PROCLAMATION 6982—APR. 1, 1997 111 STAT. 2871 to Implement an Agreement to Eliminate Tariffs on Certain Pharmaceuticals and Ch
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Customs Tariff - Schedule
CUSTOMS TARIFF - SCHEDULE 99 - i Chapter 99 SPECIAL CLASSIFICATION PROVISIONS - COMMERCIAL Notes. 1. The provisions of this Chapter are not subject to the rule of specificity in General Interpretative Rule 3 (a). 2. Goods which may be classified under the provisions of Chapter 99, if also eligible for classification under the provisions of Chapter 98, shall be classified in Chapter 98. 3. Goods may be classified under a tariff item in this Chapter and be entitled to the Most-Favoured-Nation Tariff or a preferential tariff rate of customs duty under this Chapter that applies to those goods according to the tariff treatment applicable to their country of origin only after classification under a tariff item in Chapters 1 to 97 has been determined and the conditions of any Chapter 99 provision and any applicable regulations or orders in relation thereto have been met. 4. The words and expressions used in this Chapter have the same meaning as in Chapters 1 to 97. Issued January 1, 2020 99 - 1 CUSTOMS TARIFF - SCHEDULE Tariff Unit of MFN Applicable SS Description of Goods Item Meas. Tariff Preferential Tariffs 9901.00.00 Articles and materials for use in the manufacture or repair of the Free CCCT, LDCT, GPT, UST, following to be employed in commercial fishing or the commercial MT, MUST, CIAT, CT, harvesting of marine plants: CRT, IT, NT, SLT, PT, COLT, JT, PAT, HNT, Artificial bait; KRT, CEUT, UAT, CPTPT: Free Carapace measures; Cordage, fishing lines (including marlines), rope and twine, of a circumference not exceeding 38 mm; Devices for keeping nets open; Fish hooks; Fishing nets and netting; Jiggers; Line floats; Lobster traps; Lures; Marker buoys of any material excluding wood; Net floats; Scallop drag nets; Spat collectors and collector holders; Swivels. -

An Introduction to Medicinal
An Introduction to Medicinal Chemistry FIFTH EDITION Graham L. Patrick 1 Patrick97397.indb iii 11/28/2012 9:11:45 PM 1 Great Clarendon Street, Oxford, OX2 6DP, United Kingdom Oxford University Press is a department of the University of Oxford. It furthers the University’s objective of excellence in research, scholarship, and education by publishing worldwide. Oxford is a registered trade mark of Oxford University Press in the UK and in certain other countries © Graham L. Patrick 2013 Th e moral rights of the author have been asserted Second Edition copyright 2001 Th ird Edition copyright 2005 Fourth Edition copyright 2009 Impression: 1 All rights reserved. No part of this publication may be reproduced, stored in a retrieval system, or transmitted, in any form or by any means, without the prior permission in writing of Oxford University Press, or as expressly permitted by law, by licence or under terms agreed with the appropriate reprographics rights organization. Enquiries concerning reproduction outside the scope of the above should be sent to the Rights Department, Oxford University Press, at the address above You must not circulate this work in any other form and you must impose this same condition on any acquirer British Library Cataloguing in Publication Data Data available ISBN 978–0–19–969739–7 Printed in Italy by L.E.G.O. S.p.A.—Lavis TN Links to third party websites are provided by Oxford in good faith and for information only. Oxford disclaims any responsibility for the materials contained in any third party website referenced in this work. -

A Synthetic Overview of New Molecules with 5-HT1A Binding Affinities
77 A Synthetic Overview of New Molecules with 5-HT1A Binding Affinities Hernán Pessoa-Mahana* 1 ; Ramiro Araya-Maturana1 , Claudio Saitz, B.1 and C. David Pessoa-Mahana2 1Departamento de Química Orgánica y Fisicoquímica. Facultad de Ciencias Químicas y Farmacéuticas. Universidad de Chile. Olivos 1007.Casilla 233. Santiago 1. Chile 2Departamento de Farmacia. Facultad de Química. Pontificia Universidad Católica de Chile. Vicuña Mackenna 4860-Casilla 306, Correo 22 Santiago-Chile Abstract: The present review discusses the synthetic strategies of new ligands exhibiting mainly 5-HT1A binding affinities. Specifically we focused our attention in the synthesis of compounds structurally related to arylpiperazine, 2-aminotetralin, and benzopyran derivatives. Keywords: serotonin, 5-HT1A ligands, arylpiperazines, aminotetralins, benzopyrans. INTRODUCTION During the last 15 years, seven distinct families of 5-HT receptors have been identified (5-HT1–5-HT7), and at least Depression is one of the most common illnesses, 15 subpopulations have been described for several of these affecting up to one-third of all people at the same time. [4,5]. The 5-HT1A receptors represent a major target for Depressive disorders encompass a variety of conditions neurobiological research and drug developments. A study on including two major forms of unipolar depression (i.e. major distribution of 5-HT1A receptors in the brains of various depression and dysthymia), adjustment disorders, animal species indicates that the highest densities are located subsyndromal depression (minor depression), seasonal in the hippocampus, septum, amygdale, and cortical limbic affective disorder (SAD), premenstrual dysphoric disorder areas. The 5-HT1A receptors located in the raphe nuclei are (PMDD), postpartum depression, atypical depression and known as somatodendritic autoreceptors. -

Stems for Nonproprietary Drug Names
USAN STEM LIST STEM DEFINITION EXAMPLES -abine (see -arabine, -citabine) -ac anti-inflammatory agents (acetic acid derivatives) bromfenac dexpemedolac -acetam (see -racetam) -adol or analgesics (mixed opiate receptor agonists/ tazadolene -adol- antagonists) spiradolene levonantradol -adox antibacterials (quinoline dioxide derivatives) carbadox -afenone antiarrhythmics (propafenone derivatives) alprafenone diprafenonex -afil PDE5 inhibitors tadalafil -aj- antiarrhythmics (ajmaline derivatives) lorajmine -aldrate antacid aluminum salts magaldrate -algron alpha1 - and alpha2 - adrenoreceptor agonists dabuzalgron -alol combined alpha and beta blockers labetalol medroxalol -amidis antimyloidotics tafamidis -amivir (see -vir) -ampa ionotropic non-NMDA glutamate receptors (AMPA and/or KA receptors) subgroup: -ampanel antagonists becampanel -ampator modulators forampator -anib angiogenesis inhibitors pegaptanib cediranib 1 subgroup: -siranib siRNA bevasiranib -andr- androgens nandrolone -anserin serotonin 5-HT2 receptor antagonists altanserin tropanserin adatanserin -antel anthelmintics (undefined group) carbantel subgroup: -quantel 2-deoxoparaherquamide A derivatives derquantel -antrone antineoplastics; anthraquinone derivatives pixantrone -apsel P-selectin antagonists torapsel -arabine antineoplastics (arabinofuranosyl derivatives) fazarabine fludarabine aril-, -aril, -aril- antiviral (arildone derivatives) pleconaril arildone fosarilate -arit antirheumatics (lobenzarit type) lobenzarit clobuzarit -arol anticoagulants (dicumarol type) dicumarol -

The Use of Stems in the Selection of International Nonproprietary Names (INN) for Pharmaceutical Substances
WHO/PSM/QSM/2006.3 The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances 2006 Programme on International Nonproprietary Names (INN) Quality Assurance and Safety: Medicines Medicines Policy and Standards The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances FORMER DOCUMENT NUMBER: WHO/PHARM S/NOM 15 © World Health Organization 2006 All rights reserved. Publications of the World Health Organization can be obtained from WHO Press, World Health Organization, 20 Avenue Appia, 1211 Geneva 27, Switzerland (tel.: +41 22 791 3264; fax: +41 22 791 4857; e-mail: [email protected]). Requests for permission to reproduce or translate WHO publications – whether for sale or for noncommercial distribution – should be addressed to WHO Press, at the above address (fax: +41 22 791 4806; e-mail: [email protected]). The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of the World Health Organization concerning the legal status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. Dotted lines on maps represent approximate border lines for which there may not yet be full agreement. The mention of specific companies or of certain manufacturers’ products does not imply that they are endorsed or recommended by the World Health Organization in preference to others of a similar nature that are not mentioned. Errors and omissions excepted, the names of proprietary products are distinguished by initial capital letters. -

Pharmacokinetics and Related Topics
Pharmacokinetics and 11 related topics 11.1 The three phases of drug may be absorbed through the mucosal membranes of the mouth, but most passes down into the stomach where action it encounters gastric juices and hydrochloric acid. Th ese chemicals aid in the digestion of food and will treat a Th ere are three phases involved in drug action. Th e drug in a similar fashion if it is susceptible to breakdown fi rst of these is the pharmaceutical phase . For an orally and is not protected within an acid-resistant pill or cap- administered drug, this includes the disintegration of a sule. For example, the fi rst clinically useful penicillin was pill or capsule in the gastrointestinal tract (GIT), the broken down in the stomach and had to be administered release of the drug, and its dissolution. Th e pharmaceu- by injection. Other acid-labile drugs include the local tical phase is followed by the pharmacokinetic phase , anaesthetics and insulin . If the drug does survive the which includes absorption from the GIT into the blood stomach, it enters the upper intestine where it encoun- supply, and the various factors that aff ect a drug’s survival ters digestive enzymes that serve to break down food. and progress as it travels to its molecular target. Th e fi nal Assuming the drug survives this attack, it then has to pharmacodynamic phase involves the mechanism by pass through the cells lining the gut wall. Th is means that which a drug interacts with its molecular target and the it has to pass through a cell membrane on two occasions: resulting pharmacological eff ect. -

Pharmaceutical Appendix to the Tariff Schedule 2
Harmonized Tariff Schedule of the United States (2007) (Rev. 2) Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE Harmonized Tariff Schedule of the United States (2007) (Rev. 2) Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 2 Table 1. This table enumerates products described by International Non-proprietary Names (INN) which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service (CAS) registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. ABACAVIR 136470-78-5 ACIDUM LIDADRONICUM 63132-38-7 ABAFUNGIN 129639-79-8 ACIDUM SALCAPROZICUM 183990-46-7 ABAMECTIN 65195-55-3 ACIDUM SALCLOBUZICUM 387825-03-8 ABANOQUIL 90402-40-7 ACIFRAN 72420-38-3 ABAPERIDONUM 183849-43-6 ACIPIMOX 51037-30-0 ABARELIX 183552-38-7 ACITAZANOLAST 114607-46-4 ABATACEPTUM 332348-12-6 ACITEMATE 101197-99-3 ABCIXIMAB 143653-53-6 ACITRETIN 55079-83-9 ABECARNIL 111841-85-1 ACIVICIN 42228-92-2 ABETIMUSUM 167362-48-3 ACLANTATE 39633-62-0 ABIRATERONE 154229-19-3 ACLARUBICIN 57576-44-0 ABITESARTAN 137882-98-5 ACLATONIUM NAPADISILATE 55077-30-0 ABLUKAST 96566-25-5 ACODAZOLE 79152-85-5 ABRINEURINUM 178535-93-8 ACOLBIFENUM 182167-02-8 ABUNIDAZOLE 91017-58-2 ACONIAZIDE 13410-86-1 ACADESINE 2627-69-2 ACOTIAMIDUM 185106-16-5 ACAMPROSATE 77337-76-9 -
Downloaded from Survive Nursing | Survivenursing.Com V20110426
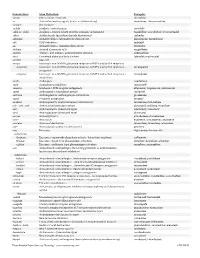
Generic Stem Stem Definition Examples -abine (see -arabine, -citabine) decitabine -ac Anti-inflammatory agents (acetic acid derivatives) bromfenac; dexpemedolac -acetam See -racetam -actide Synthetic corticotropins seractide -adol or -aldol- Analgesics (mixed opiate receptor agonists/ antagonists) tazadolene; spiradolene; levonantradol -adox Antibacterials (quinoline dioxide derivatives) carbadox -afenone Antiarrhythmics (propafenone derivatives) alprafenone; diprafenone -afil PDE5 inhibitors tadalafil -aj- Antiarrhythmics (ajmaline derivatives) lorajmine -aldrate Antacid aluminum salts magaldrate -algron Alpha1 - and alpha2 - adrenoreceptor agonists dabuzalgron -alol Combined alpha and beta blockers labetalol; medroxalol -amivir (see -vir) -ampa Ionotropic non-NMDA glutamate receptors (AMPA and/or KA receptors) -ampanel Ionotropic non-NMDA glutamate receptors (AMPA and/or KA receptors) ; becampanel antagonists -ampator Ionotropic non-NMDA glutamate receptors (AMPA and/or KA receptors) ; forampator modulators -andr- Androgens nandrolone -anib Angiogenesis inhibitors semaxanib -anserin Serotonin 5-HT2 receptor antagonists altanserin; tropanserin; adatanserin -antel Anthelmintics (undefined group) carbantel -antrone Antineoplastics; anthraquinone derivatives pixantrone -apsel P-selectin antagonists torapsel -arabine Antineoplastics (arabinofuranosyl derivatives) fazarabine; fludarabine aril-, -aril, -aril- Antiviral (arildone derivatives) pleconaril; arildone; fosarilate -arit Antirheumatics (lobenzarit type) lobenzarit; clobuzarit -arol -

(12) Patent Application Publication (10) Pub. No.: US 2015/0202317 A1 Rau Et Al
US 20150202317A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2015/0202317 A1 Rau et al. (43) Pub. Date: Jul. 23, 2015 (54) DIPEPTDE-BASED PRODRUG LINKERS Publication Classification FOR ALPHATIC AMNE-CONTAINING DRUGS (51) Int. Cl. A647/48 (2006.01) (71) Applicant: Ascendis Pharma A/S, Hellerup (DK) A638/26 (2006.01) A6M5/9 (2006.01) (72) Inventors: Harald Rau, Heidelberg (DE); Torben A 6LX3/553 (2006.01) Le?mann, Neustadt an der Weinstrasse (52) U.S. Cl. (DE) CPC ......... A61K 47/48338 (2013.01); A61 K3I/553 (2013.01); A61 K38/26 (2013.01); A61 K (21) Appl. No.: 14/674,928 47/48215 (2013.01); A61M 5/19 (2013.01) (22) Filed: Mar. 31, 2015 (57) ABSTRACT The present invention relates to a prodrug or a pharmaceuti Related U.S. Application Data cally acceptable salt thereof, comprising a drug linker conju (63) Continuation of application No. 13/574,092, filed on gate D-L, wherein D being a biologically active moiety con Oct. 15, 2012, filed as application No. PCT/EP2011/ taining an aliphatic amine group is conjugated to one or more 050821 on Jan. 21, 2011. polymeric carriers via dipeptide-containing linkers L. Such carrier-linked prodrugs achieve drug releases with therapeu (30) Foreign Application Priority Data tically useful half-lives. The invention also relates to pharma ceutical compositions comprising said prodrugs and their use Jan. 22, 2010 (EP) ................................ 10 151564.1 as medicaments. US 2015/0202317 A1 Jul. 23, 2015 DIPEPTDE-BASED PRODRUG LINKERS 0007 Alternatively, the drugs may be conjugated to a car FOR ALPHATIC AMNE-CONTAINING rier through permanent covalent bonds. -

Customs Tariff - Schedule
CUSTOMS TARIFF - SCHEDULE 99 - i Chapter 99 SPECIAL CLASSIFICATION PROVISIONS - COMMERCIAL Notes. 1. The provisions of this Chapter are not subject to the rule of specificity in General Interpretative Rule 3 (a). 2. Goods which may be classified under the provisions of Chapter 99, if also eligible for classification under the provisions of Chapter 98, shall be classified in Chapter 98. 3. Goods may be classified under a tariff item in this Chapter and be entitled to the Most-Favoured-Nation Tariff or a preferential tariff rate of customs duty under this Chapter that applies to those goods according to the tariff treatment applicable to their country of origin only after classification under a tariff item in Chapters 1 to 97 has been determined and the conditions of any Chapter 99 provision and any applicable regulations or orders in relation thereto have been met. 4. The words and expressions used in this Chapter have the same meaning as in Chapters 1 to 97. Issued January 1, 2019 99 - 1 CUSTOMS TARIFF - SCHEDULE Tariff Unit of MFN Applicable SS Description of Goods Item Meas. Tariff Preferential Tariffs 9901.00.00 Articles and materials for use in the manufacture or repair of the Free CCCT, LDCT, GPT, UST, following to be employed in commercial fishing or the commercial MT, MUST, CIAT, CT, harvesting of marine plants: CRT, IT, NT, SLT, PT, COLT, JT, PAT, HNT, Artificial bait; KRT, CEUT, UAT, CPTPT: Free Carapace measures; Cordage, fishing lines (including marlines), rope and twine, of a circumference not exceeding 38 mm; Devices for keeping nets open; Fish hooks; Fishing nets and netting; Jiggers; Line floats; Lobster traps; Lures; Marker buoys of any material excluding wood; Net floats; Scallop drag nets; Spat collectors and collector holders; Swivels. -

Providing for Duty-Free Treatment for Specified Pharmaceutical Active
13 . 3 . 97 I EN I Official Journal of the European Communities No L 71 / 1 I (Acts whose publication is obligatory) COUNCIL REGULATION (EC) No 467/97 of 3 March 1997 providing for duty-free treatment for specified pharmaceutical active ingredients bearing an 'international non-proprietary name' (INN) from the World Health Organization and specified products used for the manufacture of finished pharmaceuticals and withdrawing duty-free treatment as pharmaceutical products from certain INNs whose predominant use is not pharmaceutical THE COUNCIL OF THE EUROPEAN UNION , whereas in the context of the review it was concluded that it was appropriate to rectify the situation with regard Having regard to the Treaty establishing the European to certain INNs whose use was predominantly non Community, and in particular Article 113 thereof, pharmaceutical and which had been inadvertently included among those INNs already receiving duty-free treatment, Having regard to the proposal from the Commission , Whereas, in the course of the Uruguay Round negotia HAS ADOPTED THIS REGULATION : tions the Community and a number of countries discussed duty-free treatment of pharmaceutical products ; Article 1 Whereas the participants in those discussions concluded that in addition to products falling within the Harm From 1 April 1997 the Community shall also accord onized System (HS) Chapter 30 and HS headings 2936 , duty-free treatment for the INNs listed in Annex I as well 2937, 2939 and 2941 , duty-free treatment should be given as the salts, esters and hydrates of such products . to designated pharmaceutical active ingredients bearing an 'international non-proprietary name' (INN) from the Article 2 World Health Organization as well as specified salts , esters and hydrates of such INNs, and also to designated From 1 April 1997 the Community shall also grant duty products used for the production and manufacture of free treatment for the products used in the production finished products ; and manufacture of pharmaceutical products listed in Annex II . -

(12) United States Patent (10) Patent No.: US 6,197,764 B1 Bradley Et Al
USOO6197764B1 (12) United States Patent (10) Patent No.: US 6,197,764 B1 Bradley et al. (45) Date of Patent: *Mar. 6, 2001 (54) CLOZAPINE COMPOSITIONS AND USES FOREIGN PATENT DOCUMENTS THEREOF 0599 576A1 1/1994 (EP). (75) Inventors: Matthews O. Bradley, Laytonsville, 693498 1/1996 (EP). MD (US); Victor E. Shashoua, 61204136 11/1984 (JP). Belmont, MA (US); Charles S. 06-072868 3/1994 (JP). Swindell, Merion; Nigel L. Webb, 6072868 3/1994 (JP). Bryn Mawr, both of PA (US) 7082146 3/1996 (JP). 8151334 6/1996 (JP). (73) Assignee: Protarga, Inc., Conshohocken, PA (US) 9030963 2/1997 (JP). (*) Notice: Subject to any disclaimer, the term of this WO 89/07938 9/1989 (WO). patent is extended or adjusted under 35 WO 96/04001 2/1996 (WO). U.S.C. 154(b) by 0 days. WO 96/22303 7/1996 (WO). WO 96/27380 9/1996 (WO). This patent is Subject to a terminal dis WO98/17325 4/1998 (WO). claimer. OTHER PUBLICATIONS (21) Appl. No.: 08/978,541 (22) Filed: Nov. 26, 1997 Bourat, et al., "Long Chain Esters of Pipotiazine as Lon g-Acting Psychotropic Pro-Drug, Med. Chem. Proc. Int. (51) Int. Cl. .............................................. A61K 31/00 Symp. 5th (1976) pp. 105-114. (52) U.S. Cl. ........................... 514/218; 514/219; 514/220 Lohr, et al., “Neuroleptic-Induced Movement Disorders . (58) Field of Search ..................................... 514/218, 219, ..", Psychiatry, vol. 3, (1989). 514/220 Makino, et al., Chemical Abstracts, vol. 106, No. 12, (56) References Cited (90.177x) issued Mar. 23, 1987, “Pharmaceuticals Permeable to Blood-Brain Barrier'.