” Nicolaou, K.C.; Snyder, S. “Classics in Total Synthesis II” Larock, R. C. "Comprehensive Organic Transformations" Hartwig, J. “Organotransition Metal Chemistry: From Bonding to Catalysis” Tsuji, J. “Palladium Reagents and Catalysts” Hegedus, L. “Transition Metals in the Synthesis of Complex Organic Molecules” Problem Sets: Problem Sets will be distributed on Tuesdays (or before) and are due by the next Saturday at the Homework Session. -

Therapeutic Review Exploring Antimicrobial Potential of Hydrazones As Promising Lead
Available online a t www.derpharmachemica.com Scholars Research Library Der Pharma Chemica, 2011, 3(1):250-268 (http://derpharmachemica.com/archive.html) ISSN 0975-413X CODEN (USA): PCHHAX Therapeutic Review Exploring Antimicrobial Potential of Hydrazones as Promising Lead Goldie Uppal, Suman Bala*, Sunil Kamboj and Minaxi Saini M.M. College of Pharmacy, Maharishi Markandeshwar University, Ambala, Haryana, India ______________________________________________________________________________ ABSTRACT This review includes detailed study of structures of various hydrazones synthesized and evaluated for their antimicrobial activity. Hydrazone is a class of organic compounds with structure R 1R2C=NNH 2. Hydrazones containing an azomethine -NHN=CH- proton which leads to an important class of compounds for new drug development. Hydrazones are present in many of the bioactive heterocyclic compounds that are of very important use because of their various biological and clinical applications. Therefore, many researchers have synthesized these compounds as target structures and evaluated their antimicrobial activities. These observations have been guiding for the development of new hydrazones that possess varied biological activities. Key Words: Hydrazones, Hydrazide, Antifungal Activity, Antimicrobial Activity. ______________________________________________________________________________ INTRODUCTION The need to design new compounds to deal with the resistant strains has become one of the most important areas of research today. Hydrazone is a versatile moiety that exhibits a wide variety of biological activities. A hydrazone is a class of organic compounds with the structure R1R2C=NNH 2. Hydrazones are basically related to ketones and aldehydes. Hydrazones are formed by the replacement of the oxygen of carbonyl compounds with the -NNH 2 functional group. Hydrazones act as reactants in many important reactions e.g. -
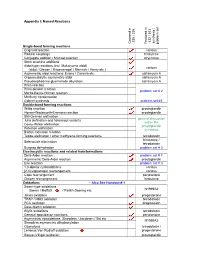
Appendix I: Named Reactions Single-Bond Forming Reactions Co
Appendix I: Named Reactions 235 / 335 432 / 533 synthesis / / synthesis Covered in Covered Featured in problem set problem Single-bond forming reactions Grignard reaction various Radical couplings hirstutene Conjugate addition / Michael reaction strychnine Stork enamine additions Aldol-type reactions (incl. Mukaiyama aldol) various (aldol / Claisen / Knoevenagel / Mannich / Henry etc.) Asymmetric aldol reactions: Evans / Carreira etc. saframycin A Organocatalytic asymmetric aldol saframycin A Pseudoephedrine glycinamide alkylation saframycin A Prins reaction Prins-pinacol reaction problem set # 2 Morita-Baylis-Hillman reaction McMurry condensation Gabriel synthesis problem set #3 Double-bond forming reactions Wittig reaction prostaglandin Horner-Wadsworth-Emmons reaction prostaglandin Still-Gennari olefination general discussion Julia olefination and heteroaryl variants within the Corey-Winter olefination prostaglandin Peterson olefination synthesis Barton extrusion reaction Tebbe olefination / other methylene-forming reactions tetrodotoxin hirstutene / Selenoxide elimination tetrodotoxin Burgess dehydration problem set # 3 Electrocyclic reactions and related transformations Diels-Alder reaction problem set # 1 Asymmetric Diels-Alder reaction prostaglandin Ene reaction problem set # 3 1,3-dipolar cycloadditions various [2,3] sigmatropic rearrangement various Cope rearrangement periplanone Claisen rearrangement hirstutene Oxidations – Also See Handout # 1 Swern-type oxidations (Swern / Moffatt / Parikh-Doering etc. N1999A2 Jones oxidation -

Convergent Total Synthesis and Preliminary Biological Investigations
Norrislide: Convergent Total Synthesis and Preliminary Biological Investigations Author: Krista Elizabeth Granger Persistent link: http://hdl.handle.net/2345/731 This work is posted on eScholarship@BC, Boston College University Libraries. Boston College Electronic Thesis or Dissertation, 2009 Copyright is held by the author, with all rights reserved, unless otherwise noted. Boston College The Graduate School of Arts and Sciences Department of Chemistry NORRISOLIDE: CONVERGENT TOTAL SYNTHESIS AND PRELIMINARY BIOLOGICAL INVESTIGATIONS a dissertation by KRISTA ELIZABETH GRANGER submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy August 2009 © copyright by KRISTA ELIZABETH GRANGER 2009 Norrisolide: Convergent Total Synthesis and Preliminary Biological Investigations Krista Elizabeth Granger Thesis Advisor: Professor Marc L. Snapper Abstract • Chapter 1: A review of Shapiro reactions as a coupling strategy in natural product total synthesis. The syntheses of lycoramine, galanthamine, yuehchukene analogues, ovalicin, studies toward the ingenol core, haemanthidine, pretazettine, tazettine, crinamine, Taxol, colombiasin A, elisapterosin B, the AB ring fragment of spongistatin 1 and 8-epipuupewhedione are discussed. Ar O S O nBuLi Li E+ E NH R' R' N R R R' R • Chapter 2: The convergent total synthesis of the marine natural product norrisolide is described. Both subunits, the hydrindane core and the norrisane side chain, are prepared in an asymmetric fashion through kinetic resolution and enantioselective cyclopropanation, respectively. A Shapiro reaction couples the two fragments and a Peterson olefination installs the 1,1-disubstituted olefin. O O MeO OTBS AcO MeO O O O O N Me Me Me MeO O O Me Li O OP H H Me Me Me Me norrisolide H Me Me • Chapter 3: Preliminary experiments to isolate the biological target of norrisolide through reductive alkylation and tritium labeling are investigated. -

Rearrangement and Trapping of Organozinc Carbenoids
Rearrangement and Trapping of Organozinc Carbenoids A Thesis Presented by Donogh John Roger O’Mahony In Partial Fulfilment of the Requirements for the Award of the Degree DOCTOR OF PHILOSOPHY of the UNIVERSITY OF LONDON Christopher Ingold Laboratories, Department of Chemistry, University College London, London WCIH OAJ. August 1995 ProQuest Number: 10016794 All rights reserved INFORMATION TO ALL USERS The quality of this reproduction is dependent upon the quality of the copy submitted. In the unlikely event that the author did not send a complete manuscript and there are missing pages, these will be noted. Also, if material had to be removed, a note will indicate the deletion. uest. ProQuest 10016794 Published by ProQuest LLC(2016). Copyright of the Dissertation is held by the Author. All rights reserved. This work is protected against unauthorized copying under Title 17, United States Code. Microform Edition © ProQuest LLC. ProQuest LLC 789 East Eisenhower Parkway P.O. Box 1346 Ann Arbor, Ml 48106-1346 ABSTRACT Abstract This thesis is divided into six chapters. Chapter one presents a review on metallocarbenoid chemistry and is divided into three parts. The first part gives a general survey of the influence of substituents on the reactivity and stability of free carbenes. The second part describes the reactions of transition metal carbenoids, particularly in relation to the oxidation state of the metal and the stoichiometry of carbenoid generation. The final part discusses the reactions of zinc carbenoids and the means of their formation. Chapter two is prefaced by a review on the generation and reactivity of organozinc carbenoids within the group, coupled with a mechanistic study of their formation. -

Shapiro Reaction
Robinson annulation The Robinson annulation is an organic reaction used to convert a ketone and an α,β-unsaturated ketone to a cyclohexenone using base. The mechanism begins with deprotonation with the base of the α-hydrogen of the ketone to form an enolate. The enolate then does a 1,4 addition to the conjugated olefin (Michael addition), which then abstracts a proton from water to form a diketone. Deprotonation of the other α-hydrogen with base forms another enolate which then does in intramolecular attack on the ketone group to give a cyclic alkoxy intermediate. Protonation of the alkoxy and a final elimination step result in the cyclo-hexenone produc. Mechanism The Wittig reaction or Wittig olefination is a chemical reaction of an aldehyde or ketone with a triphenyl phosphonium ylide (often called a Wittig reagent) to give an alkene and triphenylphosphine oxide. Reaction type:Nucleophilic Addition then Elimination Alkene formation from carbonyl compounds and phosphonium ylides, proceeding primarily through the proposed betaine and/or oxaphosphetane intermediates. The stereoselectivity can be controlled by the choice of ylide, carbonyl compound, and reaction conditions. When the ylide is replaced with a phosphine oxide carbanion, the reaction is referred to as the Horner reaction. When the ylide is replaced with a phosphonate carbanion, the reaction is referred to as the Horner-Emmons- Wadsworth reaction. The “Wittig Reaction” is one of the premier methods for the synthesis of alkenes. It uses a carbonyl compound as an electrophile, which is attacked by a “phosphorus ylide”. The Wittig reaction is nicely complementary to the aldol condensation, in which carbonyl compounds are attacked not by a phosphorus ylide but by an enolate. -

Name Reactions
Name Reactions A Collection of Detailed Reaction Mechanisms Bearbeitet von Jie Jack Li 1. Auflage 2002. Buch. xviii, 417 S. Hardcover ISBN 978 3 540 43024 7 Format (B x L): 15,5 x 23,5 cm Gewicht: 780 g Weitere Fachgebiete > Chemie, Biowissenschaften, Agrarwissenschaften > Chemie Allgemein Zu Leseprobe schnell und portofrei erhältlich bei Die Online-Fachbuchhandlung beck-shop.de ist spezialisiert auf Fachbücher, insbesondere Recht, Steuern und Wirtschaft. Im Sortiment finden Sie alle Medien (Bücher, Zeitschriften, CDs, eBooks, etc.) aller Verlage. Ergänzt wird das Programm durch Services wie Neuerscheinungsdienst oder Zusammenstellungen von Büchern zu Sonderpreisen. Der Shop führt mehr als 8 Millionen Produkte. Table of Contents Preface ................................................................................................................XIII Abbreviations.......................................................................................................XV 1. Abnormal Claisen rearrangement..............................................................1 2. Alder ene reaction......................................................................................2 3. Allan–Robinson reaction ...........................................................................3 4. Alper carbonylation...................................................................................5 5. Amadori glucosamine rearrangement........................................................7 6. Angeli–Rimini hydroxamic acid synthesis................................................8 -

The Merck Index
Browse Organic Name Reactions ● Preface ● 4CC ● Acetoacetic Ester Condensation ● Acetoacetic Ester Synthesis ● Acyloin Condensation ● Addition ● Akabori Amino Acid Reactions ● Alder (see Diels-Alder Reaction) ● Alder-Ene Reaction ● Aldol Reaction (Condensation) ● Algar-Flynn-Oyamada Reaction ● Allan-Robinson Reaction ● Allylic Rearrangements ● Aluminum Alkoxide Reduction ● Aluminum Alkoxide Reduction (see Meerwein-Ponndorf-Verley Reduction) ● Amadori Rearrangement ● Amidine and Ortho Ester Synthesis ● Aniline Rearrangement ● Arbuzov (see Michaelis-Arbuzov Reaction) ● Arens-van Dorp Synthesis ● Arndt-Eistert Synthesis ● Auwers Synthesis ● Babayan (see Favorskii-Babayan Synthesis) ● Bachmann (see Gomberg-Bachmann Reaction) ● Bäcklund (see Ramberg-Bäcklund Reaction) ● Baeyer-Drewson Indigo Synthesis ● Baeyer-Villiger Reaction ● Baker-Venkataraman Rearrangement ● Bakshi (see Corey-Bakshi-Shibata Reduction) ● Balz-Schiemann Reaction ● Bamberger Rearrangement ● Bamford-Stevens Reaction ● Barbier(-type) Reaction ● Barbier-Wieland Degradation ● Bart Reaction ● Barton Decarboxylation ● Barton Deoxygenation ● Barton Olefin Synthesis ● Barton Reaction http://themerckindex.cambridgesoft.com/TheMerckIndex/NameReactions/TOC.asp (1 of 17)19/4/2005 20:00:21 Browse Organic Name Reactions ● Barton-Kellogg Reaction ● Barton-McCombie Reaction ● Barton-Zard Reaction ● Baudisch Reaction ● Bauer (see Haller-Bauer Reaction) ● Baumann (see Schotten-Baumann Reaction) ● Baylis-Hillman Reaction ● Béchamp Reduction ● Beckmann Fragmentation ● Beckmann Rearrangement -

A Synthetic Route to Polymeric Carbohelicenes and Cyclic Derivatives
University of Nevada, Reno A Synthetic Route to Polymeric Carbohelicenes and Cyclic Derivatives A thesis submitted in partial fulfillment of the Requirements for the degree of Master of Science in Chemistry By William B. Thompson Dr. Benjamin T. King/Thesis Advisor December, 2014 i Abstract The King group’s interest is in the synthesis of polycylic aromatic hydrocarbons (PAHs) for their wide range of applications in materials, electronics, and mechanical properties.1 The primary focus of my own synthetic efforts in PAHs involves the production of polymeric carbohelicenes and their cyclic derivatives. Traditional methods for synthesizing helicenes structure use cross coupling of two phenanthrenes with desired functional groups. The final synthetic step usually involves a single reaction to close the ring and complete the helicenes such as Diels-Alder,2 carbenoid couplings,3or photocyclisation.4 Our own approach to synthesizing carbohelicenes is to synthesize a polymer precursor, poly{4,6-di((1E)-(2-2H)-propenyl])-m-phenylene), followed by a ring closing metathesis reaction (RCM) to stitch up the helicene. Our route is eight synthetic steps starting from commercially available m-xyene. The sequential steps involve bromination of m-xylene by electrophilic aromatic substitution, with a 70-74% yield, followed by a benzylic bromination with a 75-78% yield. Next a hydrolysis is performed using ZnBr2 in acetic acid yielding the dialdehyde in 75-80%. A double Wittig reaction follows using ethyltriphenylphosphonium bromide to obtain on average 65% yield of a mixture of EE, EZ, and ZZ isomers. Through optimization, we obtained a 95:5:0 ratio of our Wittig product (EE, EZ, and ZZ, checked by GC-MS). -

Wilkes, Antonia (2015) Towards the Synthesis of the ABC Tricycle of Taxol
Wilkes, Antonia (2015) Towards the synthesis of the ABC tricycle of Taxol. PhD thesis. http://theses.gla.ac.uk/5885/ Copyright and moral rights for this thesis are retained by the author A copy can be downloaded for personal non-commercial research or study, without prior permission or charge This thesis cannot be reproduced or quoted extensively from without first obtaining permission in writing from the Author The content must not be changed in any way or sold commercially in any format or medium without the formal permission of the Author When referring to this work, full bibliographic details including the author, title, awarding institution and date of the thesis must be given Glasgow Theses Service http://theses.gla.ac.uk/ [email protected] Towards the Synthesis of the ABC Tricycle of Taxol Antonia Wilkes (MChem) Thesis submitted in part fulfillment of the requirements for the Degree of Doctor of Philosophy School of Chemistry College of Science & Engineering July 2013 Abstract Taxol is one of the world’s most successful drugs used in the treatment of cancers. Isolated from the bark of the Pacific yew tree (Taxus brevifolia), it is a molecule of great interest within organic chemistry; with six total syntheses and a number of synthetic works having been published since its discovery. A semi-convergent synthesis of an intermediate in Holton’s synthesis was planned. The overall synthetic plan is shown below. The A ring would be installed by an intramolecular pinacol condensation. The BC bicycle would be closed by ring-closing metathesis at C10- C11. The ketone at C12 would be protected as an alkyne and the BC bicycle precursor would be obtained by coupling fragment A and the C ring. -

Name Reactions for Functional Group Transformations Published in 2007
Name Reactions for Homologations Edited by Jie Jack Li Copyright 0 2009 John Wiley & Sons, Inc. 678 Name Reactions for Homologations-1 Appendix 2, Table of Contents for Volume 2: Name Reactions for Functional Group Transformations Published in 2007 Chapter 1 Asymmetric Synthesis 1 1.1 CBS reduction 2 1.2 Davis chiral oxaziridine reagents 22 1.3 Midland reduction 40 1.4 Noyori catalytic asymmetric hydrogenation 46 1.5 Sharpless asymmetric hydroxylation reactions 67 Chapter 2 Reduction 85 2.1 Eschweiler-Clark reductive alkylation of amines 86 2.2 Gribble reduction of diaryl ketones 93 2.3 Luche reduction 112 2.4 Meerwein-Ponndorf-Verley reduction 123 2.5 Staudinger reaction 129 2.6 Wharton reaction 152 Chapter 3 Oxidation 159 3.1 Baeyer-Villiger oxidation 160 3.2 Brown hydroboration reaction 183 3.3 Burgess dehydrating reagent 189 3.4 Corey-Kim oxidation 207 3.5 Dess-Martin periodinane oxidation 218 3.6 Tamao-Kumada-Fleming oxidation 237 3.7 Martin's sulfurane dehydrating reagent 248 3.8 Oppenauer oxidation 265 3.9 Prilezhaev reaction 274 3.10 Rubottom oxidation 282 3.11 Swern oxidation 291 3.12 Wacker-Tsuji oxidation 309 3.13 Woodward cw-dihydroxylation 327 Chapter 4 Olefination 333 4.1 Chugaev elimination 334 4.2 Cope elimination reaction 343 4.3 Corey-Winter olefin synthesis 354 4.4 Perkin reaction (cinnamic acid synthesis) 363 4.5 Perkow vinyl phosphate synthesis 369 4.6 Ramberg-Backlund olefin synthesis 386 4.7 Shapiro reaction 405 Appendixes 679 4.8 Zaitsev elimination 414 Chapter 5 Amine Synthesis 423 5.1 Fukuyama amine synthesis