Spironolactone 25 Mg
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Azelaic Acid
Azelaic Acid (FINACEA) Topical Foam 15% National Drug Monograph August 2016 VA Pharmacy Benefits Management Services, Medical Advisory Panel, and VISN Pharmacist Executives The purpose of VA PBM Services drug monographs is to provide a focused drug review for making formulary decisions. Updates will be made when new clinical data warrant additional formulary discussion. Documents will be placed in the Archive section when the information is deemed to be no longer current. FDA Approval Information Description/Mechanism of Azelaic acid is a naturally occurring C9-dicarboxylic acid that is found in plants Action (such as whole grain cereals), animals and humans. Azelaic acid has antiinflammatory, antioxidative and antikeratinizing effects. In rosacea skin, azelaic acid decreases cathelicidin levels and kallikrein 5 (KLK5) activity and possibly inhibits toll-like receptor 2 (TLR2) expression.1 A 15% gel formulation has been marketed for rosacea, and 20% cream has been available for acne vulgaris. The newer foam formulation consists of an oil- in-water emulsion and was designed to have a higher lipid content than the gel for dry and sensitive skin. Indication(s) Under Review Topical treatment of inflammatory papules and pustules of mild to moderate in This Document rosacea. Dosage Form(s) Under Foam, 15% Review REMS REMS No REMS Postmarketing Requirements See Other Considerations for additional REMS information Pregnancy Rating Category B Executive Summary Efficacy There have been no head-to-head trials comparing the foam and gel formulations of azelaic acid in terms of safety, tolerability and efficacy in the treatment of papulopustular (PP) rosacea.. In two major randomized clinical trials, azelaic acid foam produced small benefits over vehicle foam in achieving Investigator’s Global Assessment (IGA) treatment success (NNTs of 9.2 and 11.5) and in reducing inflammatory lesion counts. -

Compositions Comprising Drospirenone Molecularly
(19) & (11) EP 1 748 756 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: A61K 9/00 (2006.01) A61K 9/107 (2006.01) 29.04.2009 Bulletin 2009/18 A61K 9/48 (2006.01) A61K 9/16 (2006.01) A61K 9/20 (2006.01) A61K 9/14 (2006.01) (2006.01) (2006.01) (21) Application number: 05708751.2 A61K 9/70 A61K 31/565 (22) Date of filing: 10.03.2005 (86) International application number: PCT/IB2005/000665 (87) International publication number: WO 2005/087194 (22.09.2005 Gazette 2005/38) (54) COMPOSITIONS COMPRISING DROSPIRENONE MOLECULARLY DISPERSED ZUSAMMENSETZUNGEN AUS DROSPIRENON IN MOLEKULARER DISPERSION DES COMPOSITIONS CONTENANT DROSPIRENONE A DISPERSION MOLECULAIRE (84) Designated Contracting States: (72) Inventors: AT BE BG CH CY CZ DE DK EE ES FI FR GB GR • FUNKE, Adrian HU IE IS IT LI LT LU MC NL PL PT RO SE SI SK TR D-14055 Berlin (DE) Designated Extension States: • WAGNER, Torsten AL BA HR MK YU 13127 Berlin (DE) (30) Priority: 10.03.2004 EP 04075713 (74) Representative: Wagner, Kim 10.03.2004 US 551355 P Plougmann & Vingtoft a/s Sundkrogsgade 9 (43) Date of publication of application: P.O. Box 831 07.02.2007 Bulletin 2007/06 2100 Copenhagen Ø (DK) (60) Divisional application: (56) References cited: 08011577.7 / 1 980 242 EP-A- 1 260 225 EP-A- 1 380 301 WO-A-01/52857 WO-A-20/04022065 (73) Proprietor: Bayer Schering Pharma WO-A-20/04041289 US-A- 5 569 652 Aktiengesellschaft US-A- 5 656 622 US-A- 5 789 442 13353 Berlin (DE) Note: Within nine months of the publication of the mention of the grant of the European patent in the European Patent Bulletin, any person may give notice to the European Patent Office of opposition to that patent, in accordance with the Implementing Regulations. -

Skin - Sulfa*Derm Sulfaderm Or Sulfa*Derm - Helps Promote Healthy Skin Function
Skin - Sulfa*Derm Sulfaderm or Sulfa*Derm - Helps promote Healthy skin Function. Has been shown to help clear up most Acne in 24 hours, Bacterial infections, Rashes, Bed Sores, Dermatitis, Eczema, Fungus/Yeast, Psoriasis type problems, Ring Worm and Wounds that won't heal. HISTORY OF SULFUR Harnessing the power of volcanos, our cutting edge formula is considered a breakthrough for the treatment of acne. Sulfa*Derm has been shown to clear up acne in one day. The active ingredient sulfur from volcanic ash destroys bacteria quickly. The Zinc Oxide combats rashes. Tea Tree Oil is a well known antiseptic and anti-fungal, while the Aloe Vera re-nourishes the skin. Vitamin E is used as the base instead of oils that clog up the skin. Sulfa*Derm is probably the best acne and everything else cream that has ever been made. When treating skin problems it is recommended to detoxify your liver. Sulfur is a yellow mineral that quite often occurs in nature in powder form. It can, under pressure bond to water, as in mineral springs or mix into the earth, as in mud baths. Normally sulfur is expunged from under the ground through the craters of venting volcanoes, where it is spread around in chunky blocks. Sulfur has been around for a long time. Native Polynesians claim to have cured a variety of infections in their hot sulfur mineral springs. Russian mud treatments, high in sulfur, have been used for centuries as a reputed therapy for arthritis. California's Napa Valley, specifically the city called Sulfur Springs, was the health spa for the wealthy until the early 1900's. -

Specifications of Approved Drug Compound Library
Annexure-I : Specifications of Approved drug compound library The compounds should be structurally diverse, medicinally active, and cell permeable Compounds should have rich documentation with structure, Target, Activity and IC50 should be known Compounds which are supplied should have been validated by NMR and HPLC to ensure high purity Each compound should be supplied as 10mM solution in DMSO and at least 100µl of each compound should be supplied. Compounds should be supplied in screw capped vial arranged as 96 well plate format. -

COVID-19—The Potential Beneficial Therapeutic Effects of Spironolactone During SARS-Cov-2 Infection
pharmaceuticals Review COVID-19—The Potential Beneficial Therapeutic Effects of Spironolactone during SARS-CoV-2 Infection Katarzyna Kotfis 1,* , Kacper Lechowicz 1 , Sylwester Drozd˙ zal˙ 2 , Paulina Nied´zwiedzka-Rystwej 3 , Tomasz K. Wojdacz 4, Ewelina Grywalska 5 , Jowita Biernawska 6, Magda Wi´sniewska 7 and Miłosz Parczewski 8 1 Department of Anesthesiology, Intensive Therapy and Acute Intoxications, Pomeranian Medical University in Szczecin, 70-111 Szczecin, Poland; [email protected] 2 Department of Pharmacokinetics and Monitored Therapy, Pomeranian Medical University, 70-111 Szczecin, Poland; [email protected] 3 Institute of Biology, University of Szczecin, 71-412 Szczecin, Poland; [email protected] 4 Independent Clinical Epigenetics Laboratory, Pomeranian Medical University, 71-252 Szczecin, Poland; [email protected] 5 Department of Clinical Immunology and Immunotherapy, Medical University of Lublin, 20-093 Lublin, Poland; [email protected] 6 Department of Anesthesiology and Intensive Therapy, Pomeranian Medical University in Szczecin, 71-252 Szczecin, Poland; [email protected] 7 Clinical Department of Nephrology, Transplantology and Internal Medicine, Pomeranian Medical University, 70-111 Szczecin, Poland; [email protected] 8 Department of Infectious, Tropical Diseases and Immune Deficiency, Pomeranian Medical University in Szczecin, 71-455 Szczecin, Poland; [email protected] * Correspondence: katarzyna.kotfi[email protected]; Tel.: +48-91-466-11-44 Abstract: In March 2020, coronavirus disease 2019 (COVID-19) caused by SARS-CoV-2 was declared Citation: Kotfis, K.; Lechowicz, K.; a global pandemic by the World Health Organization (WHO). The clinical course of the disease is Drozd˙ zal,˙ S.; Nied´zwiedzka-Rystwej, unpredictable but may lead to severe acute respiratory infection (SARI) and pneumonia leading to P.; Wojdacz, T.K.; Grywalska, E.; acute respiratory distress syndrome (ARDS). -

Hormonal Treatment Strategies Tailored to Non-Binary Transgender Individuals
Journal of Clinical Medicine Review Hormonal Treatment Strategies Tailored to Non-Binary Transgender Individuals Carlotta Cocchetti 1, Jiska Ristori 1, Alessia Romani 1, Mario Maggi 2 and Alessandra Daphne Fisher 1,* 1 Andrology, Women’s Endocrinology and Gender Incongruence Unit, Florence University Hospital, 50139 Florence, Italy; [email protected] (C.C); jiska.ristori@unifi.it (J.R.); [email protected] (A.R.) 2 Department of Experimental, Clinical and Biomedical Sciences, Careggi University Hospital, 50139 Florence, Italy; [email protected]fi.it * Correspondence: fi[email protected] Received: 16 April 2020; Accepted: 18 May 2020; Published: 26 May 2020 Abstract: Introduction: To date no standardized hormonal treatment protocols for non-binary transgender individuals have been described in the literature and there is a lack of data regarding their efficacy and safety. Objectives: To suggest possible treatment strategies for non-binary transgender individuals with non-standardized requests and to emphasize the importance of a personalized clinical approach. Methods: A narrative review of pertinent literature on gender-affirming hormonal treatment in transgender persons was performed using PubMed. Results: New hormonal treatment regimens outside those reported in current guidelines should be considered for non-binary transgender individuals, in order to improve psychological well-being and quality of life. In the present review we suggested the use of hormonal and non-hormonal compounds, which—based on their mechanism of action—could be used in these cases depending on clients’ requests. Conclusion: Requests for an individualized hormonal treatment in non-binary transgender individuals represent a future challenge for professionals managing transgender health care. For each case, clinicians should balance the benefits and risks of a personalized non-standardized treatment, actively involving the person in decisions regarding hormonal treatment. -

Enhanced Catalytic Ozonation of Ibuprofen Using a 3D Structured
www.nature.com/scientificreports OPEN Enhanced catalytic ozonation of ibuprofen using a 3D structured catalyst with MnO2 nanosheets on carbon microfbers Guhankumar Ponnusamy1, Hajar Farzaneh2, Yongfeng Tong1, Jenny Lawler1, Zhaoyang Liu1* & Jayaprakash Saththasivam1* Heterogeneous catalytic ozonation is an efective approach to degrade refractory organic pollutants in water. However, ozonation catalysts with combined merits of high activity, good reusability and low cost for practical industrial applications are still rare. This study aims to develop an efcient, stable and economic ozonation catalyst for the degradation of Ibuprofen, a pharmaceutical compound frequently detected as a refractory pollutant in treated wastewaters. The novel three-dimensional network-structured catalyst, comprising of δ-MnO2 nanosheets grown on woven carbon microfbers (MnO2 nanosheets/carbon microfber), was synthesized via a facile hydrothermal approach. Catalytic ozonation performance of Ibuprofen removal in water using the new catalyst proves a signifcant enhancement, where Ibuprofen removal efciency of close to 90% was achieved with a catalyst loading of 1% (w/v). In contrast, conventional ozonation was only able to achieve 65% removal efciency under the same operating condition. The enhanced performance with the new catalyst could be attributed to its signifcantly increased available surface active sites and improved mass transfer of reaction media, as a result of the special surface and structure properties of this new three- dimensional network-structured catalyst. Moreover, the new catalyst displays excellent stability and reusability for ibuprofen degradation over successive reaction cycles. The facile synthesis method and low-cost materials render the new catalyst high potential for industrial scaling up. With the combined advantages of high efciency, high stability, and low cost, this study sheds new light for industrial applications of ozonation catalysts. -

)&F1y3x PHARMACEUTICAL APPENDIX to THE
)&f1y3X PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE )&f1y3X PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 3 Table 1. This table enumerates products described by International Non-proprietary Names (INN) which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service (CAS) registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known. Product CAS No. Product CAS No. ABAMECTIN 65195-55-3 ACTODIGIN 36983-69-4 ABANOQUIL 90402-40-7 ADAFENOXATE 82168-26-1 ABCIXIMAB 143653-53-6 ADAMEXINE 54785-02-3 ABECARNIL 111841-85-1 ADAPALENE 106685-40-9 ABITESARTAN 137882-98-5 ADAPROLOL 101479-70-3 ABLUKAST 96566-25-5 ADATANSERIN 127266-56-2 ABUNIDAZOLE 91017-58-2 ADEFOVIR 106941-25-7 ACADESINE 2627-69-2 ADELMIDROL 1675-66-7 ACAMPROSATE 77337-76-9 ADEMETIONINE 17176-17-9 ACAPRAZINE 55485-20-6 ADENOSINE PHOSPHATE 61-19-8 ACARBOSE 56180-94-0 ADIBENDAN 100510-33-6 ACEBROCHOL 514-50-1 ADICILLIN 525-94-0 ACEBURIC ACID 26976-72-7 ADIMOLOL 78459-19-5 ACEBUTOLOL 37517-30-9 ADINAZOLAM 37115-32-5 ACECAINIDE 32795-44-1 ADIPHENINE 64-95-9 ACECARBROMAL 77-66-7 ADIPIODONE 606-17-7 ACECLIDINE 827-61-2 ADITEREN 56066-19-4 ACECLOFENAC 89796-99-6 ADITOPRIM 56066-63-8 ACEDAPSONE 77-46-3 ADOSOPINE 88124-26-9 ACEDIASULFONE SODIUM 127-60-6 ADOZELESIN 110314-48-2 ACEDOBEN 556-08-1 ADRAFINIL 63547-13-7 ACEFLURANOL 80595-73-9 ADRENALONE -

Precipitated Sulfur: Summary Report
Precipitated Sulfur: Summary Report Item Type Report Authors Yuen, Melissa V.; Gianturco, Stephanie L.; Pavlech, Laura L.; Storm, Kathena D.; Yoon, SeJeong; Mattingly, Ashlee N. Publication Date 2020-02 Keywords Precipitated sulfur; Compounding; Food, Drug, and Cosmetic Act, Section 503B; Food and Drug Administration; Outsourcing facility; Drug compounding; Legislation, Drug; United States Food and Drug Administration Rights Attribution-NoDerivatives 4.0 International Download date 02/10/2021 22:32:18 Item License http://creativecommons.org/licenses/by-nd/4.0/ Link to Item http://hdl.handle.net/10713/12274 Summary Report Precipitated Sulfur Prepared for: Food and Drug Administration Clinical use of bulk drug substances nominated for inclusion on the 503B Bulks List Grant number: 2U01FD005946 Prepared by: University of Maryland Center of Excellence in Regulatory Science and Innovation (M-CERSI) University of Maryland School of Pharmacy February 2020 This report was supported by the Food and Drug Administration (FDA) of the U.S. Department of Health and Human Services (HHS) as part of a financial assistance award (U01FD005946) totaling $2,342,364, with 100 percent funded by the FDA/HHS. The contents are those of the authors and do not necessarily represent the official views of, nor an endorsement by, the FDA/HHS or the U.S. Government. 1 Table of Contents REVIEW OF NOMINATIONS ................................................................................................... 4 METHODOLOGY ................................................................................................................... -

Us Anti-Doping Agency
2019U.S. ANTI-DOPING AGENCY WALLET CARDEXAMPLES OF PROHIBITED AND PERMITTED SUBSTANCES AND METHODS Effective Jan. 1 – Dec. 31, 2019 CATEGORIES OF SUBSTANCES PROHIBITED AT ALL TIMES (IN AND OUT-OF-COMPETITION) • Non-Approved Substances: investigational drugs and pharmaceuticals with no approval by a governmental regulatory health authority for human therapeutic use. • Anabolic Agents: androstenediol, androstenedione, bolasterone, boldenone, clenbuterol, danazol, desoxymethyltestosterone (madol), dehydrochlormethyltestosterone (DHCMT), Prasterone (dehydroepiandrosterone, DHEA , Intrarosa) and its prohormones, drostanolone, epitestosterone, methasterone, methyl-1-testosterone, methyltestosterone (Covaryx, EEMT, Est Estrogens-methyltest DS, Methitest), nandrolone, oxandrolone, prostanozol, Selective Androgen Receptor Modulators (enobosarm, (ostarine, MK-2866), andarine, LGD-4033, RAD-140). stanozolol, testosterone and its metabolites or isomers (Androgel), THG, tibolone, trenbolone, zeranol, zilpaterol, and similar substances. • Beta-2 Agonists: All selective and non-selective beta-2 agonists, including all optical isomers, are prohibited. Most inhaled beta-2 agonists are prohibited, including arformoterol (Brovana), fenoterol, higenamine (norcoclaurine, Tinospora crispa), indacaterol (Arcapta), levalbuterol (Xopenex), metaproternol (Alupent), orciprenaline, olodaterol (Striverdi), pirbuterol (Maxair), terbutaline (Brethaire), vilanterol (Breo). The only exceptions are albuterol, formoterol, and salmeterol by a metered-dose inhaler when used -

Downloaded 9/24/2021 7:05:26 PM
Environmental Science Processes & Impacts PAPER View Article Online View Journal | View Issue Solid-phase extraction as sample preparation of water samples for cell-based and other in vitro Cite this: Environ. Sci.: Processes Impacts,2018,20,493 bioassays† a bc d b Peta A. Neale, Werner Brack, Selim A¨ıt-A¨ıssa, Wibke Busch, ef b d Juliane Hollender, Martin Krauss, Emmanuelle Maillot-Marechal,´ Nicole A. Munz,ef Rita Schlichting,b Tobias Schulze, b Bernadette Voglere and Beate I. Escher *abg In vitro bioassays are increasingly used for water quality monitoring. Surface water samples often need to be enriched to observe an effect and solid-phase extraction (SPE) is commonly applied for this purpose. The applied methods are typically optimised for the recovery of target chemicals and not for effect recovery for bioassays. A review of the few studies that have evaluated SPE recovery for bioassays showed a lack of experimentally determined recoveries. Therefore, we systematically measured effect recovery of a mixture of 579 organic chemicals covering a wide range of physicochemical properties that were spiked into a pristine water sample and extracted using large volume solid-phase extraction (LVSPE). Assays indicative of activation of xenobiotic metabolism, hormone receptor-mediated effects and adaptive stress responses were applied, with non-specificeffects determined through cytotoxicity measurements. Overall, effect recovery was found to be similar to chemical recovery for the majority of bioassays and LVSPE blanks had no effect. Multi-layer SPE exhibited greater recovery of spiked chemicals compared to LVSPE, but the blanks triggered cytotoxicity at high enrichment. Chemical recovery data together with single chemical effect data were used to retrospectively estimate with reverse recovery modelling that there was typically Received 19th November 2017 less than 30% effect loss expected due to reduced SPE recovery in published surface water monitoring Accepted 15th February 2018 studies. -
Guidance for the Format and Content of the Protocol of Non-Interventional
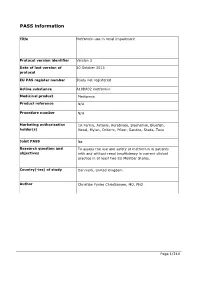
PASS information Title Metformin use in renal impairment Protocol version identifier Version 2 Date of last version of 30 October 2013 protocol EU PAS register number Study not registered Active substance A10BA02 metformin Medicinal product Metformin Product reference N/A Procedure number N/A Marketing authorisation 1A Farma, Actavis, Aurobindo, Biochemie, Bluefish, holder(s) Hexal, Mylan, Orifarm, Pfizer, Sandoz, Stada, Teva Joint PASS No Research question and To assess the use and safety of metformin in patients objectives with and without renal insufficiency in current clinical practice in at least two EU Member States. Country(-ies) of study Denmark, United Kingdom Author Christian Fynbo Christiansen, MD, PhD Page 1/214 Marketing authorisation holder(s) Marketing authorisation N/A holder(s) MAH contact person N/A Page 2/214 1. Table of Contents PASS information .......................................................................................................... 1 Marketing authorisation holder(s) .................................................................................... 2 1. Table of Contents ...................................................................................................... 3 2. List of abbreviations ................................................................................................... 4 3. Responsible parties .................................................................................................... 5 4. Abstract ..................................................................................................................