CRISPR/Cas-Mediated Base Editing
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Large-Scale Analysis of Genome and Transcriptome Alterations in Multiple Tumors Unveils Novel Cancer-Relevant Splicing Networks
Downloaded from genome.cshlp.org on September 28, 2021 - Published by Cold Spring Harbor Laboratory Press Research Large-scale analysis of genome and transcriptome alterations in multiple tumors unveils novel cancer-relevant splicing networks Endre Sebestyén,1,5 Babita Singh,1,5 Belén Miñana,1,2 Amadís Pagès,1 Francesca Mateo,3 Miguel Angel Pujana,3 Juan Valcárcel,1,2,4 and Eduardo Eyras1,4 1Universitat Pompeu Fabra, E08003 Barcelona, Spain; 2Centre for Genomic Regulation, E08003 Barcelona, Spain; 3Program Against Cancer Therapeutic Resistance (ProCURE), Catalan Institute of Oncology (ICO), Bellvitge Institute for Biomedical Research (IDIBELL), E08908 L’Hospitalet del Llobregat, Spain; 4Catalan Institution for Research and Advanced Studies, E08010 Barcelona, Spain Alternative splicing is regulated by multiple RNA-binding proteins and influences the expression of most eukaryotic genes. However, the role of this process in human disease, and particularly in cancer, is only starting to be unveiled. We system- atically analyzed mutation, copy number, and gene expression patterns of 1348 RNA-binding protein (RBP) genes in 11 solid tumor types, together with alternative splicing changes in these tumors and the enrichment of binding motifs in the alter- natively spliced sequences. Our comprehensive study reveals widespread alterations in the expression of RBP genes, as well as novel mutations and copy number variations in association with multiple alternative splicing changes in cancer drivers and oncogenic pathways. Remarkably, the altered splicing patterns in several tumor types recapitulate those of undifferen- tiated cells. These patterns are predicted to be mainly controlled by MBNL1 and involve multiple cancer drivers, including the mitotic gene NUMA1. We show that NUMA1 alternative splicing induces enhanced cell proliferation and centrosome am- plification in nontumorigenic mammary epithelial cells. -

Investigation of Key Genes and Pathways in Inhibition of Oxycodone on Vincristine-Induced Microglia Activation by Using Bioinformatics Analysis
Hindawi Disease Markers Volume 2019, Article ID 3521746, 10 pages https://doi.org/10.1155/2019/3521746 Research Article Investigation of Key Genes and Pathways in Inhibition of Oxycodone on Vincristine-Induced Microglia Activation by Using Bioinformatics Analysis Wei Liu,1 Jishi Ye,2 and Hong Yan 1 1Department of Anesthesiology, the Central Hospital of Wuhan, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430014, China 2Department of Anesthesiology, Renmin Hospital of Wuhan University, Wuhan, 430060 Hubei, China Correspondence should be addressed to Hong Yan; [email protected] Received 2 November 2018; Accepted 31 December 2018; Published 10 February 2019 Academic Editor: Hubertus Himmerich Copyright © 2019 Wei Liu et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Introduction. The neurobiological mechanisms underlying the chemotherapy-induced neuropathic pain are only partially understood. Among them, microglia activation was identified as the key component of neuropathic pain. The aim of this study was to identify differentially expressed genes (DEGs) and pathways associated with vincristine-induced neuropathic pain by using bioinformatics analysis and observe the effects of oxycodone on these DEG expressions in a vincristine-induced microglia activation model. Methods. Based on microarray profile GSE53897, we identified DEGs between vincristine-induced neuropathic pain rats and the control group. Using the ToppGene database, the prioritization DEGs were screened and performed by gene ontology (GO) and signaling pathway enrichment. A protein-protein interaction (PPI) network was used to explore the relationship among DEGs. -

The Rnase H-Like Superfamily: New Members, Comparative Structural Analysis and Evolutionary Classification Karolina A
4160–4179 Nucleic Acids Research, 2014, Vol. 42, No. 7 Published online 23 January 2014 doi:10.1093/nar/gkt1414 The RNase H-like superfamily: new members, comparative structural analysis and evolutionary classification Karolina A. Majorek1,2,3,y, Stanislaw Dunin-Horkawicz1,y, Kamil Steczkiewicz4, Anna Muszewska4,5, Marcin Nowotny6, Krzysztof Ginalski4 and Janusz M. Bujnicki1,3,* 1Laboratory of Bioinformatics and Protein Engineering, International Institute of Molecular and Cell Biology, ul. Ks. Trojdena 4, PL-02-109 Warsaw, Poland, 2Department of Molecular Physiology and Biological Physics, University of Virginia, 1340 Jefferson Park Avenue, Charlottesville, VA USA-22908, USA, 3Bioinformatics Laboratory, Institute of Molecular Biology and Biotechnology, Adam Mickiewicz University, Umultowska 89, PL-61-614 Poznan, Poland, 4Laboratory of Bioinformatics and Systems Biology, Centre of New Technologies, University of Warsaw, Zwirki i Wigury 93, PL-02-089 Warsaw, Poland, 5Institute of Biochemistry and Biophysics PAS, Pawinskiego 5A, PL-02-106 Warsaw, Poland and 6Laboratory of Protein Structure, International Institute of Molecular and Cell Biology, ul. Ks. Trojdena 4, PL-02-109 Warsaw, Poland Received September 23, 2013; Revised December 12, 2013; Accepted December 26, 2013 ABSTRACT revealed a correlation between the orientation of Ribonuclease H-like (RNHL) superfamily, also called the C-terminal helix with the exonuclease/endo- the retroviral integrase superfamily, groups together nuclease function and the architecture of the numerous enzymes involved in nucleic acid metab- active site. Our analysis provides a comprehensive olism and implicated in many biological processes, picture of sequence-structure-function relation- including replication, homologous recombination, ships in the RNHL superfamily that may guide func- DNA repair, transposition and RNA interference. -

Overlap of Quantitative Trait Loci for Early Growth Rate, and for Body
REPRODUCTIONRESEARCH Functional reconstruction of NANOS3 expression in the germ cell lineage by a novel transgenic reporter reveals distinct subcellular localizations of NANOS3 Masashi Yamaji1, Takashi Tanaka2, Mayo Shigeta1, Shinichiro Chuma2, Yumiko Saga3 and Mitinori Saitou1,4,5 1Laboratory for Mammalian Germ Cell Biology, RIKEN Center for Developmental Biology, 2-2-3 Minatojima- Minamimachi, Chuo-ku, Kobe 650-0047, Japan, 2Department of Development and Differentiation, Institute for Frontier Medical Sciences, Kyoto University, Shogoin, Sakyo-ku, Kyoto 606-8507, Japan, 3Department of Genetics and Division of Mammalian Development, National Institute of Genetics, SOKENDAI, 1111 Yata, Mishima, Shizuoka 411-8540, Japan, 4Department of Anatomy and Cell Biology, Graduate School of Medicine, Kyoto University, Yoshida- Konoe-cho, Sakyo-ku, Kyoto 606-8501, Japan and 5JST, CREST, Yoshida-Konoe-cho, Sakyo-ku, Kyoto 606-8501, Japan Correspondence should be addressed to M Saitou at Department of Anatomy and Cell Biology, Graduate School of Medicine, Kyoto University; Email: [email protected] Abstract Mutations of RNA-binding proteins such as NANOS3, TIAL1, and DND1 in mice have been known to result in the failure of survival and/or proliferation of primordial germ cells (PGCs) soon after their fate is specified (around embryonic day (E) 8.0), leading to the infertility of these animals. However, the mechanisms of actions of these RNA-binding proteins remain largely unresolved. As a foundation to explore the role of these RNA-binding proteins in germ cells, we established a novel transgenic reporter strain that expresses NANOS3 fused with EGFP under the control of Nanos3 regulatory elements. NANOS3–EGFP exhibited exclusive expression in PGCs as early as E7.25, and continued to be expressed in female germ cells until around E14.5 and in male germ cells throughout the fetal period with declining expression levels after E16.5. -

Identification and Functional Analysis of Novel Genes Associated with Inherited Bone Marrow Failure Syndromes
Identification and Functional Analysis of Novel Genes Associated with Inherited Bone Marrow Failure Syndromes by Anna Matveev A thesis submitted in conformity with the requirements for the degree of Master of Science Institute of Medical Science University of Toronto © Copyright by Anna Matveev 2020 Abstract Identification and Functional Analysis of Novel Genes Associated with Inherited Bone Marrow Failure Syndromes Anna Matveev Master of Science Institute of Medical Science University of Toronto 2020 Inherited bone marrow failure syndromes are multisystem-disorders that affect development of hematopoietic system. One of IBMFSs is Shwachman-Diamond-syndrome and about 80-90% of patients have mutations in the Shwachman-Bodian-Diamond-Syndrome gene. To unravel the genetic cause of the disease in the remaining 10-20% of patients, we performed WES as well as SNP-genotyping in families with SDS-phenotype and no mutations in SBDS. The results showed a region of homozygosity in chromosome 5p-arm DNAJC21 is in this region. Western blotting revealed reduced/null protein in patient. DNAJC21-homolog in yeast has been shown facilitating the release of the Arx1/Alb1 heterodimer from pre-60S.To investigate the cellular functions of DNAJC21 we knocked-down it in HEK293T-cells. We observed a high-level of ROS, which led to reduced cell proliferation. Our data indicate that mutations in DNAJC21 contribute to SDS. We hypothesize that DNAJC21 related ribosomal defects lead to increased levels of ROS therefore altering development and maturation of hematopoietic cells. ii Acknowledgments I would like to take this opportunity to extend my deepest gratitude to everyone who has helped me throughout my degree. -

Bioinformatics Analyses of Genomic Imprinting
Bioinformatics Analyses of Genomic Imprinting Dissertation zur Erlangung des Grades des Doktors der Naturwissenschaften der Naturwissenschaftlich-Technischen Fakultät III Chemie, Pharmazie, Bio- und Werkstoffwissenschaften der Universität des Saarlandes von Barbara Hutter Saarbrücken 2009 Tag des Kolloquiums: 08.12.2009 Dekan: Prof. Dr.-Ing. Stefan Diebels Berichterstatter: Prof. Dr. Volkhard Helms Priv.-Doz. Dr. Martina Paulsen Vorsitz: Prof. Dr. Jörn Walter Akad. Mitarbeiter: Dr. Tihamér Geyer Table of contents Summary________________________________________________________________ I Zusammenfassung ________________________________________________________ I Acknowledgements _______________________________________________________II Abbreviations ___________________________________________________________ III Chapter 1 – Introduction __________________________________________________ 1 1.1 Important terms and concepts related to genomic imprinting __________________________ 2 1.2 CpG islands as regulatory elements ______________________________________________ 3 1.3 Differentially methylated regions and imprinting clusters_____________________________ 6 1.4 Reading the imprint __________________________________________________________ 8 1.5 Chromatin marks at imprinted regions___________________________________________ 10 1.6 Roles of repetitive elements ___________________________________________________ 12 1.7 Functional implications of imprinted genes _______________________________________ 14 1.8 Evolution and parental conflict ________________________________________________ -

Congenital Disorders of Glycosylation from a Neurological Perspective
brain sciences Review Congenital Disorders of Glycosylation from a Neurological Perspective Justyna Paprocka 1,* , Aleksandra Jezela-Stanek 2 , Anna Tylki-Szyma´nska 3 and Stephanie Grunewald 4 1 Department of Pediatric Neurology, Faculty of Medical Science in Katowice, Medical University of Silesia, 40-752 Katowice, Poland 2 Department of Genetics and Clinical Immunology, National Institute of Tuberculosis and Lung Diseases, 01-138 Warsaw, Poland; [email protected] 3 Department of Pediatrics, Nutrition and Metabolic Diseases, The Children’s Memorial Health Institute, W 04-730 Warsaw, Poland; [email protected] 4 NIHR Biomedical Research Center (BRC), Metabolic Unit, Great Ormond Street Hospital and Institute of Child Health, University College London, London SE1 9RT, UK; [email protected] * Correspondence: [email protected]; Tel.: +48-606-415-888 Abstract: Most plasma proteins, cell membrane proteins and other proteins are glycoproteins with sugar chains attached to the polypeptide-glycans. Glycosylation is the main element of the post- translational transformation of most human proteins. Since glycosylation processes are necessary for many different biological processes, patients present a diverse spectrum of phenotypes and severity of symptoms. The most frequently observed neurological symptoms in congenital disorders of glycosylation (CDG) are: epilepsy, intellectual disability, myopathies, neuropathies and stroke-like episodes. Epilepsy is seen in many CDG subtypes and particularly present in the case of mutations -

A Comprehensive Analysis of 3' End Sequencing Data Sets Reveals Novel
bioRxiv preprint doi: https://doi.org/10.1101/033001; this version posted November 26, 2015. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. A comprehensive analysis of 3’ end sequencing data sets reveals novel polyadenylation signals and the repressive role of heterogenous ribonucleoprotein C on cleavage and polyadenylation 1 1 1 1 1 Andreas J. Gruber , Ralf Schmidt , Andreas R. Gruber , Georges Martin , Souvik Ghosh , 1,2 1 1,§ Manuel Belmadani , Walter Keller , Mihaela Zavolan 1 Computational and Systems Biology, Biozentrum, University of Basel, Klingelbergstrasse 5070, 4056 Basel, Switzerland 2 Current address: University of British Columbia, 177 Michael Smith Laboratories, 2185 East Mall Vancouver BC V6T 1Z4 § To whom correspondence should be addressed ([email protected]) Abstract Alternative polyadenylation (APA) is a general mechanism of transcript diversification in mammals, which has been recently linked to proliferative states and cancer. Different 3’ untranslated region (3’ UTR) isoforms interact with different RNA binding proteins (RBPs), which modify the stability, translation, and subcellular localization of the corresponding transcripts. Although the heterogeneity of premRNA 3’ end processing has been established with highthroughput approaches, the mechanisms that underlie systematic changes in 3’ UTR lengths remain to be characterized. Through a uniform analysis of a large number of 3’ end sequencing data sets we have uncovered 18 signals, 6 of which novel, whose positioning with respect to premRNA cleavage sites indicates a role in premRNA 3’ end processing in both mouse and human. -

Genetic Variant in 3' Untranslated Region of the Mouse Pycard Gene
bioRxiv preprint doi: https://doi.org/10.1101/2021.03.26.437184; this version posted March 26, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. 1 2 3 Title: 4 Genetic Variant in 3’ Untranslated Region of the Mouse Pycard Gene Regulates Inflammasome 5 Activity 6 Running Title: 7 3’UTR SNP in Pycard regulates inflammasome activity 8 Authors: 9 Brian Ritchey1*, Qimin Hai1*, Juying Han1, John Barnard2, Jonathan D. Smith1,3 10 1Department of Cardiovascular & Metabolic Sciences, Lerner Research Institute, Cleveland Clinic, 11 Cleveland, OH 44195 12 2Department of Quantitative Health Sciences, Lerner Research Institute, Cleveland Clinic, Cleveland, OH 13 44195 14 3Department of Molecular Medicine, Cleveland Clinic Lerner College of Medicine of Case Western 15 Reserve University, Cleveland, OH 44195 16 *, These authors contributed equally to this study. 17 Address correspondence to Jonathan D. Smith: email [email protected]; ORCID ID 0000-0002-0415-386X; 18 mailing address: Cleveland Clinic, Box NC-10, 9500 Euclid Avenue, Cleveland, OH 44195, USA. 19 1 bioRxiv preprint doi: https://doi.org/10.1101/2021.03.26.437184; this version posted March 26, 2021. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY 4.0 International license. 20 Abstract 21 Quantitative trait locus mapping for interleukin-1 release after inflammasome priming and activation 22 was performed on bone marrow-derived macrophages (BMDM) from an AKRxDBA/2 strain intercross. -
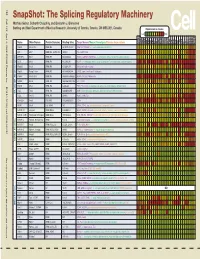
Snapshot: the Splicing Regulatory Machinery Mathieu Gabut, Sidharth Chaudhry, and Benjamin J
192 Cell SnapShot: The Splicing Regulatory Machinery Mathieu Gabut, Sidharth Chaudhry, and Benjamin J. Blencowe 133 Banting and Best Department of Medical Research, University of Toronto, Toronto, ON M5S 3E1, Canada Expression in mouse , April4, 2008©2008Elsevier Inc. Low High Name Other Names Protein Domains Binding Sites Target Genes/Mouse Phenotypes/Disease Associations Amy Ceb Hip Hyp OB Eye SC BM Bo Ht SM Epd Kd Liv Lu Pan Pla Pro Sto Spl Thy Thd Te Ut Ov E6.5 E8.5 E10.5 SRp20 Sfrs3, X16 RRM, RS GCUCCUCUUC SRp20, CT/CGRP; −/− early embryonic lethal E3.5 9G8 Sfrs7 RRM, RS, C2HC Znf (GAC)n Tau, GnRH, 9G8 ASF/SF2 Sfrs1 RRM, RS RGAAGAAC HipK3, CaMKIIδ, HIV RNAs; −/− embryonic lethal, cond. KO cardiomyopathy SC35 Sfrs2 RRM, RS UGCUGUU AChE; −/− embryonic lethal, cond. KO deficient T-cell maturation, cardiomyopathy; LS SRp30c Sfrs9 RRM, RS CUGGAUU Glucocorticoid receptor SRp38 Fusip1, Nssr RRM, RS ACAAAGACAA CREB, type II and type XI collagens SRp40 Sfrs5, HRS RRM, RS AGGAGAAGGGA HipK3, PKCβ-II, Fibronectin SRp55 Sfrs6 RRM, RS GGCAGCACCUG cTnT, CD44 DOI 10.1016/j.cell.2008.03.010 SRp75 Sfrs4 RRM, RS GAAGGA FN1, E1A, CD45; overexpression enhances chondrogenic differentiation Tra2α Tra2a RRM, RS GAAARGARR GnRH; overexpression promotes RA-induced neural differentiation SR and SR-Related Proteins Tra2β Sfrs10 RRM, RS (GAA)n HipK3, SMN, Tau SRm160 Srrm1 RS, PWI AUGAAGAGGA CD44 SWAP Sfrs8 RS, SWAP ND SWAP, CD45, Tau; possible asthma susceptibility gene hnRNP A1 Hnrnpa1 RRM, RGG UAGGGA/U HipK3, SMN2, c-H-ras; rheumatoid arthritis, systemic lupus -

Gigantea: Uncovering New Functions in Flower Development
G C A T T A C G G C A T genes Review Gigantea: Uncovering New Functions in Flower Development Claudio Brandoli 1, Cesar Petri 2 , Marcos Egea-Cortines 1 and Julia Weiss 1,* 1 Genética Molecular, Instituto de Biotecnología Vegetal, Edificio I+D+I, Plaza del Hospital s/n, Universidad Politécnica de Cartagena, 30202 Cartagena, Spain; [email protected] (C.B.); [email protected] (M.E.-C.) 2 Instituto de Hortofruticultura Subtropical y Mediterránea-UMA-CSIC, Departamento de Fruticultura Subtropical y Mediterránea, 29750 Algarrobo-costa, Málaga, Spain; [email protected] * Correspondence: [email protected]; Tel.: +34-868-071-078 Received: 27 August 2020; Accepted: 22 September 2020; Published: 28 September 2020 Abstract: GIGANTEA (GI) is a gene involved in multiple biological functions, which have been analysed and are partially conserved in a series of mono- and dicotyledonous plant species. The identified biological functions include control over the circadian rhythm, light signalling, cold tolerance, hormone signalling and photoperiodic flowering. The latter function is a central role of GI, as it involves a multitude of pathways, both dependent and independent of the gene CONSTANS(CO), as well as on the basis of interaction with miRNA. The complexity of the gene function of GI increases due to the existence of paralogs showing changes in genome structure as well as incidences of sub- and neofunctionalization. We present an updated report of the biological function of GI, integrating late insights into its role in floral initiation, flower development and volatile flower production. Keywords: Gene ontology; molecular function; cellular localisation; biological function; circadian clock; flowering time; flower development; floral scent 1. -

Current Trends in Gene Recovery Mediated by the CRISPR-Cas System Hyeon-Ki Jang 1, Beomjong Song2,Gue-Hohwang1 and Sangsu Bae 1
Jang et al. Experimental & Molecular Medicine (2020) 52:1016–1027 https://doi.org/10.1038/s12276-020-0466-1 Experimental & Molecular Medicine REVIEW ARTICLE Open Access Current trends in gene recovery mediated by the CRISPR-Cas system Hyeon-Ki Jang 1, Beomjong Song2,Gue-HoHwang1 and Sangsu Bae 1 Abstract The CRISPR-Cas system has undoubtedly revolutionized the genome editing field, enabling targeted gene disruption, regulation, and recovery in a guide RNA-specific manner. In this review, we focus on currently available gene recovery strategies that use CRISPR nucleases, particularly for the treatment of genetic disorders. Through the action of DNA repair mechanisms, CRISPR-mediated DNA cleavage at a genomic target can shift the reading frame to correct abnormal frameshifts, whereas DNA cleavage at two sites, which can induce large deletions or inversions, can correct structural abnormalities in DNA. Homology-mediated or homology-independent gene recovery strategies that require donor DNAs have been developed and widely applied to precisely correct mutated sequences in genes of interest. In contrast to the DNA cleavage-mediated gene correction methods listed above, base-editing tools enable base conversion in the absence of donor DNAs. In addition, CRISPR-associated transposases have been harnessed to generate a targeted knockin, and prime editors have been developed to edit tens of nucleotides in cells. Here, we introduce currently developed gene recovery strategies and discuss the pros and cons of each. 1234567890():,; 1234567890():,; 1234567890():,; 1234567890():,; Introduction differs from that of the endogenous gene2. Furthermore, Human genetic disorders, often associated with severe the mutated endogenous gene, which is malfunctional and pathological phenotypes, are caused by genomic aberra- potentially cytotoxic, might still be transcribed.