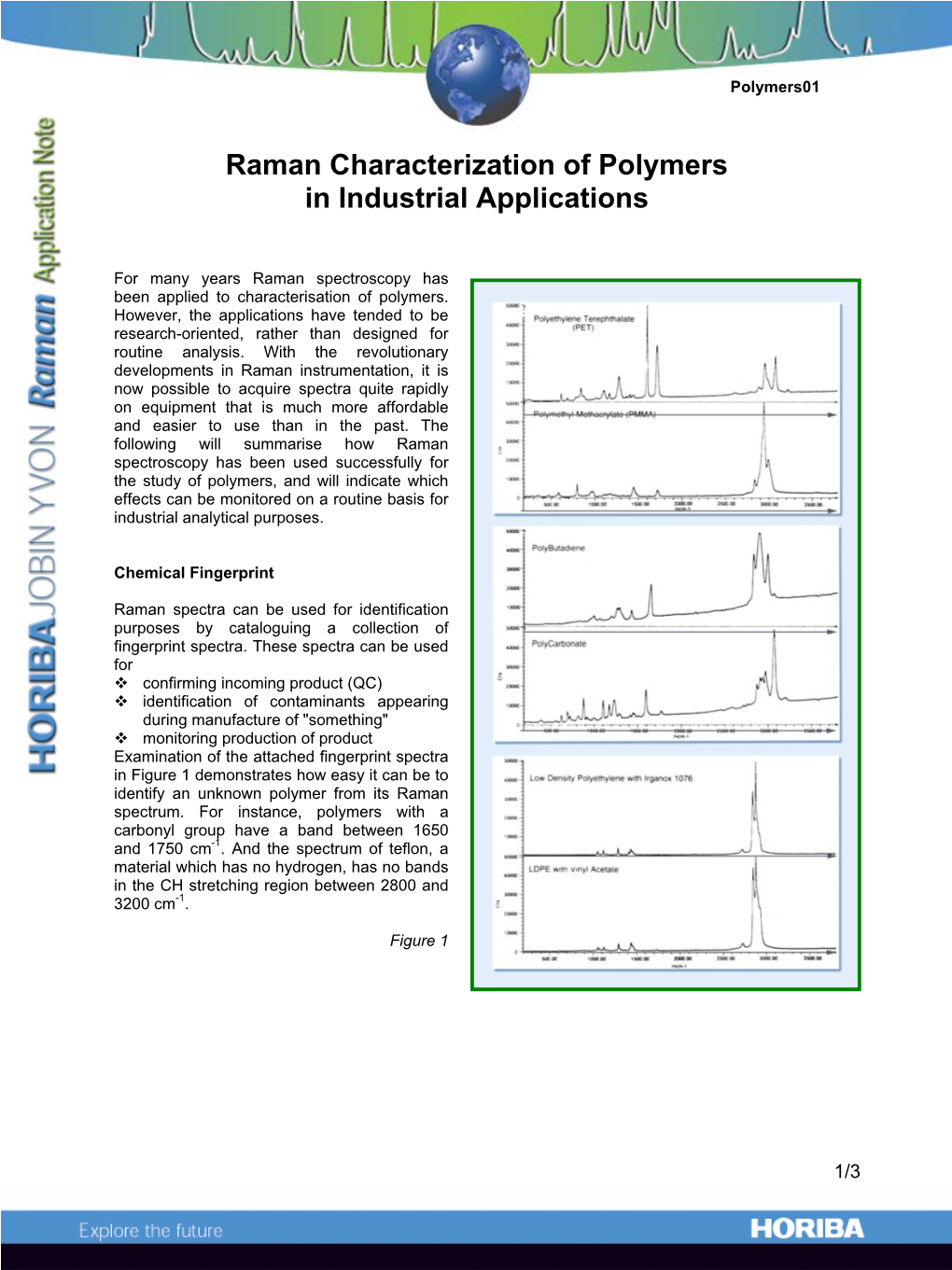
Raman Characterization of Polymers in Industrial Applications
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Three Methods for in Situ Cross-Linking of Polyvinyl Alcohol Films for Application As Ion-Conducting Membranes in Potassium Hydroxide Electrolyte
NASA TP 1407 c.1 t . ... J 1 NASA Technical Paper 1407 Three Methods for In Situ Cross-Linking of Polyvinyl Alcohol Films for Application as Ion-Conducting Membranes in Potassium Hydroxide Electrolyte Warren H. Philipp and Li-Chen Hsu I APRIL 1979 b. NASA a TECH LIBRARY KAFB, NM 0134803 NASA Technical Paper 1407 i Three Methods for In Situ Cross-Linking of Polyvinyl Alcohol Films for Application as Ion-Conducting Membranes in Potassium Hydroxide Electrolyte Warren H. Philipp and Li-Chen Hsu Lewis Research Center Clevelaizd, Ohio A NASA National Aeronautics and Space Administration Scientific and Technical Information Office 1979 Ir SUMMARY Three methods for in situ cross-linking of water soluble polyvinyl alcohol films are presented. These cross-linked films show promise for use as battery separators in T aqueous potassium hydroxide (KOH) electrolyte. Electrical resistivities in KOH of cross-linked membranes representing the three procedures are given with a brief dis- ; cussion of the chemical mechanism involved in their preparation. Physical properties, such as mechanical strength and swelling in alkaline electrolyte, are discussed. The three cross-linking techniques entail: (l)Treating a polyvinyl alcohol membrane containing a specified amount of a dial- dehyde such as glutaraldehyde with an acid solution which catalyzes acetalization cross- linking. (2) Treating a polyvinyl alcohol film with periodic acid which cleaves the few 1,2 diol units present in polyvinyl alcohol with the formation of aldehyde groups which then I causes cross-linking via acetalation of the 1, 3 diol units. (3) Reacting a polyvinyl alcohol film with hydrogen atoms and hydroxyl radicals from irradiated water whereby cross-linking is accomplished by polymer radicals formed as a consequence of hydrogen abstraction. -

Poly(Acrylic Acid) Surface Grafted Polypropylene Films: Near Surface and Bulk Mechanical Response
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by CONICET Digital eXPRESS Polymer Letters Vol.2, No.11 (2008) 779–790 Available online at www.expresspolymlett.com DOI: 10.3144/expresspolymlett.2008.91 Poly(acrylic acid) surface grafted polypropylene films: Near surface and bulk mechanical response L. A. Fasce1, V. Costamagna2, V. Pettarin1, M. Strumia2, P. M. Frontini1* 1Universidad Nacional de Mar del Plata, Instituto de Investigaciones en Ciencia y Tecnología de Materiales, INTEMA, J.B. Justo 4302 - B7608 FDQ - Mar del Plata, Argentina 2Departamento de Química Orgánica, Facultad de Ciencias Químicas, Universidad Nacional de Córdoba, Argentina Received 19 August 2008; accepted in revised form 20 September 2008 Abstract. Radical photo-grafting polymerization constitutes a promising technique for introducing functional groups onto surfaces of polypropylene films. According to their final use, surface grafting should be done without affecting overall mechanical properties. In this work the tensile drawing, fracture and biaxial impact response of biaxially oriented polypropylene commercial films grafted with poly(acrylic acid) (PAA) were investigated in terms of film orientation and surface modification. The variations of surface roughness, elastic modulus, hardness and resistance to permanent deforma- tion induced by the chemical treatment were assessed by depth sensing indentation. As a consequence of chemical modifi- cation the optical, transport and wettability properties of the films were successfully varied. The introduced chains generated a PAA-grafted layer, which is stiffer and harder than the neat polypropylene surface. Regardless of the surface changes, it was proven that this kind of grafting procedure does not detriment bulk mechanical properties of the PP film. -

Reactive Melt Blending of Low-Density Polyethylene with Poly (Acrylic Acid)
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector Arabian Journal of Chemistry (2015) 8, 191–199 King Saud University Arabian Journal of Chemistry www.ksu.edu.sa www.sciencedirect.com ORIGINAL ARTICLE Reactive melt blending of low-density polyethylene with poly (acrylic acid) Tahseen A. Saki * Basrah University, College of Science, Chemistry Department, Basrah, Iraq Received 17 May 2011; accepted 19 May 2011 Available online 17 June 2011 KEYWORDS Abstract Reactive melt blending of low-density polyethylene LDPE with a low molecular weight Low-density polyethylene; poly (acrylic acid) PAA in the presence of benzoyl peroxide (BPO) free radical initiator was studied Poly(acrylic acid); by means of a HAAKE torque rheometer and a capillary rheometer FTIR spectroscopy and was Reactive blend; used to characterize the structure of LDPE/PAA blends. The rheological behavior of blends has FTIR; been investigated at different temperatures (170, 190, 210 and 230 °C) and shear rates (5.43, 18.1, Rheological behavior; 54.3, 181 and 543 SÀ1). With a rise of temperature and increasing shear rates the shear stress of Flow index; blends increases nonlinearly. The melt viscosity increases with the increase in the PAA proportion Activation energy but decreases with the increasing shear rate and shear stress. The melts of all the blends are pseudo- plastic in nature. The flow behavior index (n) of the blends decreases with the increase in the PAA proportion and with the increase in the temperature. The temperature sensitivity of the flow behav- ior of the blends is studied using Arrhenius plots. -

Synthesis, Characterization of a New Polyacrylic Acid Superabsorbent, Some Heavy Metal Ion Sorption, the Adsorption Isotherms, and Quantum Chemical Investigation
materials Article Synthesis, Characterization of a New Polyacrylic Acid Superabsorbent, Some Heavy Metal Ion Sorption, the Adsorption Isotherms, and Quantum Chemical Investigation Sevil Savaskan Yilmaz 1,* , Nuri Yildirim 1, Murat Misir 2, Yasin Misirlioglu 2 and Emre Celik 1 1 Department of Chemistry, Faculty of Sciences, Karadeniz Technical University, University Avanue, 61080 Trabzon, Turkey; [email protected] (N.Y.); [email protected] (E.C.) 2 Faculty of Engineering and Architecture, Ahi Evran University, 40100 Kır¸sehir, Turkey; [email protected] (M.M.); [email protected] (Y.M.) * Correspondence: [email protected]; Tel.: +90-462-377-2506; Fax: +90-462-325-3195 Received: 3 July 2020; Accepted: 13 August 2020; Published: 1 October 2020 Abstract: Poly(acrylic acid/Kryptofix 23-Dimethacrylate) superabsorbent polymer [P (AA/Kry23-DM) SAP] was synthesized by solution polymerization to remove Co, Ni, Cu, Cd, Mn, Zn, Pb, Cr, and Fe ions in water and improve the quality of the water. Kry23-DM cross-linker (1,4,7,13,16-Pentaoxa-10,19 diazo cyclohexene icosane di methacrylate) was synthesized using Kry23 and methacryloyl chloride. The characterization of the molecules was done by FTIR, TGA, DSC, and SEM techniques. The effects of parameters such as pH, concentration, and the metal ion interaction on the heavy metal ions uptaking of SAP was investigated. It was observed that P (AA/Kry23-DM) SAP has maximum water absorption, and the absorption increases with the pH increase. Adsorption rates and sorption capacity, desorption ratios, competitive sorption (qcs), and distribution coefficient (log D) of P(AA/Kry23-DM) SAP were studied as a function of time and pH with the heavy metal ion concentration. -

Surface Modification of Polyvinyl Chloride by Polyacrylic Acid Graft As a Polyelectrolyte Membrane Using Ar Plasma
Turkish Journal of Chemistry Turk J Chem (2019) 43: 1686 – 1696 http://journals.tubitak.gov.tr/chem/ © TÜBİTAK Research Article doi:10.3906/kim-1903-48 Surface modification of polyvinyl chloride by polyacrylic acid graft as a polyelectrolyte membrane using Ar plasma Alaa FAHMY1;∗,, Mouhamed ABU-SAIED2,, Nasser MORGAN3,, Walid QUTOP1,, Hassan ABDELBARY1,, Tarek SALAMA1, 1Department of Chemistry, Faculty of Science, Al-Azhar University, Cairo, Egypt 2Department of Polymeric Materials Research, Advanced Technology and New Materials Research Institute, City of Scientific Research & Technological Applications (SRTA-City), New Borg Al-Arab City, Alexandria, Egypt 3Department of Physics, Faculty of Science, Al-Azhar University, Cairo, Egypt Received: 16.03.2019 • Accepted/Published Online: 12.11.2019 • Final Version: 09.12.2019 Abstract: This work reports the synthesis and properties of a new membrane based on polyvinyl chloride (PVC) grafting with polyacrylic acid (PAA) using argon (Ar) plasma. The membranes of PVC were synthesized by solution- casting method, where PAA was deposited as an ultrathin film onto PVC using dielectric barrier discharge at atmospheric pressure with Ar gas. The surface characteristics and chemical composition of the modified membranes were analyzed by water contact angle, scanning electron microscopy, and Fourier transform infrared spectroscopy. Moreover, the electrochemical properties of the membrane were investigated via ion exchange capacity for the purpose of using it as a polyelectrolyte membrane. Key words: Atmospheric plasma, electrolyte membrane, grafting polymerization, polyacrylic acid, polyvinyl chloride 1. Introduction Fuel cells have advantages that have made them the first source of portable energy used in vehicles and simple buildings as an alternative to rechargeable batteries [1,2]. -

Page 1 of 9 RSC Advances
RSC Advances This is an Accepted Manuscript, which has been through the Royal Society of Chemistry peer review process and has been accepted for publication. Accepted Manuscripts are published online shortly after acceptance, before technical editing, formatting and proof reading. Using this free service, authors can make their results available to the community, in citable form, before we publish the edited article. This Accepted Manuscript will be replaced by the edited, formatted and paginated article as soon as this is available. You can find more information about Accepted Manuscripts in the Information for Authors. Please note that technical editing may introduce minor changes to the text and/or graphics, which may alter content. The journal’s standard Terms & Conditions and the Ethical guidelines still apply. In no event shall the Royal Society of Chemistry be held responsible for any errors or omissions in this Accepted Manuscript or any consequences arising from the use of any information it contains. www.rsc.org/advances Page 1 of 9 RSC Advances Self-assembled multimicellar vesicles via complexation of a rigid conjugated polymer with an amphiphilic block copolymer Anbazhagan Palanisamy and Qipeng Guo* Polymers Research Group, Institute for Frontier Materials, Deakin University, Locked Bag 2000, Geelong, Victoria 3220, Australia. We report here a facile method for fabrication of multimicellar vesicles from self-assembled complexes of a flexible coil-like block copolymer and a rigid rod conjugated homopolymer. Manuscript Accepted Advances RSC RSC Advances Page 2 of 9 Journal Name RSCPublishing ARTICLE Self-assembled multimicellar vesicles via complexation of a rigid conjugated polymer with Cite this: DOI: 10.1039/x0xx00000x amphiphilic block copolymer Anbazhagan Palanisamy and Qipeng Guo* Received 00th January 2012, Accepted 00th January 2012 A viable method of encapsulating block copolymer micelles inside vesicles using a conjugated polymer is DOI: 10.1039/x0xx00000x reported in this study. -

(Meta-)Cresol 1,1,1,2,3,3,3 Hepta Fluoropropane 1,1,1,2,3,3,3
(META-)CRESOL CARBOXYLATED STYRENE BUTADINE POLYMER 1,1,1,2,3,3,3 HEPTA FLUOROPROPANE CHLORINATED POLYVINYL CHLORIDE 1,1,1,2,3,3,3-HEPTAFLUOROPROPANE CHLOROACETIC ACID 1,1-METHYLENEBIS[4-ISOCYANATO-BENZENE CHLOROFORM 1,2 - PROPANEDIOL CHLOROMETHYL CHLOROFORMATE 1,2-PROPANEDIOL CYCLOHEXANEDIAMINETETRAACETIC ACID 1-METHOXY-2-PROPANOL CYCLOHEXANONE 1-OCTENE POLYMER WITH ETHENE CYCLO-SOL (BENZENE) 1-THIOGLYCEROL DIACETONE ALCOHOL 50%, ROSIN ESTER 2-(2-AMINOETHOXY) ETHANOL DIAMINOCYCLOHEXANE 2,4-TOLUENE DIISOCYANATE DIETHYLENE GLYCOL ETHER ACETATE 2,5-FURANDIONE, POLYMER WITH 1-PROPENE DIETHYLENE GLYCOL MONO ETHYL ETHER 2-BENZYLIDENEHEPTANAL DIETHYLENE GLYCOL MONOBUTYL ETHER 2-BROMO-2-NITRO-1,3-PROPANEDIOL DIETHYLENE GLYCOL MONOETHYL ETHER 2-BUTOXY ETHANOL DIETHYLETHANOLAMINE 2-BUTOXYETHANOL DIGLYCIDYL ETHER OF BISPHENOL A 2-HEPTANONE DIMETHYLETHANOLAMINE-EPICHLORHYDRIN 2-METHOXY-1-METHYL ACETATE DIPROPYLENE GLYCOL 4,4-DIPHENYLMETHANE DIISOCYANATE DIPROPYLENE GLYCOL METHYL ETHER 4-4-ISOPROPYLIDENEDIPHENOL DIPROPYLENE GLYCOL MONOMETHYL ETHER 4-AMINO BENZENE ETHANOL DIPROPYLENE GLYCOL TERTBUTYL ETHER 4-METHYL-2-PENTANOL DODECYLBENZENE SULFONIC ACID 4-NONYLPHENOL, BRANCHED EMULSIFIED STYRENE BUTADIENE / ACRYLONIT 5-CHLORO-2-PENTANONE EPICHLORHYDRIIN/BISPHENOL-A EPOXY RESIN 6-DIAZO-5,6-DIHYDRO-5-OXY-1- ETHANE, 1,1,1,2-TETRAFLUORO (HFC-134A) NAPHTHALENES ETHANOL ACETALDEHYDE DIETHYL ACETAL, NATURAL ETHANOL, 2-((2-AMINO-1- ACETIC ACID METHYLETHYL)AMINO ACETIC ACID/SULFURIC ACID/SILVER ETHANOL, 2,2',2"-NITRILOTRIS- ACETONE ETHANOLAMINE ACETYLENE DISSOLVED -

Nanostructure of Poly(Acrylic Acid) Adsorption Layer on the Surface of Activated Carbon Obtained from Residue After Supercritical Extraction of Hops M
Wiśniewska et al. Nanoscale Research Letters (2017) 12:2 DOI 10.1186/s11671-016-1772-3 NANOEXPRESS Open Access Nanostructure of Poly(Acrylic Acid) Adsorption Layer on the Surface of Activated Carbon Obtained from Residue After Supercritical Extraction of Hops M. Wiśniewska1, A. Nosal-Wiercińska2, I. Ostolska1, D. Sternik3, P. Nowicki4, R. Pietrzak4, A. Bazan-Wozniak4 and O. Goncharuk5* Abstract The nanostructure of poly(acrylic acid) (PAA) adsorption layer on the surface of mesoporous-activated carbon HPA obtained by physical activation of residue after supercritical extraction of hops was characterized. This characterization has been done based on the analysis of determination of adsorbed polymer amount, surface charge density, and zeta potential of solid particles (without and in the PAA presence). The SEM, thermogravimetric, FTIR, and MS techniques have allowed one to examine the solid surface morphology and specify different kinds of HPA surface groups. The effects of solution pH, as well as polymer molecular weight and concentration, were studied. The obtained results indicated that the highest adsorption on the activated carbon surface was exhibited by PAA with lower molecular weight (i.e., 2000 Da) at pH 3. Under such conditions, polymeric adsorption layer is composed of nanosized PAA coils (slightly negatively charged) which are densely packed on the positive surface of HPA. Additionally, the adsorption of polymeric macromolecules into solid pores is possible. Keywords: Mesoporous activated carbon, Poly(acrylic acid) adsorption, Nanostructure of adsorption layer, Physical activation, Electrokinetic properties Background The conformation of polymeric chains (especially Specific structure of polymeric adsorption layer formed those classified as ionic polymers) can be influenced by on the solid-liquid interface determines the surface prop- many factors, among which the most essential are as erties of colloidal suspension. -

Improved Cement Admixture Product Having Improved Rheological Properties and Process of Forming Same
Patentamt Europaisches || || 1 1| || || ||| 1 1| 1 1 1|| || || (19) J European Patent Office Office europeen des brevets (11) EP 0 739 320 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) int. CI.6: C04B 24/26, C08F 8/30, of the grant of the patent: C08F 8/48 08.12.1999 Bulletin 1999/49 (86) International application number: number: 94928051.5 (21) Application PCT/US94/10134 (22) Date of filing: 13.09.1994 (87) International publication number: WO 95/09821 (13.04.1995 Gazette 1 995/1 6) (54) IMPROVED CEMENT ADMIXTURE PRODUCT HAVING IMPROVED RHEOLOGICAL PROPERTIES AND PROCESS OF FORMING SAME ZUSAMMENSETZUNG ZUM VERBESSERN DER RHEOLOGISCHEN EIGENSCHAFTEN ZEMENTBASIERTER PRODUKTE UND VERFAHREN ZUR HERSTELLUNG DAVON ADJUVANT DU CIMENT AMELIORE PRESENTANT DE MEILLEURS PROPRIETES RHEOLOGIQUES ET SON PROCEDE DE FABRICATION (84) Designated Contracting States: • KOYATA, Hideo DE DK ES FR GB IT Yokohama, Kanagawa 245 (JP) • KUO, Lawrence, Lu (30) Priority: 29.09.1993 US 128939 Columbia, MD 21045 (US) 06.09.1994 US 299028 (74) Representative: (43) Date of publication of application: Senior, Alan Murray 30.10.1996 Bulletin 1996/44 J.A. KEMP & CO., 14 South Square, (73) Proprietor: Gray's Inn W.R. GRACE & CO.-CONN. London WC1 R 5LX (GB) New York New York 10036-7794 (US) (56) References cited: (72) Inventors: EP-A- 0 056 627 EP-A- 0 300 760 • ARFAEI, Ahmad EP-A- 0 448 717 WO-A-92/07010 Chelmsford, MA 01824-4153 (US) DE-A- 4 034 708 FR-A- 2 224 498 • DARWIN, David, Charles FR-A- 2 470 140 GB-A- 879 950 Columbia, MD 21045 (US) • GARTNER, Ellis, Martin PATENT ABSTRACTS OF JAPAN vol. -

Polyacrylic Acid Sds, Polyacrylic Acid Msds
Safety Data Sheet 24 hour Emergency Response No. +86-532-8027466 SECTION 1: PRODUCT AND COMPANY IDENTIFICATION Shandong IRO Water Treatment Co.,Ltd Mushi Town, Tengzhou, Zaozhuang, IR-PAA (50) China IRO phone: +86-532-8027466 PRODUCT USE: Water Treatment Application Formulation Personal Protection Equipment GHS DOT HMIS Pictograph Symbol(s) (See last page for guide to HMIS) DOT Proper HEALTH 1 Shipping Name: Not FLAMMABILITY 0 regulated as a hazardous PHYSICAL HAZARD 0 material PERSONAL PROTECTION B SECTION 2: HAZARDSShandong IDENTIFICATION IRO Water Co.,Ltd EMERGENCY OVERVIEW GHS CLASSIFICATION Prevention Precautionary Statements Signal Word Warning: P234: Keep only in original container. DANGER P260: Do not breathe. P264: Wash hands thoroughly after handling. P280: Wear protective gloves/eye protection/face protection. Response Precautionary Statements Co.,Ltd P305+351+338: IF IN EYES: Rinse cautiously with water for several minutes. IRRITANT Remove contact lenses, if present and easy to do. Continue rinsing. GHS Hazard Statements P337+313: If eye irritation persists, get medical advice/attention. H290: May be corrosive to metals. H303: May be harmful if swallowed. Water H319: Causes serious eye irritation RESPONSE: See section 4: FIRST AID MEASURES.IRO Also, see below: • Do not handle until all safety precautions have been read and understood. • Wear gloves, eye protection and face protection (as needed to prevent skin and eye contact Wash hands or liquid-contacted skin thoroughly after handling. • Do not eat, drink or smoke when using this product. STORAGE: Store in a well ventilated place. Keep cool. Store locked up. Keep container tightly closed. Use only approved containers. DISPOSAL: Dispose of contents/containers to approved disposal site in accordance with local, regional, and/or international regulations. -

The Molecular Weight Effects of Poly(Acrylic Acid) on Calcium Carbonate Inhibition in the Kraft Pulping Process
The Molecular Weight Effects of Poly(acrylic acid) on Calcium Carbonate Inhibition in the Kraft Pulping Process A DISSERTATION SUBMITTED TO THE FACULTY OF THE GRADUATE SCHOOL OF THE UNIVERSITY OF MINNESOTA BY Matthew Richard Dubay IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY Steven J. Severtson, Advisor May, 2011 © Matthew R. Dubay 2011 Acknowledgements First and foremost, I want to thank the Creator, my Savior, and the Comforter for their priceless influences on my life. Also, I hold an enormous amount of gratitude and respect for all of the people responsible for helping and guiding me as a graduate student. In particular, I would like to sincerely thank Dr. Steve Severtson for his seemingly unending supply of experiential wisdom and genuine concern for my progress not only as a student but also as a teacher, independent researcher, and leader. Beyond the classroom, he has been my mentor (and friend) in innumerable ways. All the members of his research group have been instrumental in my success as a graduate student –especially Dr. Gang Pu for the massive amount of time he generously donated while helping me organize this thesis and Dr. Jiguang Zhang for his help and guidance with polymerization techniques. I would also like to thank Dr. Andreas Stein and his group members (particularly Melissa Fierke) for their continual support and help with many of the issues that sprung up during this research. The other members of my thesis committee, Dr. William Tze and Dr. Shri Ramaswamy, were instrumental in their support and review of this thesis, of which I am grateful for. -

Water Soluble Polymers for Pharmaceutical Applications
Polymers 2011, 3, 1972-2009; doi:10.3390/polym3041972 OPEN ACCESS polymers ISSN 2073-4360 www.mdpi.com/journal/polymers Review Water Soluble Polymers for Pharmaceutical Applications Veeran Gowda Kadajji and Guru V. Betageri * Department of Pharmaceutical Sciences, Western University of Health Sciences, Pomona, CA 91766, USA; E-Mail: [email protected] * Author to whom correspondence should be addressed; E-Mail: [email protected]; Tel.: +1-909-469-5682; Fax: +1-909-469-5600. Received: 22 September 2011 / Accepted: 10 November 2011 / Published: 11 November 2011 Abstract: Advances in polymer science have led to the development of novel drug delivery systems. Some polymers are obtained from natural resources and then chemically modified for various applications, while others are chemically synthesized and used. A large number of natural and synthetic polymers are available. In the present paper, only water soluble polymers are described. They have been explained in two categories (1) synthetic and (2) natural. Drug polymer conjugates, block copolymers, hydrogels and other water soluble drug polymer complexes have also been explained. The general properties and applications of different water soluble polymers in the formulation of different dosage forms, novel delivery systems and biomedical applications will be discussed. Keywords: polymers; natural; synthetic; hydrogels; gums; cellulose ethers; povidone; polyethylene glycol; polyacrylamides; polyacrylic acid copolymers 1. Introduction Advances in polymer science have led to the development of novel delivery systems. The introduction of new polymers has resulted in development of polymers with unique properties. Initially polymers were used as solubilisers, stabilizers and mechanical supports for sustained release of drugs. But over a period of time, the functionalities of polymers have changed.