WO 2018/218208 Al 29 November 2018 (29.11.2018) W !P O PCT
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Investigator Initiated Study IRB-29839 an Open-Label Pilot Study To
Investigator Initiated Study IRB-29839 An open-label pilot study to evaluate the efficacy and safety of a combination treatment of Sonidegib and BKM120 for the treatment of advanced basal cell carcinomas Version 05 September 2016 NCT02303041 DATE: 12Dec2018 1 Coordinating Center Stanford Cancer Center 875 Blake Wilbur Drive Stanford, CA 94305 And 450 Broadway, MC 5334 Redwood City, CA 94603 Protocol Director and Principal Investigator Anne Lynn S Chang, MD, Director of Dermatological Clinical Trials 450 Broadway St, MC 5334 Redwood City, CA 94603 [email protected] Co-Investigator Anthony Oro, MD PhD 450 Broadway St, MC 5334 Redwood City, CA 94603 [email protected] Biostatistician Shufeng Li, MS 450 Broadway St, MC 5334 Redwood City, CA 94603 [email protected] Study Coordinator Ann Moffat 450 Broadway St, MC 5334 Redwood City, CA 94603 [email protected] 2 Table of Contents 1 Background ................................................................. 7 1.1 Disease Background ..................................................... 7 1.2 Hedgehog Pathway and mechanism of action ............................... 7 1.3 PI3K Pathway and mechanism of action ................................... 9 1.4 Sonidegib Compound Information ............ Error! Bookmark not defined. 1.4.1 Preclinical Studies for Sonidegib ....................................................................11 1.4.2 Muscular system...............................................................................................13 1.4.3 Skeletal system ................................................................................................13 -

Table of Contents
ANTICANCER RESEARCH International Journal of Cancer Research and Treatment ISSN: 0250-7005 Volume 32, Number 4, April 2012 Contents Experimental Studies * Review: Multiple Associations Between a Broad Spectrum of Autoimmune Diseases, Chronic Inflammatory Diseases and Cancer. A.L. FRANKS, J.E. SLANSKY (Aurora, CO, USA)............................................ 1119 Varicella Zoster Virus Infection of Malignant Glioma Cell Cultures: A New Candidate for Oncolytic Virotherapy? H. LESKE, R. HAASE, F. RESTLE, C. SCHICHOR, V. ALBRECHT, M.G. VIZOSO PINTO, J.C. TONN, A. BAIKER, N. THON (Munich; Oberschleissheim, Germany; Zurich, Switzerland) .................................... 1137 Correlation between Adenovirus-neutralizing Antibody Titer and Adenovirus Vector-mediated Transduction Efficiency Following Intratumoral Injection. K. TOMITA, F. SAKURAI, M. TACHIBANA, H. MIZUGUCHI (Osaka, Japan) .......................................................................................................... 1145 Reduction of Tumorigenicity by Placental Extracts. A.M. MARLEAU, G. MCDONALD, J. KOROPATNICK, C.-S. CHEN, D. KOOS (Huntington Beach; Santa Barbara; Loma Linda; San Diego, CA, USA; London, ON, Canada) ...................................................................................................................................... 1153 Stem Cell Markers as Predictors of Oral Cancer Invasion. A. SIU, C. LEE, D. DANG, C. LEE, D.M. RAMOS (San Francisco, CA, USA) ................................................................................................ -

Effect of Bioactive Compound of Aronia Melanocarpa on Cardiovascular System in Experimental Hypertension
Hindawi Oxidative Medicine and Cellular Longevity Volume 2017, Article ID 8156594, 8 pages https://doi.org/10.1155/2017/8156594 Research Article Effect of Bioactive Compound of Aronia melanocarpa on Cardiovascular System in Experimental Hypertension 1 1 1,2 1 Martina Cebova, Jana Klimentova, Pavol Janega, and Olga Pechanova 1Institute of Normal and Pathological Physiology, Slovak Academy of Sciences, Bratislava, Slovakia 2Department of Pathology, Faculty of Medicine, Comenius University, Bratislava, Slovakia Correspondence should be addressed to Martina Cebova; [email protected] Received 4 August 2017; Revised 10 October 2017; Accepted 18 October 2017; Published 30 November 2017 Academic Editor: Victor M. Victor Copyright © 2017 Martina Cebova et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Aronia melanocarpa has attracted scientific interest due to its dense contents of different polyphenols. We aimed to analyse effects of Aronia melanocarpa (AME) extract on blood pressure (BP), lipid peroxidation, cytokine level, total NOS activity in the left ventricle (LV), and aorta of L-NAME-induced hypertensive rats. 12-week-old male WKY rats were assigned to the control group and groups treated with AME extract (57.90 mg/kg/day), L-NAME (40 mg/kg/day), or combination of L-NAME (40 mg/kg/day) and AME (57.90 mg/kg/day) in tap water for 3 weeks. NOS activity, eNOS protein expression, and conjugated diene (CD) concentration were determined in the LV and aorta. After 3 weeks of L-NAME treatment, BP was increased by 28% and concomitant treatment with AME reduced it by 21%. -

Therapeutic Class Overview Colony Stimulating Factors
Therapeutic Class Overview Colony Stimulating Factors Therapeutic Class Overview/Summary: This review will focus on the granulocyte colony stimulating factors (G-CSFs) and granulocyte- macrophage colony stimulating factors (GM-CSFs).1-5 Colony-stimulating factors (CSFs) fall under the naturally occurring glycoprotein cytokines, one of the main groups of immunomodulators.6 In general, these proteins are vital to the proliferation and differentiation of hematopoietic progenitor cells.6-8 The G- CSFs commercially available in the United States include pegfilgrastim (Neulasta®), filgrastim (Neupogen®), filgrastim-sndz (Zarxio®), and tbo-filgrastim (Granix®). While filgrastim-sndz and tbo- filgrastim are the same recombinant human G-CSF as filgrastim, only filgrastim-sndz is considered a biosimilar drug as it was approved through the biosimilar pathway. At the time tbo-filgrastim was approved, a regulatory pathway for biosimilar drugs had not yet been established in the United States and tbo-filgrastim was filed under its own Biologic License Application.9 Only one GM-CSF is currently available, sargramostim (Leukine). These agents are Food and Drug Administration (FDA)-approved for a variety of conditions relating to neutropenia or for the collection of hematopoietic progenitor cells for collection by leukapheresis.1-5 Due to the pathway taken, tbo-filgrastim does not share all of the same indications as filgrastim and these two products are not interchangeable. It is important to note that although filgrastim-sndz is a biosimilar product, and it was approved with all the same indications as filgrastim at the time, filgrastim has since received FDA-approval for an additional indication that filgrastim-sndz does not have, to increase survival in patients with acute exposure to myelosuppressive doses of radiation.1-3A complete list of indications for each agent can be found in Table 1. -

Assessment of Overall Survival, Quality of Life, And
Supplementary Online Content Salas-Vega S, Iliopoulos O, Mossialos E. Assesment of overall survival, quality of life, and safety benefits associated with new cancer medicines [published online December 29, 2016]. JAMA Oncol. doi:10.1001/jamaoncol.2016.4166 eTable 1. Therapeutic profile of all cancer medicines approved by the FDA between 2003- 2013 eTable 2. Regulatory evidence in support of classification of drug clinical benefits eFigure 1. Number of cancer drugs that were evaluated by all three HTA agencies, sorted by magnitude of clinical benefits eTable 3. Interagency agreement–Krippendorff’s alpha coefficients eMethods This supplementary material has been provided by the authors to give readers additional information about their work. Online-Only Supplement © 2016 American Medical Association. All rights reserved. Downloaded From: https://jamanetwork.com/ on 09/25/2021 Clinical value of cancer medicines Contents eExhibits ......................................................................................................................................................... 3 eTable 1. Therapeutic profile of all cancer medicines approved by the FDA between 2003- 2013 (Summary of eTable 2) ................................................................................................................... 3 eTable 2. Regulatory evidence in support of classification of drug clinical benefits ....................... 6 eFigure 1. Number of cancer drugs that were evaluated by all three HTA agencies, sorted by magnitude of clinical benefits -

Palifermin (Kepivance™) in the Treatment of Mucositis
Session V • From cell biology to cell therapy Palifermin (Kepivance™) in the treatment of mucositis [haematologica reports] 2005;1(8):41-45 EMMANOUILIDES C n patients undergoing high dose chemo- factor (KGF) is a 28 kD member of the therapy and hematopoietic stem cell fibroblast growth factor family with epithe- Associate Profesor transplant, oral mucositis (OM) is one of lial cell proliferative properties.26 Palifermin of Medicine, UCLA, I Division Haematology-Oncology, the most debilitating and annoying side (Kepivance™) is a truncated, recombinant Thessaloniki, Greece effects. This complication results from form of human keratinocyte growth factor cytotoxic injury to the epithelial lining of (rHuKGF) that has been approved in the the oropharyngeal mucosa, although USA in 2004 to decrease the incidence and lesions of the whole gastrointestinal tract duration of severe OM in patients with also occur.1 The severity of OM varies from hematologic malignancies receiving myelo- erythema and edema accompanied by mild toxic therapy requiring HSCT support. Pal- soreness to full mucosal thickness ulcera- ifermin (recombinant human keratinocyte tions penetrating into the submucosa, growth factor) is an N-terminal, truncated often resulting in severe pain requiring version of endogenous keratinocyte growth narcotic analgesia and impaired swallow- factor with biologic activity similar to that ing, prolonged hospitalization, and of the native protein, but with increased increased risks for infections and poten- stability 26 In animal models of chemother- tially life-threatening sequelae.2,3 Between apy, radiotherapy, and hematopoietic stem- 40% to 80% of cancer patients undergo- cell transplantation27,28 palifermin protected ing intensive treatment regimens requiring several types of epithelial tissues. -
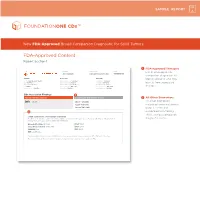
FDA-Approved Content Report Section 1
SAMPLE REPORT New FDA-Approved Broad Companion Diagnostic for Solid Tumors FDA-Approved Content Report Section 1 1 FDA-Approved Therapies PATIENT TUMOR TYPE TRF# List of FDA-approved Jane Sample Lung adenocarcinoma TRFXXXXXX companion diagnostics to PATIENT PHYSICIAN SPECIMEN identify patients who may DISEASE Lung adenocarcinoma ORDERING PHYSICIAN Not Given SPECIMEN SITE Not Given NAME Not Given MEDICAL FACILITY Not Given SPECIMEN ID Not Given benefi t from associated DATE OF BIRTH Not Given ADDITIONAL RECIPIENT Not Given SPECIMEN TYPE Not Given SEX Female MEDICAL FACILITY ID Not Given DATE OF COLLECTION Not Given therapies MEDICAL RECORD # Not Given PATHOLOGIST Not Given SPECIMEN RECEIVED Not Given CDx Associated Findings 1 GENOMIC FINDINGS DETECTED FDA-APPROVED THERAPEUTIC OPTIONS 2 All Other Biomarkers EGFR L858R Gilotrif® (Afatinib) All other biomarkers, Iressa® (Gefitinib) including tumor mutational Tarceva® (Erlotinib) burden (TMB) and 2 microsatellite instability (MSI), without companion OTHER ALTERATIONS & BIOMARKERS IDENTIFIED Results reported in this section are not prescriptive or conclusive for labeled use of any specific therapeutic product. See diagnostic claims professional services section for additional information. Microsatellite Status MS-Stable PTCH1 T416S Tumor Mutation Burden 11 Muts/Mb RBM10 Q494* CDKN2A/B loss TP53 R267P EGFR amplification § Refer to appendix for limitation statements related to detection of any copy number alterations, gene rearrangements, MSI or TMB result in this section. Please refer to appendix -

Encapsulation of Nedaplatin in Novel Pegylated Liposomes Increases Its Cytotoxicity and Genotoxicity Against A549 and U2OS Human Cancer Cells
pharmaceutics Article Encapsulation of Nedaplatin in Novel PEGylated Liposomes Increases Its Cytotoxicity and Genotoxicity against A549 and U2OS Human Cancer Cells 1, 2, 1 1 2, Salma El-Shafie y, Sherif Ashraf Fahmy y , Laila Ziko , Nada Elzahed , Tamer Shoeib * and Andreas Kakarougkas 1,* 1 Department of Biology, School of Sciences and Engineering, The American University in Cairo, Cairo 11835, Egypt; [email protected] (S.E.-S.); [email protected] (L.Z.); [email protected] (N.E.) 2 Department of Chemistry, School of Sciences and Engineering, The American University in Cairo, Cairo 11835 Egypt; sheriff[email protected] * Correspondence: [email protected] (T.S.); [email protected] (A.K.) These authors contribute equally to this paper. y Received: 7 April 2020; Accepted: 25 August 2020; Published: 10 September 2020 Abstract: Following the discovery of cisplatin over 50 years ago, platinum-based drugs have been a widely used and effective form of cancer therapy, primarily causing cell death by inducing DNA damage and triggering apoptosis. However, the dose-limiting toxicity of these drugs has led to the development of second and third generation platinum-based drugs that maintain the cytotoxicity of cisplatin but have a more acceptable side-effect profile. In addition to the creation of new analogs, tumor delivery systems such as liposome encapsulated platinum drugs have been developed and are currently in clinical trials. In this study, we have created the first PEGylated liposomal form of nedaplatin using thin film hydration. Nedaplatin, the main focus of this study, has been exclusively used in Japan for the treatment of non-small cell lung cancer, head and neck, esophageal, bladder, ovarian and cervical cancer. -

Comparison Between Filgrastim and Lenograstim Plus Chemotherapy For
Bone Marrow Transplantation (2010) 45, 277–281 & 2010 Macmillan Publishers Limited All rights reserved 0268-3369/10 $32.00 www.nature.com/bmt ORIGINAL ARTICLE Comparison between filgrastim and lenograstim plus chemotherapy for mobilization of PBPCs R Ria, T Gasparre, G Mangialardi, A Bruno, G Iodice, A Vacca and F Dammacco Department of Biomedical Sciences and Human Oncology, Section of Internal Medicine and Clinical Oncology, University of Bari Medical School, Bari, Italy Recombinant human (rHu) G-CSF has been widely used during transplantation.4 However, the use of G-CSF after to treat neutropenia and mobilize PBPCs for their autologous PBPC transplantation has been queried, as its autologous and allogeneic transplantation. It shortens further reduction in time to a safe neutrophil count5,6 does neutropenia and thus reduces the frequency of neutropenic not always imply fewer significant clinical events, such as fever. We compared the efficiency of glycosylated rHu infections, length of hospitalization, extrahematological and non-glycosylated Hu G-CSF in mobilizing hemato- toxicities or mortality.7,8 Even so, the ASCO guidelines still poietic progenitor cells (HPCs). In total, 86 patients were recommend the use of growth factors after autologous consecutively enrolled for mobilization with CY plus either PBPC transplantation.9 glycosylated or non-glycosylated G-CSF, and under- G-CSF induces the proliferation and differentiation of went leukapheresis. The HPC content of each collection, myeloid precursor cells, and also provides a functional toxicity, days of leukapheresis needed to reach the activity that influences chemotaxis, respiratory burst and minimum HPC target and days to recover WBC (X500 Ag expression of neutrophils. -

WO 2017/176265 Al
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date W O 2017/176265 A l 12 October 2017 (12.10.2017) P O P C T (51) International Patent Classification: (74) Agent: COLLINS, Daniel W.; Foley & Lardner LLP, A61K 9/00 (2006.01) A61K 9/51 (2006.01) 3000 K Street, NW, 6th Floor, Washington, DC 20007- A61K 47/42 (20 ) 5109 (US). (21) International Application Number: (81) Designated States (unless otherwise indicated, for every PCT/US20 16/026270 kind of national protection available): AE, AG, AL, AM, AO, AT, AU, AZ, BA, BB, BG, BH, BN, BR, BW, BY, (22) International Filing Date: BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, 6 April 2016 (06.04.2016) DO, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, (25) Filing Language: English HN, HR, HU, ID, IL, EST, IR, IS, JP, KE, KG, KN, KP, KR, KZ, LA, LC, LK, LR, LS, LU, LY, MA, MD, ME, MG, (26) Publication Language: English MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, OM, (71) Applicant: MAYO FOUNDATION FOR MEDICAL PA, PE, PG, PH, PL, PT, QA, RO, RS, RU, RW, SA, SC, EDUCATION AND RESEARCH [US/US]; 200 First SD, SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, TN, Street, NW, Rochester, Minnesota 55905 (US). TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, ZW. (84) Designated States (72) Inventors: MARKOVIC, Svetomir N.; c/o Mayo Founda (unless otherwise indicated, for every tion For Medical Education And Research, 200 First kind of regional protection available): ARIPO (BW, GH, Street, NW, Rochester, Minnesota 55905 (US). -

G-CSF Protects Motoneurons Against Axotomy-Induced Apoptotic Death In
Henriques et al. BMC Neuroscience 2010, 11:25 http://www.biomedcentral.com/1471-2202/11/25 RESEARCH ARTICLE Open Access G-CSF protects motoneurons against axotomy- induced apoptotic death in neonatal mice Alexandre Henriques1,2,3, Claudia Pitzer1*, Luc Dupuis2,3, Armin Schneider1* Abstract Background: Granulocyte colony stimulating factor (G-CSF) is a growth factor essential for generation of neutrophilic granulocytes. Apart from this hematopoietic function, we have recently uncovered potent neuroprotective and regenerative properties of G-CSF in the central nervous system (CNS). The G-CSF receptor and G-CSF itself are expressed in a motoneurons, G-CSF protects motoneurons, and improves outcome in the SOD1(G93A) transgenic mouse model for amyotrophic lateral sclerosis (ALS). In vitro, G-CSF acts anti- apoptotically on motoneuronal cells. Due to the pleiotrophic effects of G-CSF and the complexity of the SOD1 transgenic ALS models it was however not possible to clearly distinguish between directly mediated anti- apoptotic and indirectly protective effects on motoneurons. Here we studied whether G-CSF is able to protect motoneurons from purely apoptotic cell death induced by a monocausal paradigm, neonatal sciatic nerve axotomy. Results: We performed sciatic nerve axotomy in neonatal mice overexpressing G-CSF in the CNS and found that G-CSF transgenic mice displayed significantly higher numbers of surviving lumbar motoneurons 4 days following axotomy than their littermate controls. Also, surviving motoneurons in G-CSF overexpressing animals were larger, suggesting additional trophic effects of this growth factor. Conclusions: In this model of pure apoptotic cell death the protective effects of G-CSF indicate direct actions of G-CSF on motoneurons in vivo. -

September 2017 ~ Resource #330909
−This Clinical Resource gives subscribers additional insight related to the Recommendations published in− September 2017 ~ Resource #330909 Medications Stored in the Refrigerator (Information below comes from current U.S. and Canadian product labeling and is current as of date of publication) Proper medication storage is important to ensure medication shelf life until the manufacturer expiration date and to reduce waste. Many meds are recommended to be stored at controlled-room temperature. However, several meds require storage in the refrigerator or freezer to ensure stability. See our toolbox, Medication Storage: Maintaining the Cold Chain, for helpful storage tips and other resources. Though most meds requiring storage at temperatures colder than room temperature should be stored in the refrigerator, expect to see a few meds require storage in the freezer. Some examples of medications requiring frozen storage conditions include: anthrax immune globulin (Anthrasil [U.S. only]), carmustine wafer (Gliadel [U.S. only]), cholera (live) vaccine (Vaxchora), dinoprostone vaginal insert (Cervidil), dinoprostone vaginal suppository (Prostin E2 [U.S.]), varicella vaccine (Varivax [U.S.]; Varivax III [Canada] can be stored in the refrigerator or freezer), zoster vaccine (Zostavax [U.S.]; Zostavax II [Canada] can be stored in the refrigerator or freezer). Use the list below to help identify medications requiring refrigerator storage and become familiar with acceptable temperature excursions from recommended storage conditions. Abbreviations: RT = room temperature Abaloparatide (Tymlos [U.S.]) Aflibercept (Eylea) Amphotericin B (Abelcet, Fungizone) • Once open, may store at RT (68°F to 77°F • May store at RT (77°F [25°C]) for up to Anakinra (Kineret) [20°C to 25°C]) for up to 30 days.