Antipatharia and Alcyonacea) on Scleractinian Cold-Water Coral
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Octocoral Diversity of Balıkçı Island, the Marmara Sea
J. Black Sea/Mediterranean Environment Vol. 19, No. 1: 46-57 (2013) RESEARCH ARTICLE Octocoral diversity of Balıkçı Island, the Marmara Sea Eda Nur Topçu1,2*, Bayram Öztürk1,2 1Faculty of Fisheries, Istanbul University, Ordu St., No. 200, 34470, Laleli, Istanbul, TURKEY 2Turkish Marine Research Foundation (TUDAV), P.O. Box: 10, Beykoz, Istanbul, TURKEY *Corresponding author: [email protected] Abstract We investigated the octocoral diversity of Balıkçı Island in the Marmara Sea. Three sites were sampled by diving, from 20 to 45 m deep. Nine species were found, two of which are first records for Turkish fauna: Alcyonium coralloides and Paralcyonium spinulosum. Scientific identification of Alcyonium acaule in the Turkish seas was also done for the first time in this study. Key words: Octocoral, soft coral, gorgonian, diversity, Marmara Sea. Introduction The Marmara Sea is a semi-enclosed sea connecting the Black Sea to the Aegean Sea via the Turkish Straits System, with peculiar oceanographic, ecological and geomorphologic characteristics (Öztürk and Öztürk 1996). The benthic fauna consists of Black Sea species until approximately 20 meters around the Prince Islands area, where Mediterranean species take over due to the two layer stratification in the Marmara Sea. The Sea of Marmara, together with the straits of Istanbul and Çanakkale, serves as an ecological barrier, a biological corridor and an acclimatization zone for the biota of Mediterranean and Black Seas (Öztürk and Öztürk 1996). The Islands in the Sea of Marmara constitute habitats particularly for hard bottom communities of Mediterranean origin. Ten Octocoral species were reported by Demir (1954) from the Marmara Sea but amongst them, Gorgonia flabellum was probably reported by mistake and 46 should not be considered as a valid record from the Sea of Marmara. -

Di Camillo Et Al 2017
This is a post-peer-review, pre-copyedit version of an article published in Biodiversity and Conservation on 23 December 2017 (First Online). The final authenticated version is available online at: https://doi.org/10.1007/s10531-017-1492-8 https://link.springer.com/article/10.1007%2Fs10531-017-1492-8 An embargo period of 12 months applies to this Journal. This paper has received funding from the European Union (EU)’s H2020 research and innovation programme under the Marie Sklodowska-Curie grant agreement No 643712 to the project Green Bubbles RISE for sustainable diving (Green Bubbles). This paper reflects only the authors’ view. The Research Executive Agency is not responsible for any use that may be made of the information it contains. © 2017. This manuscript version is made available under the CC-BY-NC-ND 4.0 AUTHORS' ACCEPTED MANUSCRIPT Building a baseline for habitat-forming corals by a multi-source approach, including Web Ecological Knowledge - Cristina G Di Camillo, Department of Life and Environmental Sciences, Marche Polytechnic University, CoNISMa, Ancona, Italy, [email protected] - Massimo Ponti, Department of Biological, Geological and Environmental Sciences and Interdepartmental Research Centre for Environmental SciencesUniversity of Bologna, CoNISMa, Ravenna, Italy - Giorgio Bavestrello, Department of Earth, Environment and Life Sciences, University of Genoa, CoNISMa, Genoa, Italy - Maja Krzelj, Department of Marine Studies, University of Split, Split, Croatia - Carlo Cerrano, Department of Life and Environmental Sciences, Marche Polytechnic University, CoNISMa, Ancona, Italy Received: 12 January 2017 Revised: 10 December 2017 Accepted: 14 December 2017 First online: 23 December 2017 Cite as: Di Camillo, C.G., Ponti, M., Bavestrello, G. -

THE LARVAL STAGES of HOMOLA BARBATA (FABRICIUS) (CRUSTACEA, DECAPODA, HOMOLIDAE) REARED in the Laboratoryl
BIOLOGICAL RESULTS OF THE UNIVERSITY OF MIAMI DEEP-SEA EXPEDITIONS. 55. THE LARVAL STAGES OF HOMOLA BARBATA (FABRICIUS) (CRUSTACEA, DECAPODA, HOMOLIDAE) REARED IN THE LABORATORyl A. L. RICE British Museum (Natural History), Cromwell Road, London, S.W. 7, England AND ANTHONY J. PROVENZANO, JR. University of Miami, Rosenstiel School of Marine and Atmospheric Sciences ABSTRACT Larvae yielded by a female collected off Yucatan were reared to the seventh, and final, zoeal stage at 20°C and 36.2%0 salinity. The larvae, the first complete series available of any species of the family, are described and illustrated. Comparison of the reared larvae with others attributed to the same species but captured in Mediterranean and South African waters confirmed the morphological differences which previously had led to sug- gestions of specific or subspecific separation of the parent populations. Examination of adult material failed to reveal positive evidence of specific status of the separated populations. The setation of the uropodal endopods in Homola appears in the third zoeal stage, earlier than for any other decapod known. Recent studies of larval forms tend to confirm the view that the Homolidae are related to the Raninidae and are close to the line of descent of the true crabs, and that the family is well separated from both the typical Anomura and the much more primitive Dromiidea. INTRODUCTION Homola barbata has been recorded in the eastern and western North Atlantic, in the Mediterranean, and in the South Atlantic near South Africa. Throughout its range (Fig. 1), H. barbata is the only recognized repre- sentative of the genus, except off the east coast of North America where a closely related species, H. -

Microbiomes of Gall-Inducing Copepod Crustaceans from the Corals Stylophora Pistillata (Scleractinia) and Gorgonia Ventalina
www.nature.com/scientificreports OPEN Microbiomes of gall-inducing copepod crustaceans from the corals Stylophora pistillata Received: 26 February 2018 Accepted: 18 July 2018 (Scleractinia) and Gorgonia Published: xx xx xxxx ventalina (Alcyonacea) Pavel V. Shelyakin1,2, Sofya K. Garushyants1,3, Mikhail A. Nikitin4, Sofya V. Mudrova5, Michael Berumen 5, Arjen G. C. L. Speksnijder6, Bert W. Hoeksema6, Diego Fontaneto7, Mikhail S. Gelfand1,3,4,8 & Viatcheslav N. Ivanenko 6,9 Corals harbor complex and diverse microbial communities that strongly impact host ftness and resistance to diseases, but these microbes themselves can be infuenced by stresses, like those caused by the presence of macroscopic symbionts. In addition to directly infuencing the host, symbionts may transmit pathogenic microbial communities. We analyzed two coral gall-forming copepod systems by using 16S rRNA gene metagenomic sequencing: (1) the sea fan Gorgonia ventalina with copepods of the genus Sphaerippe from the Caribbean and (2) the scleractinian coral Stylophora pistillata with copepods of the genus Spaniomolgus from the Saudi Arabian part of the Red Sea. We show that bacterial communities in these two systems were substantially diferent with Actinobacteria, Alphaproteobacteria, and Betaproteobacteria more prevalent in samples from Gorgonia ventalina, and Gammaproteobacteria in Stylophora pistillata. In Stylophora pistillata, normal coral microbiomes were enriched with the common coral symbiont Endozoicomonas and some unclassifed bacteria, while copepod and gall-tissue microbiomes were highly enriched with the family ME2 (Oceanospirillales) or Rhodobacteraceae. In Gorgonia ventalina, no bacterial group had signifcantly diferent prevalence in the normal coral tissues, copepods, and injured tissues. The total microbiome composition of polyps injured by copepods was diferent. -

Long-Term Recruitment of Soft-Corals (Octocorallia: Alcyonacea) on Artificial Substrata at Eilat (Red Sea)
MARINE ECOLOGY - PROGRESS SERIES Vol. 38: 161-167, 1987 Published June 18 Mar. Ecol. Prog. Ser. Long-term recruitment of soft-corals (Octocorallia: Alcyonacea) on artificial substrata at Eilat (Red Sea) Y.Benayahu & Y.Loya Department of Zoology. The George S. Wise Center for Life Sciences, Tel Aviv University, Tel Aviv 69978. Israel ABSTRACT: Recruitment of soft corals (Octocorallia: Alcyonacea) on concrete plates was studied in the reefs of the Nature Reserve of Eilat at depths of 17 to 29 m over 12 yr. Xenia macrospiculata was the pioneering species, appealing on the vast majority of the plates before any other spat. This species remained the most conspicuous inhabitant of the substrata throughout the whole study. Approximately 10 % of the plates were very extensively colonized by X. rnacrospiculata, resembling the percentage of living coverage by the species in the surrounding reef, thus suggesting that during the study X. rnacrospiculata populations reached their maximal potential to capture the newly available substrata. The successive appearance of an additional 11 soft coral species was recorded. The species composition of the recruits and their abundance corresponded with the soft coral community in the natural reef, indicahng that the estabhshed spat were progeny of the local populations. Soft coral recruits utilized the edges and lower surfaces of the plates most successfully, rather than the exposed upper surfaces. Such preferential settling of alcyonaceans allows the spat to escape from unfavourable conditions and maintains their high survival in the established community. INTRODUCTION determine the role played by alcyonaceans in the course of reef colonization and in the reef's space Studies on processes and dynamics of reef benthic allocation. -

Coral Feeding on Microalgae Assessed with Molecular Trophic Markers
Molecular Ecology (2013) doi: 10.1111/mec.12486 Coral feeding on microalgae assessed with molecular trophic markers MIGUEL C. LEAL,*† CHRISTINE FERRIER-PAGES,‡ RICARDO CALADO,* MEGAN E. THOMPSON,† MARC E. FRISCHER† and JENS C. NEJSTGAARD† *Departamento de Biologia & CESAM, Universidade de Aveiro, Campus Universitario de Santiago, 3810-193 Aveiro, Portugal, †Skidaway Institute of Oceanography, 10 Ocean Science Circle, 31411 Savannah, GA, USA, ‡Centre Scientifique de Monaco, Avenue St-Martin, 98000 Monaco, Monaco Abstract Herbivory in corals, especially for symbiotic species, remains controversial. To investi- gate the capacity of scleractinian and soft corals to capture microalgae, we conducted controlled laboratory experiments offering five algal species: the cryptophyte Rhodo- monas marina, the haptophytes Isochrysis galbana and Phaeocystis globosa, and the diatoms Conticribra weissflogii and Thalassiosira pseudonana. Coral species included the symbiotic soft corals Heteroxenia fuscescens and Sinularia flexibilis, the asymbiotic scleractinian coral Tubastrea coccinea, and the symbiotic scleractinian corals Stylophora pistillata, Pavona cactus and Oculina arbuscula. Herbivory was assessed by end-point PCR amplification of algae-specific 18S rRNA gene fragments purified from coral tissue genomic DNA extracts. The ability to capture microalgae varied with coral and algal species and could not be explained by prey size or taxonomy. Herbivory was not detected in S. flexibilis and S. pistillata. P. globosa was the only algal prey that was never captured by any coral. Although predation defence mechanisms have been shown for Phaeocystis spp. against many potential predators, this study is the first to suggest this for corals. This study provides new insights into herbivory in symbiotic corals and suggests that corals may be selective herbivorous feeders. -

Ecological Mapping and Data Quality Assessment for the Needs of Ecosystem-Based Marine Spatial Management: Case Study Greek Ionian Sea and the Adjacent Gulfs
Research Article Mediterranean Marine Science Indexed in WoS (Web of Science, ISI Thomson) and SCOPUS The journal is available on line at http://www.medit-mar-sc.net Ecological mapping and data quality assessment for the needs of ecosystem-based marine spatial management: case study Greek Ionian Sea and the adjacent gulfs Y. ISSARIS1,2, S. KATSANEVAKIS1,3, M. PANTAZI1, V. VASSILOPOULOU1, P. PANAYOTIDIS4, S. KAVADAS1, A. KOKKALI1, M. SALOMIDI4, A. FRANTZIS5, A. PANOU6, D. DAMALAS1,7, D. KLAOUDATOS1, D. SAKELLARIOU4, V. DRAKOPOULOU4, C. KYRIAKIDOU4, I. MAINA1, J. FRIC8, C. SMITH1, S. GIAKOUMI1 and G. KARRIS9 1 Institute of Marine Biological Resources, Hellenic Centre for Marine Research, Ag. Kosmas, Greece 2 Sector of Zoology and Marine Biology, Department of Biology, University of Athens, Zografos, Greece 3 European Commission, Joint Research Centre, Institute for Environment and Sustainability, Ispra, Italy 4 Institute of Oceanography, Hellenic Centre for Marine Research, Anavyssos, Greece 5 Pelagos Cetacean Research Institute, Vouliagmeni, Greece 6 Archipelagos – environment and development, Kifissia, Greece 7 European Commission, Joint Research Centre, Institute for the Protection and Security of the Citizen, Ispra, Italy 8 Hellenic Ornithological Society, Athens, Greece 9 Department of Environmental Technology and Ecology, TEI of the Ionian Islands, Zakynthos Island, Greece Corresponding author: [email protected] Received: 30 April 2012; Accepted: 2 November 2012; Published on line: 19 November 2012 Abstract Mapping of ecosystem components (natural and socioeconomic) is a prerequisite for ecosystem-based marine spatial manage- ment (EB-MSM). To initiate the process of EB-MSM in the Greek Ionian Sea and the adjacent gulfs, the main relevant ecosystem components were mapped based on existing spatial information and expert judgment. -

Updated Classification of Benthic Marine Habitat Types for The
Updated Classification of Benthic Marine Habitat Types for the Mediterranean Region TUNISIA - 2019 Legal notice: The designations employed and the presentation of the material in this document do not imply the expression of any opinion whatsoever on the part of the Specially Protected Areas Regional Activity Centre (SPA/RAC) and UN Environment/Mediterranean Action Plan (MAP) and those of the Lebanese Ministry of Environment concerning the legal status of any State, Territory, city or area, or of its authorities, or concerning the delimitation of their frontiers or boundaries. This publication was produced with the fi nancial support of the European Union in the framework of the MedMPA Network Project. Its contents are the sole responsibility of SPA/RAC and do not necessarily refl ect the views of the European Union. Copyright : All property rights of texts and content of different types of this publication belong to SPA/RAC. Reproduction of these texts and contents, in whole or in part, and in any form, is prohibited without prior written permission from SPA/RAC, except for educational and other non-commercial purposes, provided that the source is fully acknowledged. © 2019 - United Nations Environment Programme Mediterranean Action Plan Specially Protected Areas Regional Activity Centre (SPA/RAC) Boulevard du Leader Yasser Arafat B.P. 337 1080 Tunis Cedex - Tunisia. [email protected] For bibliographic purposes, this document may be cited as: SPA/RAC–UN Environment/MAP, 2019: Updated Classifi cation of Benthic Marine Habitat Types for the Mediterranean Region Layout : Atef OUERGHI Cover photos credit: © SPA/RAC, University of Seville, University of Alicante, Trainito E. -
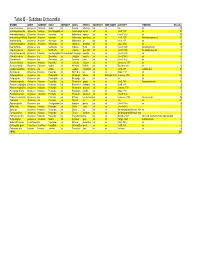
Table B – Subclass Octocorallia
Table B – Subclass Octocorallia BINOMEN ORDER SUBORDER FAMILY SUBFAMILY GENUS SPECIES SUBSPECIES COMN_NAMES AUTHORITY SYNONYMS #Records Acanella arbuscula Alcyonacea Calcaxonia Isididae n/a Acanella arbuscula n/a n/a n/a n/a 59 Acanthogorgia armata Alcyonacea Holaxonia Acanthogorgiidae n/a Acanthogorgia armata n/a n/a Verrill, 1878 n/a 95 Anthomastus agassizii Alcyonacea Alcyoniina Alcyoniidae n/a Anthomastus agassizii n/a n/a (Verrill, 1922) n/a 35 Anthomastus grandiflorus Alcyonacea Alcyoniina Alcyoniidae n/a Anthomastus grandiflorus n/a n/a Verrill, 1878 Anthomastus purpureus 37 Anthomastus sp. Alcyonacea Alcyoniina Alcyoniidae n/a Anthomastus sp. n/a n/a Verrill, 1878 n/a 1 Anthothela grandiflora Alcyonacea Scleraxonia Anthothelidae n/a Anthothela grandiflora n/a n/a (Sars, 1856) n/a 24 Capnella florida Alcyonacea n/a Nephtheidae n/a Capnella florida n/a n/a (Verrill, 1869) Eunephthya florida 44 Capnella glomerata Alcyonacea n/a Nephtheidae n/a Capnella glomerata n/a n/a (Verrill, 1869) Eunephthya glomerata 4 Chrysogorgia agassizii Alcyonacea Holaxonia Acanthogorgiidae Chrysogorgiidae Chrysogorgia agassizii n/a n/a (Verrill, 1883) n/a 2 Clavularia modesta Alcyonacea n/a Clavulariidae n/a Clavularia modesta n/a n/a (Verrill, 1987) n/a 6 Clavularia rudis Alcyonacea n/a Clavulariidae n/a Clavularia rudis n/a n/a (Verrill, 1922) n/a 1 Gersemia fruticosa Alcyonacea Alcyoniina Alcyoniidae n/a Gersemia fruticosa n/a n/a Marenzeller, 1877 n/a 3 Keratoisis flexibilis Alcyonacea Calcaxonia Isididae n/a Keratoisis flexibilis n/a n/a Pourtales, 1868 n/a 1 Lepidisis caryophyllia Alcyonacea n/a Isididae n/a Lepidisis caryophyllia n/a n/a Verrill, 1883 Lepidisis vitrea 13 Muriceides sp. -

Author's Accepted Manuscript
Author’s Accepted Manuscript This is the accepted version of the following article: Rakka, M., Bilan, M., Godinho, A., Movilla, J., Orejas, C., & Carreiro-Silva, M. (2019). First description of polyp bailout in cold-water octocorals under aquaria maintenance. Coral Reefs, 38(1), 15-20, which has been published in final form at https://doi.org/10.1007/s00338-018-01760- x. This article may be used for non-commercial purposes in accordance with Springer Terms and Conditions for Use of Self-Archived Versions 1 First description of polyp bail-out in cold-water octocorals under aquaria maintenance Maria Rakka1,2,3, Meri Bilan1,2,3, Antonio Godinho1,2,3,Juancho Movilla4,5, Covadonga Orejas4, Marina Carreiro-Silva1,2,3 1 MARE – Marine and Environmental Sciences Centre and Centre of the Institute of Marine Research 2 IMAR – University of the Azores, Rua Frederico Machado 4, 9901-862 Horta, Portugal 3 OKEANOS Research Unit, Faculty of Science and Technology, University of the Azores, 9901-862, Horta, Portugal 4 Instituto Español de Oceanografía, Centro Oceanográfico de Baleares, Moll de Ponent s/n, 07015 Palma, Spain 5 Instituto de Ciencias del Mar (ICM-CSIC). Passeig Maritim de la Barceloneta 37-49, 08003 Barcelona, Spain Corresponding author: Maria Rakka, [email protected], +351915407062 2 Abstract Cnidarians, characterized by high levels of plasticity, exhibit remarkable mechanisms to withstand or escape unfavourable conditions including reverse development which describes processes of transformation of adult stages into early developmental stages with higher mobility. Polyp bail-out is a stress-escape response common among scleractinian species, consisting of massive detachment of live polyps and subsequent death of the mother colony. -

Guide to the Identification of Precious and Semi-Precious Corals in Commercial Trade
'l'llA FFIC YvALE ,.._,..---...- guide to the identification of precious and semi-precious corals in commercial trade Ernest W.T. Cooper, Susan J. Torntore, Angela S.M. Leung, Tanya Shadbolt and Carolyn Dawe September 2011 © 2011 World Wildlife Fund and TRAFFIC. All rights reserved. ISBN 978-0-9693730-3-2 Reproduction and distribution for resale by any means photographic or mechanical, including photocopying, recording, taping or information storage and retrieval systems of any parts of this book, illustrations or texts is prohibited without prior written consent from World Wildlife Fund (WWF). Reproduction for CITES enforcement or educational and other non-commercial purposes by CITES Authorities and the CITES Secretariat is authorized without prior written permission, provided the source is fully acknowledged. Any reproduction, in full or in part, of this publication must credit WWF and TRAFFIC North America. The views of the authors expressed in this publication do not necessarily reflect those of the TRAFFIC network, WWF, or the International Union for Conservation of Nature (IUCN). The designation of geographical entities in this publication and the presentation of the material do not imply the expression of any opinion whatsoever on the part of WWF, TRAFFIC, or IUCN concerning the legal status of any country, territory, or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries. The TRAFFIC symbol copyright and Registered Trademark ownership are held by WWF. TRAFFIC is a joint program of WWF and IUCN. Suggested citation: Cooper, E.W.T., Torntore, S.J., Leung, A.S.M, Shadbolt, T. and Dawe, C. -

Composition and Abundance of Octocorals in the Sea of Marmara, Where the Mediterranean Meets the Black Sea
SCIENTIA MARINA 79(1) March 2015, 125-135, Barcelona (Spain) ISSN-L: 0214-8358 doi: http://dx.doi.org/10.3989/scimar.04120.09A Composition and abundance of octocorals in the Sea of Marmara, where the Mediterranean meets the Black Sea Eda N. Topçu 1,2, Bayram Öztürk 1,2 1 Istanbul University Fisheries Faculty, Ordu cad No 200 Laleli, 34470 Istanbul, Turkey. E-mail: [email protected] 2 Turkish Marine Research Foundation, No 10, Beykoz, 81650, Istanbul, Turkey. Summary: Species composition and abundance of octocoral assemblages were investigated in the Sea of Marmara, which forms the connection between the Mediterranean and the Black Seas, two semi-enclosed seas with peculiar oceanographic conditions. Fourteen octocoral species were collected in the saline layer of the Marmara Sea (20-40 m), with a mean coral abundance of 5.21±5.11 colonies m–2 (mean ± SD) calculated from a total of 1390 colonies counted in transects. In spite of severe anthropogenic disturbances, dense assemblages of corals/gorgonians were observed during this study. The coral- ligenous communities—one of the most valuable structures of the Mediterranean Sea—harbored either Eunicella cavolini or Paramuricea macrospina as the dominant gorgonian in the Marmara Sea. Furthermore, the gorgonian assemblages of the Marmara Sea differed from those of the Mediterranean in their high abundance of P. macrospina and Spinimuricea klavereni, two species rarely encountered in the Mediterranean Sea at the studied depth range. The factors behind the observed differ- ences are discussed in regard to the particular oceanographic conditions of the Marmara Sea. Finally, we revised the main threats to corals/gorgonians in the Marmara Sea and provided some insights on management recommendations for coral conservation in this area.