Novel Treatments for Mood Disorders
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Corporate Release No 485 12 December 2012 FDA Accepts
H. Lundbeck A/S Ottiliavej 9 Tel +45 36 30 13 11 E-mail [email protected] DK-2500 Valby, Copenhagen Fax +45 36 43 82 62 www.lundbeck.com CVR number: 56759913 Corporate Release No 485 12 December 2012 FDA accepts Takeda and Lundbeck’s filing for review of Brintellix (vortioxetine) for the treatment of major depression FDA has determined that the New Drug Application filed in October 2012 is sufficiently complete to permit a substantive review Upon the acceptance of the filing by the FDA, Lundbeck is to receive a milestone of USD 50 million (approximately DKK 285 million) from Takeda H. Lundbeck A/S (Lundbeck) announced today that the U.S. Food and Drug Administration (FDA) has accepted the submission of a New Drug Application (NDA) for BrintellixTM (vortioxetine) for the treatment of major depressive disorder (MDD) in adult patients. Brintellix (pronounced “brin′-tel-ix”) is the proposed global trade name for vortioxetine. According to the timelines established by the Prescription Drug User Fee Act (PDUFA), the review of the NDA is targeted for completion by 2 October, 2013. The NDA includes positive data from six short-term studies and one long-term maintenance study. The vortioxetine global clinical development program included more than 7,500 individuals aged 18 to 88 years old exposed to the drug. Major depression, often referred to as depression, is a common, debilitating illness affecting around 15 million Americans and 121 million people worldwide. Depression was the third leading contributor to the global burden of disease in 2004 and is projected to be the leading contributor to the worldwide burden of disease by 2030. -

Download Resume
Curriculum Vitae Full Name: David M. Marks, M.D. Contact: [email protected] Mobile (619) 822-7117 Credentials: Diplomate, American Board of Psychiatry and Neurology (Psychiatry) Subspecialty Certification in Psychosomatic Medicine Diplomate, American Board of Pain Medicine Position Title: Associate Professor Department of Psychiatry and Behavioral Sciences Department of Community and Family Medicine Duke University Medical Center Duke Clinical Research Institute Duke Pain and Palliative Care Clinic Education: Institution & Location Degree Year Conferred Field of Study University of California at San Diego Fellowship 1999 - 2000 Consultation and San Diego, CA Liaison Psychiatry Medical College of Pennsylvania / Senior 1998 - 1999 Psychiatry Clinical Neuroscience Research Unit Resident Philadelphia, PA University of California at San Diego Resident 1995 - 1998 Psychiatry San Diego, CA University of Texas Medical Branch M.D. 1995 Galveston, TX Rice University B.A. 1991 Psychology Houston, TX Research and Professional Experience: Position Institution/Employer & Location Dates of Employment Attending Faculty Physician Duke Pain and Palliative Care Clinic 09/08-present (Chronic Pain Management) Attending Faculty Physician Duke University Medical Center, 07/06-present Inpatient Psychiatric Service, Emergency Service, Consultation/Liaison Service Attending Faculty Physician Durham Regional Hospital, 09/08-07/11 Consultation/Liaison Service Medical Director, Inpatient and Duke University Medical Center 07/06 – 02/07 Emergency Psychiatry Services Medical Director, CNS Division EStudy Site 05/05 -- 07/06 La Mesa, Oceanside, National City CA Chief Executive Officer/Medical Optimum Health Services 01/02 – 05/05 Director La Mesa, Oceanside CA Chief of Staff Alvarado Parkway Institute 01/04 – 01/05 1 La Mesa, CA Page _____________________________________________________________________ David M. -

New Drug Evaluation Monograph Template
© Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 | Fax 503-947-1119 Class Update: Second Generation Antidepressant Medications Month/Year of Review: May 2014 Last Oregon Review: April 2012 PDL Classes: Psychiatric: Antidepressants Source Document: OSU College of Pharmacy New drug(s): vortioxetine (Brintellix®) Manufacturer: Takeda & Lundbeck/Forest levomilnacipran extended-release (Fetzima®) Dossier Received: Yes/Pending Current Status of Voluntary PDL Class: Preferred Agents: BUPROPION HCL TABLET/TABLET ER, CITALOPRAM TABLET/SOLUTION, FLUOXETINE CAPSULE/SOLUTION/TABLET, FLUVOXAMINE, MIRTAZEPINE TAB RAPDIS/TABLET, PAROXETINE TABLET, SERTRALINE ORAL CONC/TABLET, VENLAFAXINE TABLET, VENLAFAXINE ER Non-Preferred Agents: BUPROPRION XL, DESVENLAFAXINE (PRISTIQ ER), DULOXETINE (CYMBALTA®), ESCITALOPRAM, FLUOXETINE DF (PROZAC® WEEKLY), NEFAZODONE, PAROXETINE HCL (PAXIL CR®), SELEGILINE PATCH (ENSAM®), VILAZODONE (VIIBRYD®), OLANZAPINE/FLUOXETINE (SYMBYAX®) Status of the Voluntary Mental Health Preferred Drug List Currently, all antidepressants are available without prior authorization for non-preferred placement. Oregon law prohibits traditional methods of PDL enforcement on mental health drugs. Second generation antidepressants have been reviewed for clinical efficacy and safety and specific agents were chosen as clinically preferred; this eliminates a copay. Oregon’s Medicaid program currently -

Pristiq (Desvenlafaxine Succinate) – First-Time Generic
Pristiq® (desvenlafaxine succinate) – First-time generic • On March 1, 2017, Teva launched AB-rated generic versions of Pfizer’s Pristiq (desvenlafaxine succinate) 25 mg, 50 mg, and 100 mg extended-release tablets for the treatment of major depressive disorder. — Teva launched the 25 mg tablet with 180-day exclusivity. — In addition, Alembic/Breckenridge, Mylan, and West-Ward have launched AB-rated generic versions of Pristiq 50 mg and 100 mg extended-release tablets. — Greenstone’s launch plans for authorized generic versions of Pristiq 25 mg, 50 mg, and 100 mg tablets are pending. — Lupin and Sandoz received FDA approval of AB-rated generic versions of Pristiq 50 mg and 100 mg tablets on June 29, 2015. Lupin’s and Sandoz’s launch plans are pending. • Other serotonin-norepinephrine reuptake inhibitors approved for the treatment of major depressive disorder include desvenlafaxine fumarate, duloxetine, Fetzima™ (levomilnacipran), Khedezla™ (desvenlafaxine) , venlafaxine, and venlafaxine extended-release. • Pristiq and the other serotonin-norepinephrine reuptake inhibitors carry a boxed warning for suicidal thoughts and behaviors. • According to IMS Health data, the U.S. sales of Pristiq were approximately $883 million for the 12 months ending on December 31, 2016. optumrx.com OptumRx® specializes in the delivery, clinical management and affordability of prescription medications and consumer health products. We are an Optum® company — a leading provider of integrated health services. Learn more at optum.com. All Optum® trademarks and logos are owned by Optum, Inc. All other brand or product names are trademarks or registered marks of their respective owners. This document contains information that is considered proprietary to OptumRx and should not be reproduced without the express written consent of OptumRx. -

Stems for Nonproprietary Drug Names
USAN STEM LIST STEM DEFINITION EXAMPLES -abine (see -arabine, -citabine) -ac anti-inflammatory agents (acetic acid derivatives) bromfenac dexpemedolac -acetam (see -racetam) -adol or analgesics (mixed opiate receptor agonists/ tazadolene -adol- antagonists) spiradolene levonantradol -adox antibacterials (quinoline dioxide derivatives) carbadox -afenone antiarrhythmics (propafenone derivatives) alprafenone diprafenonex -afil PDE5 inhibitors tadalafil -aj- antiarrhythmics (ajmaline derivatives) lorajmine -aldrate antacid aluminum salts magaldrate -algron alpha1 - and alpha2 - adrenoreceptor agonists dabuzalgron -alol combined alpha and beta blockers labetalol medroxalol -amidis antimyloidotics tafamidis -amivir (see -vir) -ampa ionotropic non-NMDA glutamate receptors (AMPA and/or KA receptors) subgroup: -ampanel antagonists becampanel -ampator modulators forampator -anib angiogenesis inhibitors pegaptanib cediranib 1 subgroup: -siranib siRNA bevasiranib -andr- androgens nandrolone -anserin serotonin 5-HT2 receptor antagonists altanserin tropanserin adatanserin -antel anthelmintics (undefined group) carbantel subgroup: -quantel 2-deoxoparaherquamide A derivatives derquantel -antrone antineoplastics; anthraquinone derivatives pixantrone -apsel P-selectin antagonists torapsel -arabine antineoplastics (arabinofuranosyl derivatives) fazarabine fludarabine aril-, -aril, -aril- antiviral (arildone derivatives) pleconaril arildone fosarilate -arit antirheumatics (lobenzarit type) lobenzarit clobuzarit -arol anticoagulants (dicumarol type) dicumarol -

Desvenlafaxine
https://providers.amerigroup.com CONTAINS CONFIDENTIAL PATIENT INFORMATION desvenlafaxine Prior Authorization of Benefits (PAB) Form Complete form in its entirety and fax to: Prior Authorization of Benefits Center at 1-844-512-9004 Provider Help Desk 1-800-454-3730 1. PATIENT INFORMATION 2. PHYSICIAN INFORMATION Patient Name: Prescribing Physician: Patient ID #: Physician Address: Patient DOB: Physician Phone #: Date of Rx: Physician Fax #: Patient Phone #: Physician Specialty: Patient Email Address: Physician DEA: Physician NPI #: Physician Email Address: 3. MEDICATION 4. STRENGTH 5. DIRECTIONS 6. QUANTITY PER 30 DAYS desvenlafaxine Specify: 7. DIAGNOSIS: 8. APPROVAL CRITERIA: CHECK ALL BOXES THAT APPLY NOTE: Any areas not filled out are considered not applicable to your patient & MAY AFFECT THE OUTCOME of this request. □ Yes □ No Documentation of a previous trial and therapy failure at a therapeutic dose with two preferred generic SSRIs* has been provided If No: □ Yes □ No Documented evidence is provided that the use of these agents would be medically contraindicated □ Yes □ No Documentation of a previous trial and therapy failure at a therapeutic dose with one preferred generic SNRI** has been provided If No: □ Yes □ No Documented evidence is provided that the use of these agents would be medically contraindicated □ Yes □ No Documentation of a previous trial and therapy failure at a therapeutic dose with one non-SSRI/SNRI generic antidepressant** has been provided If No: □ Yes □ No Documented evidence is provided that the use of these -
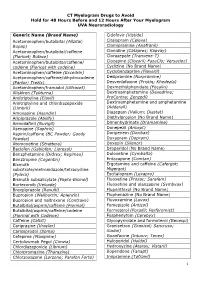
CT Myelogram Drugs to Avoid Hold for 48 Hours Before and 12 Hours After Your Myelogram UVA Neuroradiology
CT Myelogram Drugs to Avoid Hold for 48 Hours Before and 12 Hours After Your Myelogram UVA Neuroradiology Generic Name (Brand Name) Cidofovir (Vistide) Acetaminophen/butalbital (Allzital; Citalopram (Celexa) Bupap) Clomipramine (Anafranil) Acetaminophen/butalbital/caffeine Clonidine (Catapres; Kapvay) (Fioricet; Butace) Clorazepate (Tranxene-T) Acetaminophen/butalbital/caffeine/ Clozapine (Clozaril; FazaClo; Versacloz) codeine (Fioricet with codeine) Cyclizine (No Brand Name) Acetaminophen/caffeine (Excedrin) Cyclobenzaprine (Flexeril) Acetaminophen/caffeine/dihydrocodeine Desipramine (Norpramine) (Panlor; Trezix) Desvenlafaxine (Pristiq; Khedezla) Acetaminophen/tramadol (Ultracet) Dexmethylphenidate (Focalin) Aliskiren (Tekturna) Dextroamphetamine (Dexedrine; Amitriptyline (Elavil) ProCentra; Zenzedi) Amitriptyline and chlordiazepoxide Dextroamphetamine and amphetamine (Limbril) (Adderall) Amoxapine (Asendin) Diazepam (Valium; Diastat) Aripiprazole (Abilify) Diethylpropion (No Brand Name) Armodafinil (Nuvigil) Dimenhydrinate (Dramamine) Asenapine (Saphris) Donepezil (Aricept) Aspirin/caffeine (BC Powder; Goody Doripenem (Doribax) Powder) Doxapram (Dopram) Atomoxetine (Strattera) Doxepin (Silenor) Baclofen (Gablofen; Lioresal) Droperidol (No Brand Name) Benzphetamine (Didrex; Regimex) Duloxetine (Cymbalta) Benztropine (Cogentin) Entacapone (Comtan) Bismuth Ergotamine and caffeine (Cafergot; subcitrate/metronidazole/tetracycline Migergot) (Pylera) Escitalopram (Lexapro) Bismuth subsalicylate (Pepto-Bismol) Fluoxetine (Prozac; Sarafem) -

Antidepressants, Other Review 04/14/2009
Antidepressants, Other Review 04/14/2009 Copyright © 2004 - 2009 by Provider Synergies, L.L.C. All rights reserved. Printed in the United States of America. All rights reserved. No part of this publication may be reproduced or transmitted in any form or by any means, electronic or mechanical, including photocopying, recording, digital scanning, or via any information storage and retrieval system without the express written consent of Provider Synergies, L.L.C. All requests for permission should be mailed to: Attention: Copyright Administrator Intellectual Property Department Provider Synergies, L.L.C. 5181 Natorp Blvd., Suite 205 Mason, Ohio 45040 The materials contained herein represent the opinions of the collective authors and editors and should not be construed to be the official representation of any professional organization or group, any state Pharmacy and Therapeutics committee, any state Medicaid Agency, or any other clinical committee. This material is not intended to be relied upon as medical advice for specific medical cases and nothing contained herein should be relied upon by any patient, medical professional or layperson seeking information about a specific course of treatment for a specific medical condition. All readers of this material are responsible for independently obtaining medical advice and guidance from their own physician and/or other medical professional in regard to the best course of treatment for their specific medical condition. This publication, inclusive of all forms contained herein, is intended to be -

Desvenlafaxine Drug Assessment Pristiq® in Major Depressive Report Disorder in Adults @DTB Navarre.Es More Cost for Less
03 2015 DESVENLAFAXINE DRUG ASSESSMENT Pristiq® in major depressive REPORT www.dtb.navarra.es disorder in adults @DTB_Navarre.es More cost for less... ABSTRACT Desvenlafaxine is an active me- tabolite of venlafaxine. Indications1 In the study against escitalopram4 flexible Treatment of major depressive disorder doses of desvenlafaxine were used (100 and In three placebo-controlled 200 mg daily) in postmenopausal women. No trials, the results on the reduc- 1 Mechanism of action advantage of desvenlafaxine over escitalopram tion in HAM-D17 score were not This is an active metabolite of venlafaxine, that was found. consistent. inhibits the reuptake of serotonine and nora- There is only one long-term study16 that eva- dernaline. Its bioavailability reaches up to 80% luated relapse prevention. Patients responding The most common adverse while elimination occurs without alteration after 8 weeks of treatment with desvenlafaxine effects are of gastrointestinal through the urine (45%) and metabolism by 50 mg daily and with a stable response up to origin or sleep disorders. There glucurono conjugation (19%). week 20 were randomized either to placebo or are no available long-term safe- desvenlafaxine 50 mg daily for 6 months. The ty data. Dosage and administration1 endpoint was time to relapse (defined as HAM- The recommended dose is 50 mg daily. The ta- D17 score ≥16), treatment withdrawal due to In the only head-to-head trial blets are swallowed wholly, with liquid with or unsatisfactory response, hospital admission carried out in post-menopause without food and at the same time. The increa- due to depression, suicide attempt or suicide. women, desvenlafaxine at high se in doses should be gradual and up to a maxi- Time to relapse was significantly lower in the doses did not show superiority mum of 200 mg daily and at intervals of at least case of placebo compared to desvenlafaxine versus escitalopram. -

GPCR/G Protein
Inhibitors, Agonists, Screening Libraries www.MedChemExpress.com GPCR/G Protein G Protein Coupled Receptors (GPCRs) perceive many extracellular signals and transduce them to heterotrimeric G proteins, which further transduce these signals intracellular to appropriate downstream effectors and thereby play an important role in various signaling pathways. G proteins are specialized proteins with the ability to bind the nucleotides guanosine triphosphate (GTP) and guanosine diphosphate (GDP). In unstimulated cells, the state of G alpha is defined by its interaction with GDP, G beta-gamma, and a GPCR. Upon receptor stimulation by a ligand, G alpha dissociates from the receptor and G beta-gamma, and GTP is exchanged for the bound GDP, which leads to G alpha activation. G alpha then goes on to activate other molecules in the cell. These effects include activating the MAPK and PI3K pathways, as well as inhibition of the Na+/H+ exchanger in the plasma membrane, and the lowering of intracellular Ca2+ levels. Most human GPCRs can be grouped into five main families named; Glutamate, Rhodopsin, Adhesion, Frizzled/Taste2, and Secretin, forming the GRAFS classification system. A series of studies showed that aberrant GPCR Signaling including those for GPCR-PCa, PSGR2, CaSR, GPR30, and GPR39 are associated with tumorigenesis or metastasis, thus interfering with these receptors and their downstream targets might provide an opportunity for the development of new strategies for cancer diagnosis, prevention and treatment. At present, modulators of GPCRs form a key area for the pharmaceutical industry, representing approximately 27% of all FDA-approved drugs. References: [1] Moreira IS. Biochim Biophys Acta. 2014 Jan;1840(1):16-33. -

Current Phase III Clinical Trials
Toolbox: Current phase III clinical trials Tiffany-Jade Kreys, PharmD1 1Assistant Professor of Pharmacy Practice University of the Incarnate Word Feik School of Pharmacy, San Antonio, Texas KEYWORDS clinical trials, depression, bipolar, schizophrenia, medications Table 1. Novel Agents for Major Depression, Bipolar Disorder, and/or Schizophrenia Product Name Sponsor Indication Mechanism of Action (previous name) Downloaded from http://meridian.allenpress.com/mhc/article-pdf/2/6/138/2094102/mhc_n129046.pdf by guest on 26 September 2021 Amitifadine Euthymics MDD Triple Reuptake Inhibitor (EB-1010) Bioscience (1:2:8 ratio of serotonin: norepinephrine: dopamine inhibition) Bitopertin Roche Schizophrenia Glycine reuptake inhibitor (RG1678) Enhances NMDA receptor activity Brexpiprazole Lundbeck MDD, Claimed to have "broad activity across multiple monoamine (OPC-34712) Otsuka America Schizophrenia systems and exhibits reduced partial agonist activity at D2 Pharmaceuticals receptors and enhanced affinity for specific serotonin receptors" Cariprazine Forest Laboratories BPAD, MDD adjunct D3-preferring/D2 receptor partial agonist (RGH-188) therapy, Schizophrenia Citalopram/ PharmaNeuro- MDD 5HT2A/D4 antagonist Pipamperone Boost (PNB01) Edivoxetine Eli Lilly MDD adjunct therapy Norepinephrine reuptake inhibitor (LY2216684) Vortioxetine Lundbeck MDD In vitro studies: 5HT3 and 7 receptor antagonist, 5HT1B partial (LUAA21004) Takeda agonist, 5HT1A agonist, serotonin transporter inhibitor Pharmaceuticals In vivo: Increases serotonin, norepinephrine, dopamine, -

Patent Application Publication ( 10 ) Pub . No . : US 2019 / 0192440 A1
US 20190192440A1 (19 ) United States (12 ) Patent Application Publication ( 10) Pub . No. : US 2019 /0192440 A1 LI (43 ) Pub . Date : Jun . 27 , 2019 ( 54 ) ORAL DRUG DOSAGE FORM COMPRISING Publication Classification DRUG IN THE FORM OF NANOPARTICLES (51 ) Int . CI. A61K 9 / 20 (2006 .01 ) ( 71 ) Applicant: Triastek , Inc. , Nanjing ( CN ) A61K 9 /00 ( 2006 . 01) A61K 31/ 192 ( 2006 .01 ) (72 ) Inventor : Xiaoling LI , Dublin , CA (US ) A61K 9 / 24 ( 2006 .01 ) ( 52 ) U . S . CI. ( 21 ) Appl. No. : 16 /289 ,499 CPC . .. .. A61K 9 /2031 (2013 . 01 ) ; A61K 9 /0065 ( 22 ) Filed : Feb . 28 , 2019 (2013 .01 ) ; A61K 9 / 209 ( 2013 .01 ) ; A61K 9 /2027 ( 2013 .01 ) ; A61K 31/ 192 ( 2013. 01 ) ; Related U . S . Application Data A61K 9 /2072 ( 2013 .01 ) (63 ) Continuation of application No. 16 /028 ,305 , filed on Jul. 5 , 2018 , now Pat . No . 10 , 258 ,575 , which is a (57 ) ABSTRACT continuation of application No . 15 / 173 ,596 , filed on The present disclosure provides a stable solid pharmaceuti Jun . 3 , 2016 . cal dosage form for oral administration . The dosage form (60 ) Provisional application No . 62 /313 ,092 , filed on Mar. includes a substrate that forms at least one compartment and 24 , 2016 , provisional application No . 62 / 296 , 087 , a drug content loaded into the compartment. The dosage filed on Feb . 17 , 2016 , provisional application No . form is so designed that the active pharmaceutical ingredient 62 / 170, 645 , filed on Jun . 3 , 2015 . of the drug content is released in a controlled manner. Patent Application Publication Jun . 27 , 2019 Sheet 1 of 20 US 2019 /0192440 A1 FIG .