Chapter 12 Group 2: the Alkaline Earth Metals
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Comparison of Lead Calcium and Lead Selenium Alloys
A COMPARISON OF LEAD CALCIUM & LEAD SELENIUM ALLOYS Separating Fact From Fiction By: Carey O’Donnell & Chuck Finin Background Debate between lead antimony vs. lead calcium has been ongoing for almost 70 years Both are mature ‘technologies’, with major battery producers and users in both camps Batteries based on both alloy types have huge installed bases around the globe Time to take another look for US applications: • New market forces at work • Significant improvements in alloy compositions • Recognize that users are looking for viable options Objectives Provide a brief history of the development and use of both lead selenium (antimony) and lead calcium; objectively compare and contrast the performance and characteristics of each type To attempt to draw conclusions about the performance, reliability, and life expectancy of each alloy type; suitability of each for use in the US Then & Now: Primary Challenges in Battery Manufacturing The improvement of lead alloy compositions for increased tensile strength, improved casting, & conductive performance Developing better compositions & processes for the application and retention of active material on the grids Alloy Debate: Lead Calcium Vs. Lead Selenium Continues to dominate & define much of the technical and market debate in US Good reasons for this: • Impacts grid & product design, long-term product performance & reliability • Directly affects physical strength & hardness of grid; manufacturability • Influences grid corrosion & growth, retention of active material History of Antimony First -

Oregon Department of Human Services HEALTH EFFECTS INFORMATION
Oregon Department of Human Services Office of Environmental Public Health (503) 731-4030 Emergency 800 NE Oregon Street #604 (971) 673-0405 Portland, OR 97232-2162 (971) 673-0457 FAX (971) 673-0372 TTY-Nonvoice TECHNICAL BULLETIN HEALTH EFFECTS INFORMATION Prepared by: Department of Human Services ENVIRONMENTAL TOXICOLOGY SECTION Office of Environmental Public Health OCTOBER, 1998 CALCIUM CARBONATE "lime, limewater” For More Information Contact: Environmental Toxicology Section (971) 673-0440 Drinking Water Section (971) 673-0405 Technical Bulletin - Health Effects Information CALCIUM CARBONATE, "lime, limewater@ Page 2 SYNONYMS: Lime, ground limestone, dolomite, sugar lime, oyster shell, coral shell, marble dust, calcite, whiting, marl dust, putty dust CHEMICAL AND PHYSICAL PROPERTIES: - Molecular Formula: CaCO3 - White solid, crystals or powder, may draw moisture from the air and become damp on exposure - Odorless, chalky, flat, sweetish flavor (Do not confuse with "anhydrous lime" which is a special form of calcium hydroxide, an extremely caustic, dangerous product. Direct contact with it is immediately injurious to skin, eyes, intestinal tract and respiratory system.) WHERE DOES CALCIUM CARBONATE COME FROM? Calcium carbonate can be mined from the earth in solid form or it may be extracted from seawater or other brines by industrial processes. Natural shells, bones and chalk are composed predominantly of calcium carbonate. WHAT ARE THE PRINCIPLE USES OF CALCIUM CARBONATE? Calcium carbonate is an important ingredient of many household products. It is used as a whitening agent in paints, soaps, art products, paper, polishes, putty products and cement. It is used as a filler and whitener in many cosmetic products including mouth washes, creams, pastes, powders and lotions. -

The Periodic Table of Elements
The Periodic Table of Elements 1 2 6 Atomic Number = Number of Protons = Number of Electrons HYDROGENH HELIUMHe 1 Chemical Symbol NON-METALS 4 3 4 C 5 6 7 8 9 10 Li Be CARBON Chemical Name B C N O F Ne LITHIUM BERYLLIUM = Number of Protons + Number of Neutrons* BORON CARBON NITROGEN OXYGEN FLUORINE NEON 7 9 12 Atomic Weight 11 12 14 16 19 20 11 12 13 14 15 16 17 18 SODIUMNa MAGNESIUMMg ALUMINUMAl SILICONSi PHOSPHORUSP SULFURS CHLORINECl ARGONAr 23 24 METALS 27 28 31 32 35 40 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 POTASSIUMK CALCIUMCa SCANDIUMSc TITANIUMTi VANADIUMV CHROMIUMCr MANGANESEMn FeIRON COBALTCo NICKELNi CuCOPPER ZnZINC GALLIUMGa GERMANIUMGe ARSENICAs SELENIUMSe BROMINEBr KRYPTONKr 39 40 45 48 51 52 55 56 59 59 64 65 70 73 75 79 80 84 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 RUBIDIUMRb STRONTIUMSr YTTRIUMY ZIRCONIUMZr NIOBIUMNb MOLYBDENUMMo TECHNETIUMTc RUTHENIUMRu RHODIUMRh PALLADIUMPd AgSILVER CADMIUMCd INDIUMIn SnTIN ANTIMONYSb TELLURIUMTe IODINEI XeXENON 85 88 89 91 93 96 98 101 103 106 108 112 115 119 122 128 127 131 55 56 72 73 74 75 76 77 78 79 80 81 82 83 84 85 86 CESIUMCs BARIUMBa HAFNIUMHf TANTALUMTa TUNGSTENW RHENIUMRe OSMIUMOs IRIDIUMIr PLATINUMPt AuGOLD MERCURYHg THALLIUMTl PbLEAD BISMUTHBi POLONIUMPo ASTATINEAt RnRADON 133 137 178 181 184 186 190 192 195 197 201 204 207 209 209 210 222 87 88 104 105 106 107 108 109 110 111 112 113 114 115 116 117 118 FRANCIUMFr RADIUMRa RUTHERFORDIUMRf DUBNIUMDb SEABORGIUMSg BOHRIUMBh HASSIUMHs MEITNERIUMMt DARMSTADTIUMDs ROENTGENIUMRg COPERNICIUMCn NIHONIUMNh -

Llfltfits Paul Stanek, MST-6
COAJF- 9 50cDo/-~^0 LA-UR- 9 5-1666 Title: AGE HARDENING IN RAPIDLY SOLIDIFIED AND HOT ISOSTATICALLY PRESSED BERYLLIUM-ALUM1UM SILVER ALLOYS HVAUli Author(s): David H. Carter, MST-6 Andrew McGeorge, MST-6 Loren A. Jacobson, MST-6 llfltfits Paul Stanek, MST-6 IflllS,Ic 8-o° ,S8 si Proceedings of TMS Annual Meeting, Las Vegas, jJffilJSif—- Nevada, February 1995 as-IVUiS' i in Los Alamos NATIONAL LABORATORY Los Alamos National Laboratory, an affirmative action/equal opportunity employer, is operated by the University of California for the U.S. Department of Energy under contract W-7405-ENG-36. By acceptance of this article, the publisher recognizes that the U.S. Government retains a nonexclusive, royalty-free license to publish or reproduce the published form of this contribution, or to allow others to do so, for U.S. Government purposes. The Los Alamos National Laboratory requests that the publisher identify this article as work performed under the auspices of the U.S. Department of Energy* FotmNo.B36R5 MCTOIQimnM f\c mie hAMMr&T 10 i mi in ST26291M1 DISCLAIMER Portions of this document may be illegible in electronic image products. Images are produced from the best available original document. Age Hardening in Rapidly Solidified and Hot Isostatically Pressed Beryllium-Aluminum-Silver Alloys David H. Carter, Andrew C. McGeorge,* Loren A. Jacobson, and Paul W. Stanek Los Alamos National Laboratory, Los Alamos NM 87545 Abstract Three different alloys of beryllium, aluminum and silver were processed to powder by centrifugal atomization in a helium atmosphere. Alloy com• positions were, by weight, 50% Be, 47.5% Al, 2.5% Ag, 50% Be, 47% Al, 3% Ag, and 50% Be, 46% Al, 4% Ag. -

Of the Periodic Table
of the Periodic Table teacher notes Give your students a visual introduction to the families of the periodic table! This product includes eight mini- posters, one for each of the element families on the main group of the periodic table: Alkali Metals, Alkaline Earth Metals, Boron/Aluminum Group (Icosagens), Carbon Group (Crystallogens), Nitrogen Group (Pnictogens), Oxygen Group (Chalcogens), Halogens, and Noble Gases. The mini-posters give overview information about the family as well as a visual of where on the periodic table the family is located and a diagram of an atom of that family highlighting the number of valence electrons. Also included is the student packet, which is broken into the eight families and asks for specific information that students will find on the mini-posters. The students are also directed to color each family with a specific color on the blank graphic organizer at the end of their packet and they go to the fantastic interactive table at www.periodictable.com to learn even more about the elements in each family. Furthermore, there is a section for students to conduct their own research on the element of hydrogen, which does not belong to a family. When I use this activity, I print two of each mini-poster in color (pages 8 through 15 of this file), laminate them, and lay them on a big table. I have students work in partners to read about each family, one at a time, and complete that section of the student packet (pages 16 through 21 of this file). When they finish, they bring the mini-poster back to the table for another group to use. -

Radionuclides (Including Radon, Radium and Uranium)
Radionuclides (including Radon, Radium and Uranium) Hazard Summary Uranium, radium, and radon are naturally occurring radionuclides found in the environment. No information is available on the acute (short-term) noncancer effects of the radionuclides in humans. Animal studies have reported inflammatory reactions in the nasal passages and kidney damage from acute inhalation exposure to uranium. Chronic (long-term) inhalation exposure to uranium and radon in humans has been linked to respiratory effects, such as chronic lung disease, while radium exposure has resulted in acute leukopenia, anemia, necrosis of the jaw, and other effects. Cancer is the major effect of concern from the radionuclides. Radium, via oral exposure, is known to cause bone, head, and nasal passage tumors in humans, and radon, via inhalation exposure, causes lung cancer in humans. Uranium may cause lung cancer and tumors of the lymphatic and hematopoietic tissues. EPA has not classified uranium, radon or radium for carcinogenicity. Please Note: The main sources of information for this fact sheet are EPA's Integrated Risk Information System (IRIS) (5), which contains information on oral chronic toxicity and the RfD for uranium, and the Agency for Toxic Substances and Disease Registry's (ATSDR's) Toxicological Profiles for Uranium, Radium, and Radon. (1) Uses Uranium is used in nuclear power plants and nuclear weapons. Very small amounts are used in photography for toning, in the leather and wood industries for stains and dyes, and in the silk and wood industries. (2) Radium is used as a radiation source for treating neoplastic diseases, as a radon source, in radiography of metals, and as a neutron source for research. -

Beryllium Compounds (A) BERYLLIUM COMPOUNDS
Beryllium Compounds (A) BERYLLIUM COMPOUNDS 107-02-8 Hazard Summary Inhalation exposure to beryllium primarily occurs in the workplaces where it is mined, processed, or converted into alloys and chemicals, or from the burning of coal or fuel oil and in tobacco smoke. Acute (short-term) inhalation exposure to high levels of beryllium has been observed to cause inflammation of the lungs or acute pneumonitis (reddening and swelling of the lungs) in humans; after exposure ends, these symptoms may be reversible. Chronic (long-term) inhalation exposure of humans to beryllium has been reported to cause chronic beryllium disease (berylliosis), in which granulomatous lesions (noncancerous) develop in the lung. Human epidemiology studies are limited, but suggest a causal relationship between beryllium exposure and an increased risk of lung cancer. Inhalation exposure to beryllium has been demonstrated to cause lung cancer in rats and monkeys. EPA has classified beryllium as a Group B1, probable human carcinogen. Please Note: The main sources of information for this fact sheet are EPA's Integrated Risk Information System (IRIS) (3), which contains information on oral chronic toxicity and the RfD and inhalation chronic toxicity and the RfC, and the carcinogenic effects of beryllium including the unit cancer risk for inhalation exposure, EPA's Toxicological Review of Beryllium and Compounds (2), and the Agency for Toxic Substances and Disease Registry's (ATSDR's) Toxicological Profile for Beryllium. (1) Uses Pure beryllium and its metal alloys have applications in electrical components, tools, structural components for aircraft, missiles, and satellites, and other metal-fabricating uses. (1) Beryllium is also used in consumer products, such as televisions, calculators, and personal computers. -

Calcium Fluoride Scale Prevention
Tech Manual Excerpt Water Chemistry and Pretreatment Calcium Fluoride Scale Prevention Calcium Fluoride Fluoride levels in the feedwater of as low as 0.1 mg/L can create a scaling potential if Scale Prevention the calcium concentration is high. The calculation of the scaling potential is analogous to the procedure described in Calcium Sulfate Scale Prevention (Form No. 45-D01553-en) for CaSO4. Calculation 1. Calculate the ionic strength of the concentrate stream (Ic) following the procedure described in Scaling Calculations (Form No. 45-D01551-en): Eq. 1 2. Calculate the ion product (IPc) for CaF2 in the concentrate stream: Eq. 2 where: m 2+ 2+ ( Ca )f = M Ca in feed, mol/L m – – ( F )f = M F in feed, mol/L 3. Compare IPc for CaF2 with the solubility product (Ksp) of CaF2 at the ionic strength of the concentrate stream, Figure 2 /11/. If IPc ≥ Ksp, CaF2 scaling can occur, and adjustment is required. Adjustments for CaF2 Scale Control The adjustments discussed in Calcium Sulfate Scale Prevention (Form No. 45-D01553-en) for CaSO4 scale control apply as well for CaF2 scale control. Page 1 of 3 Form No. 45-D01556-en, Rev. 4 April 2020 Figure 1: Ksp for SrSO4 versus ionic strength /10/ Page 2 of 3 Form No. 45-D01556-en, Rev. 4 April 2020 Figure 2: Ksp for CaF2 versus ionic strength /11/ Excerpt from FilmTec™ Reverse Osmosis Membranes Technical Manual (Form No. 45-D01504-en), Chapter 2, "Water Chemistry and Pretreatment." Have a question? Contact us at: All information set forth herein is for informational purposes only. -

Periodic Activity of Metals Periodic Trends and the Properties of the Elements SCIENTIFIC
Periodic Activity of Metals Periodic Trends and the Properties of the Elements SCIENTIFIC Introduction Elements are classified based on similarities, differences, and trends in their properties, including their chemical reactions. The reactions of alkali and alkaline earth metals with water are pretty spectacular chemical reactions. Mixtures bubble and boil, fizz and hiss, and may even smoke and burn. Introduce the study of the periodic table and periodic trends with this exciting demonstration of the activity of metals. Concepts • Alkali and alkaline earth metals • Periodic table and trends • Physical and chemical properties • Metal activity Materials Calcium turnings, Ca, 0.3 g Beaker, Berzelius (tall-form), Pyrex®, 500-mL, 4 Lithium metal, Li, precut piece Forceps or tongs Magnesium ribbon, Mg, 3-cm Knife (optional) Sodium metal, Na, precut piece Petri dishes, disposable, 4 Phenolphthalein, 1% solution, 2 mL Scissors Water, distilled or deionized, 600 mL Safety Precautions Lithium and sodium are flammable, water-reactive, corrosive solids; dangerous when exposed to heat or flame. They react violently with water to produce flammable hydrogen gas and solutions of corrosive metal hydroxides. Hydrogen gas may be released in sufficient quantities to cause ignition. Do NOT “scale up” this demonstration using larger pieces of sodium or lithium! These metals are shipped in dry mineral oil. Store them in mineral oil until immediately before use. Do not allow these metals to stand exposed to air from one class period to another or for extended periods of time. Purchasing small, pre-cut pieces of lithium and sodium greatly reduces their potential hazard. Calcium metal is flammable in finely divided form and reacts upon contact with water to give flammable hydrogen gas and corrosive calcium hydroxide. -
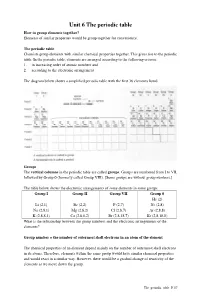
Unit 6 the Periodic Table How to Group Elements Together? Elements of Similar Properties Would Be Group Together for Convenience
Unit 6 The periodic table How to group elements together? Elements of similar properties would be group together for convenience. The periodic table Chemists group elements with similar chemical properties together. This gives rise to the periodic table. In the periodic table, elements are arranged according to the following criteria: 1. in increasing order of atomic numbers and 2. according to the electronic arrangement The diagram below shows a simplified periodic table with the first 36 elements listed. Groups The vertical columns in the periodic table are called groups . Groups are numbered from I to VII, followed by Group 0 (formerly called Group VIII). [Some groups are without group numbers.] The table below shows the electronic arrangements of some elements in some groups. Group I Group II Group VII Group 0 He (2) Li (2,1) Be (2,2) F (2,7) Ne (2,8) Na (2,8,1) Mg (2,8,2) Cl (2,8,7) Ar (2,8,8) K (2,8,8,1) Ca (2,8,8,2) Br (2,8,18,7) Kr (2,8,18,8) What is the relationship between the group numbers and the electronic arrangements of the elements? Group number = the number of outermost shell electrons in an atom of the element The chemical properties of an element depend mainly on the number of outermost shell electrons in its atoms. Therefore, elements within the same group would have similar chemical properties and would react in a similar way. However, there would be a gradual change of reactivity of the elements as we move down the group. -

Beryllium and Beryllium Compounds
BERYLLIUM AND BERYLLIUM COMPOUNDS Beryllium and beryllium compounds were considered by previous IARC Working Groups in 1971, 1979, 1987, and 1993 (IARC, 1972, 1980, 1987, 1993). Since that time, new data have become available, these have been incorporated in the Monograph, and taken into consid- eration in the present evaluation. 1. Exposure Data a very high strength-to-weight ratio. Beryllium is lighter than aluminium but is greater than 40% 1.1 Identification of the agents more rigid than steel. It has excellent electrical and thermal conductivities. Its only markedly Synonyms and molecular formulae for beryl- adverse feature is relatively pronounced brittle- lium, beryllium–aluminium and beryllium– ness, which restricts the use of metallic beryl- copper alloys, and certain beryllium compounds lium to specialized applications (WHO, 1990). are presented in Table 1.1. The list is not exhaus- Because of its low atomic number, beryllium tive, nor does it comprise necessarily the most is very permeable to X-rays. Neutron emission commercially important beryllium-containing after bombardment with α or γ rays is the most substances; rather, it indicates the range of beryl- important of its nuclear physical properties, lium compounds available. and beryllium can be used as a neutron source. Moreover, its low neutron absorptiveness and high-scattering cross-section make it a suitable 1.2 Chemical and physical properties moderator and reflector in structural materials of the agents in nuclear facilities; where most other metals absorb neutrons emitted during the fission Beryllium (atomic number, 4; relative atomic of nuclear fuel, beryllium atoms only reduce mass, 9.01) is a metal, which belongs to Group the energy of such neutrons, and reflect them IIA of the Periodic Table. -

Genius of the Periodic Table
GENIUS OF THE PERIODIC TABLE "Isn't it the work of a genius'. " exclaimed Academician V.I. Spitsyn, USSR, a member of the Scientific Advisory Committee when talking to an Agency audience in January. His listeners shared his enthusiasm. Academician Spitsyn was referring to the to the first formulation a hundred years ago by Professor Dmitry I. Mendeleyev of the Periodic Law of Elements. In conditions of enormous difficulty, considering the lack of data on atomic weights of elements, Mendeleyev created in less than two years work at St. Petersburg University, a system of chemical elements that is, in general, still being used. His law became a powerful instrument for further development of chemistry and physics. He was able immediately to correct the atomic weight numbers of some elements, including uranium, whose atomic weight he found to be double that given at the time. Two years later Mendeleyev went so far as to give a detailed description of physical or chemical properties of some elements which were as yet undiscovered. Time gave striking proof of his predictions and his periodic law. Mendeleyev published his conclusions in the first place by sending, early in March 186 9, a leaflet to many Russian and foreign scientists. It gave his system of elements based on their atomic weights and chemical resemblance. On the 18th March that year his paper on the subject was read at the meeting of the Russian Chemical Society, and two months later the Society's Journal published his article entitled "The correlation between properties of elements and their atomic weight".