Structural Studies on Mammalian Septins – New Insights Into Filament Formation
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
![FK506-Binding Protein 12.6/1B, a Negative Regulator of [Ca2+], Rescues Memory and Restores Genomic Regulation in the Hippocampus of Aging Rats](https://docslib.b-cdn.net/cover/6136/fk506-binding-protein-12-6-1b-a-negative-regulator-of-ca2-rescues-memory-and-restores-genomic-regulation-in-the-hippocampus-of-aging-rats-16136.webp)
FK506-Binding Protein 12.6/1B, a Negative Regulator of [Ca2+], Rescues Memory and Restores Genomic Regulation in the Hippocampus of Aging Rats
This Accepted Manuscript has not been copyedited and formatted. The final version may differ from this version. A link to any extended data will be provided when the final version is posted online. Research Articles: Neurobiology of Disease FK506-Binding Protein 12.6/1b, a negative regulator of [Ca2+], rescues memory and restores genomic regulation in the hippocampus of aging rats John C. Gant1, Eric M. Blalock1, Kuey-Chu Chen1, Inga Kadish2, Olivier Thibault1, Nada M. Porter1 and Philip W. Landfield1 1Department of Pharmacology & Nutritional Sciences, University of Kentucky, Lexington, KY 40536 2Department of Cell, Developmental and Integrative Biology, University of Alabama at Birmingham, Birmingham, AL 35294 DOI: 10.1523/JNEUROSCI.2234-17.2017 Received: 7 August 2017 Revised: 10 October 2017 Accepted: 24 November 2017 Published: 18 December 2017 Author contributions: J.C.G. and P.W.L. designed research; J.C.G., E.M.B., K.-c.C., and I.K. performed research; J.C.G., E.M.B., K.-c.C., I.K., and P.W.L. analyzed data; J.C.G., E.M.B., O.T., N.M.P., and P.W.L. wrote the paper. Conflict of Interest: The authors declare no competing financial interests. NIH grants AG004542, AG033649, AG052050, AG037868 and McAlpine Foundation for Neuroscience Research Corresponding author: Philip W. Landfield, [email protected], Department of Pharmacology & Nutritional Sciences, University of Kentucky, 800 Rose Street, UKMC MS 307, Lexington, KY 40536 Cite as: J. Neurosci ; 10.1523/JNEUROSCI.2234-17.2017 Alerts: Sign up at www.jneurosci.org/cgi/alerts to receive customized email alerts when the fully formatted version of this article is published. -

A Complete Compendium of Crystal Structures for the Human SEPT3 Subgroup Reveals Functional Plasticity at a Specific Septin Inte
research papers A complete compendium of crystal structures for IUCrJ the human SEPT3 subgroup reveals functional ISSN 2052-2525 plasticity at a specific septin interface BIOLOGYjMEDICINE Danielle Karoline Silva do Vale Castro,a,b‡ Sabrina Matos de Oliveira da Silva,a,b‡ Humberto D’Muniz Pereira,a Joci Neuby Alves Macedo,a,c Diego Antonio Leonardo,a Napolea˜o Fonseca Valadares,d Patricia Suemy Kumagai,a Jose´ Branda˜o- Received 2 December 2019 Neto,e Ana Paula Ulian Arau´joa and Richard Charles Garratta* Accepted 3 March 2020 aInstituto de Fı´sica de Sa˜o Carlos, Universidade de Sa˜o Paulo, Avenida Joao Dagnone 1100, Sa˜o Carlos-SP 13563-723, b Edited by Z.-J. Liu, Chinese Academy of Brazil, Instituto de Quı´mica de Sa˜o Carlos, Universidade de Sa˜o Paulo, Avenida Trabalhador Sa˜o-carlense 400, c Sciences, China Sa˜o Carlos-SP 13566-590, Brazil, Federal Institute of Education, Science and Technology of Rondonia, Rodovia BR-174, Km 3, Vilhena-RO 76980-000, Brazil, dDepartamento de Biologia Celular, Universidade de Brası´lia, Instituto de Cieˆncias Biolo´gicas, Brası´lia-DF 70910900, Brazil, and eDiamond Light Source, Diamond House, Harwell Science and Innovation ‡ These authors contributed equally to this Campus, Didcot OX11 0DE, United Kingdom. *Correspondence e-mail: [email protected] work. Keywords: septins; GTP binding/hydrolysis; Human septins 3, 9 and 12 are the only members of a specific subgroup of septins filaments; protein structure; X-ray that display several unusual features, including the absence of a C-terminal crystallography. coiled coil. This particular subgroup (the SEPT3 septins) are present in rod-like octameric protofilaments but are lacking in similar hexameric assemblies, which PDB references: SEPT3G–GTP S, 4z51; only contain representatives of the three remaining subgroups. -

Diminished Dosage of 22Q11 Genes Disrupts Neurogenesis and Cortical Development in a Mouse Model of 22Q11 Deletion/Digeorge Syndrome
Diminished dosage of 22q11 genes disrupts neurogenesis and cortical development in a mouse model of 22q11 deletion/DiGeorge syndrome Daniel W. Meechana, Eric S. Tuckera, Thomas M. Maynarda, and Anthony-Samuel LaMantiaa,b,1 aDepartment of Cell and Molecular Physiology and bNeuroscience Center, University of North Carolina School of Medicine, Chapel Hill, NC 27599 Edited by Pasko Rakic, Yale University School of Medicine, New Haven, CT, and approved July 22, 2009 (received for review May 28, 2009) The 22q11 deletion (or DiGeorge) syndrome (22q11DS), the result of flect changes in cortical neurogenesis. We focused on two a 1.5- to 3-megabase hemizygous deletion on human chromosome 22, distinct classes of cortical progenitors: basal progenitors–transit results in dramatically increased susceptibility for ‘‘diseases of cortical amplifying progenitors in the cortical subventricular zone connectivity’’ thought to arise during development, including schizo- (SVZ)–and apical progenitors–self-renewing radial glial stem phrenia and autism. We show that diminished dosage of the genes cells present in the cortical VZ. Each class can be recognized deleted in the 1.5-megabase 22q11 minimal critical deleted region in with combinations of proliferative and molecular markers (Fig. a mouse model of 22q11DS specifically compromises neurogenesis 1). We found reduced frequency of mitotic basal progenitors, and subsequent differentiation in the cerebral cortex. Proliferation of identified by phosphohistone 3 labeling (PH3; a G2/M-phase basal, but not apical, progenitors is disrupted, and subsequently, the cell-cycle marker) as well as SVZ location, throughout the frequency of layer 2/3, but not layer 5/6, projection neurons is altered. -

Cell Type-Specific Gene Expression Patterns Associated with Posttraumatic Stress Disorder in World Trade Center Responders
Kuan et al. Translational Psychiatry (2019) 9:1 https://doi.org/10.1038/s41398-018-0355-8 Translational Psychiatry ARTICLE Open Access Cell type-specific gene expression patterns associated with posttraumatic stress disorder in World Trade Center responders Pei-Fen Kuan1, Xiaohua Yang2,SeanClouston 3,XuRen1,RomanKotov4, Monika Waszczuk4,PrashantK.Singh5, Sean T. Glenn5, Eduardo Cortes Gomez 6, Jianmin Wang6,EvelynBromet4 and Benjamin J. Luft2 Abstract Posttraumatic stress disorder (PTSD), a chronic disorder resulting from severe trauma, has been linked to immunologic dysregulation. Gene expression profiling has emerged as a promising tool for understanding the pathophysiology of PTSD. However, to date, all but one gene expression study was based on whole blood or unsorted peripheral blood mononuclear cell (PBMC), a complex tissue consisting of several populations of cells. The objective of this study was to utilize RNA sequencing to simultaneously profile the gene expression of four immune cell subpopulations (CD4T, CD8T, B cells, and monocytes) in 39 World Trade Center responders (20 with and 19 without PTSD) to determine which immune subsets play a role in the transcriptomic changes found in whole blood. Transcriptome-wide analyses identified cell-specific and shared differentially expressed genes across the four cell types. FKBP5 and PI4KAP1 genes were consistently upregulated across all cell types. Notably, REST and SEPT4, genes linked to neurodegeneration, were among the top differentially expressed genes in monocytes. Pathway analyses identified differentially expressed gene sets involved in mast cell activation and regulation in CD4T, interferon-beta production in CD8T, and neutrophil- fi 1234567890():,; 1234567890():,; 1234567890():,; 1234567890():,; related gene sets in monocytes. -

Anti-SEPT7 Antibody (ARG55299)
Product datasheet [email protected] ARG55299 Package: 100 μl anti-SEPT7 antibody Store at: -20°C Summary Product Description Rabbit Polyclonal antibody recognizes 7-Sep Tested Reactivity Hu, Ms, Rat Tested Application ICC/IF, WB Host Rabbit Clonality Polyclonal Isotype IgG Target Name SEPT7 Antigen Species Human Immunogen Recombinant protein of Human SEPT7 Conjugation Un-conjugated Alternate Names Septin-7; CDC10; CDC10 protein homolog; NBLA02942; SEPT7A; CDC3 Application Instructions Application table Application Dilution ICC/IF 1:50 - 1:100 WB 1:500 - 1:2000 Application Note * The dilutions indicate recommended starting dilutions and the optimal dilutions or concentrations should be determined by the scientist. Positive Control HepG2 Calculated Mw 51 kDa Properties Form Liquid Purification Affinity purification with immunogen. Buffer PBS (pH 7.3), 0.02% Sodium azide and 50% Glycerol Preservative 0.02% Sodium azide Stabilizer 50% Glycerol Storage instruction For continuous use, store undiluted antibody at 2-8°C for up to a week. For long-term storage, aliquot and store at -20°C. Storage in frost free freezers is not recommended. Avoid repeated freeze/thaw cycles. Suggest spin the vial prior to opening. The antibody solution should be gently mixed before use. Note For laboratory research only, not for drug, diagnostic or other use. www.arigobio.com 1/2 Bioinformation Gene Symbol 42254 Gene Full Name septin 7 Background This gene encodes a protein that is highly similar to the CDC10 protein of Saccharomyces cerevisiae. The protein also shares similarity with Diff 6 of Drosophila and with H5 of mouse. Each of these similar proteins, including the yeast CDC10, contains a GTP-binding motif. -

Atherosclerosis-Susceptible and Atherosclerosis-Resistant Pigeon Aortic Cells Express Different Genes in Vivo
University of New Hampshire University of New Hampshire Scholars' Repository New Hampshire Agricultural Experiment Station Publications New Hampshire Agricultural Experiment Station 7-1-2013 Atherosclerosis-susceptible and atherosclerosis-resistant pigeon aortic cells express different genes in vivo Janet L. Anderson University of New Hampshire, [email protected] C. M. Ashwell University of New Hampshire - Main Campus S. C. Smith University of New Hampshire - Main Campus R. Shine University of New Hampshire - Main Campus E. C. Smith University of New Hampshire - Main Campus See next page for additional authors Follow this and additional works at: https://scholars.unh.edu/nhaes Part of the Poultry or Avian Science Commons Recommended Citation J. L. Anderson, C. M. Ashwell, S. C. Smith, R. Shine, E. C. Smith and R. L. Taylor, Jr. Atherosclerosis- susceptible and atherosclerosis-resistant pigeon aortic cells express different genes in vivo Poultry Science (2013) 92 (10): 2668-2680 doi:10.3382/ps.2013-03306 This Article is brought to you for free and open access by the New Hampshire Agricultural Experiment Station at University of New Hampshire Scholars' Repository. It has been accepted for inclusion in New Hampshire Agricultural Experiment Station Publications by an authorized administrator of University of New Hampshire Scholars' Repository. For more information, please contact [email protected]. Authors Janet L. Anderson, C. M. Ashwell, S. C. Smith, R. Shine, E. C. Smith, and Robert L. Taylor Jr. This article is available at University of New Hampshire Scholars' Repository: https://scholars.unh.edu/nhaes/207 Atherosclerosis-susceptible and atherosclerosis-resistant pigeon aortic cells express different genes in vivo J. -

Protein Kinase A-Mediated Septin7 Phosphorylation Disrupts Septin Filaments and Ciliogenesis
cells Article Protein Kinase A-Mediated Septin7 Phosphorylation Disrupts Septin Filaments and Ciliogenesis Han-Yu Wang 1,2, Chun-Hsiang Lin 1, Yi-Ru Shen 1, Ting-Yu Chen 2,3, Chia-Yih Wang 2,3,* and Pao-Lin Kuo 1,2,4,* 1 Department of Obstetrics and Gynecology, College of Medicine, National Cheng Kung University, Tainan 701, Taiwan; [email protected] (H.-Y.W.); [email protected] (C.-H.L.); [email protected] (Y.-R.S.) 2 Institute of Basic Medical Sciences, College of Medicine, National Cheng Kung University, Tainan 701, Taiwan; [email protected] 3 Department of Cell Biology and Anatomy, College of Medicine, National Cheng Kung University, Tainan 701, Taiwan 4 Department of Obstetrics and Gynecology, National Cheng-Kung University Hospital, Tainan 704, Taiwan * Correspondence: [email protected] (C.-Y.W.); [email protected] (P.-L.K.); Tel.: +886-6-2353535 (ext. 5338); (C.-Y.W.)+886-6-2353535 (ext. 5262) (P.-L.K.) Abstract: Septins are GTP-binding proteins that form heteromeric filaments for proper cell growth and migration. Among the septins, septin7 (SEPT7) is an important component of all septin filaments. Here we show that protein kinase A (PKA) phosphorylates SEPT7 at Thr197, thus disrupting septin filament dynamics and ciliogenesis. The Thr197 residue of SEPT7, a PKA phosphorylating site, was conserved among different species. Treatment with cAMP or overexpression of PKA catalytic subunit (PKACA2) induced SEPT7 phosphorylation, followed by disruption of septin filament formation. Constitutive phosphorylation of SEPT7 at Thr197 reduced SEPT7-SEPT7 interaction, but did not affect SEPT7-SEPT6-SEPT2 or SEPT4 interaction. -

1 Supporting Information for a Microrna Network Regulates
Supporting Information for A microRNA Network Regulates Expression and Biosynthesis of CFTR and CFTR-ΔF508 Shyam Ramachandrana,b, Philip H. Karpc, Peng Jiangc, Lynda S. Ostedgaardc, Amy E. Walza, John T. Fishere, Shaf Keshavjeeh, Kim A. Lennoxi, Ashley M. Jacobii, Scott D. Rosei, Mark A. Behlkei, Michael J. Welshb,c,d,g, Yi Xingb,c,f, Paul B. McCray Jr.a,b,c Author Affiliations: Department of Pediatricsa, Interdisciplinary Program in Geneticsb, Departments of Internal Medicinec, Molecular Physiology and Biophysicsd, Anatomy and Cell Biologye, Biomedical Engineeringf, Howard Hughes Medical Instituteg, Carver College of Medicine, University of Iowa, Iowa City, IA-52242 Division of Thoracic Surgeryh, Toronto General Hospital, University Health Network, University of Toronto, Toronto, Canada-M5G 2C4 Integrated DNA Technologiesi, Coralville, IA-52241 To whom correspondence should be addressed: Email: [email protected] (M.J.W.); yi- [email protected] (Y.X.); Email: [email protected] (P.B.M.) This PDF file includes: Materials and Methods References Fig. S1. miR-138 regulates SIN3A in a dose-dependent and site-specific manner. Fig. S2. miR-138 regulates endogenous SIN3A protein expression. Fig. S3. miR-138 regulates endogenous CFTR protein expression in Calu-3 cells. Fig. S4. miR-138 regulates endogenous CFTR protein expression in primary human airway epithelia. Fig. S5. miR-138 regulates CFTR expression in HeLa cells. Fig. S6. miR-138 regulates CFTR expression in HEK293T cells. Fig. S7. HeLa cells exhibit CFTR channel activity. Fig. S8. miR-138 improves CFTR processing. Fig. S9. miR-138 improves CFTR-ΔF508 processing. Fig. S10. SIN3A inhibition yields partial rescue of Cl- transport in CF epithelia. -
Background Methods Conclusions Results
Gene Expression Profiles of Pediatric Tuberculosis Patients and Exposed Controls from India Jeffrey A Tornheim,1 Mandar Paradkar,2 Anil Madugundu,4,5 Nikhil Gupte,2 Vandana Kulkarni,2 Sreelakshmi Sreenivasamurthy,4,5 Remya Raja,4 Neeta Pradhan,2 Shri Vijay Bala Yogendra Shivakumar,6 Chhaya Valvi,3 Rewa Kohli,2 Padmapriyadarsini Chandrasekaran,7 Vidya Mave,2 Akhilesh Pandey,4,5 and Amita Gupta1 1Center for Clinical Global Health Education, Division of Infectious Diseases, Johns Hopkins University School of Medicine, Baltimore, MD, USA, 2Byramjee Jeejeebhoy Government Medical College – Johns Hopkins University Clinical Research Site, Pune, Maharashtra, India, 3Byramjee Jeejeebhoy Government Medical College, Pune, Maharashtra, India, 4National Institute of Mental Health and Neurosciences - Institute of Bioinformatics Lab, Bangalore, India, 5Institute of Genetic Medicine, Johns Hopkins University School of Medicine, Baltimore, MD, USA, 6Johns Hopkins University – India office (CCGHE), Pune, Maharashtra, India, 7National Institute for Research in Tuberculosis, Chennai, Tamil Nadu, India BACKGROUND RESULTS—Differential Gene Expression Tuberculosis (TB) is the #1 infectious disease killer worldwide, and 27% of cases are in India (WHO 2016). Aim 1: Differential gene expression between pediatric cases (N=16) and controls (N=32) at enrollment Children frequently have either paucibacillary disease, are too young to provide adequate sputum Gene Counts Fold Change Distribution samples, or develop extrapulmonary disease, forcing clinicians to rely on diagnostic tests with poor sensitivity and frequent presumptive treatment for TB in children (Perez-Velez NEJM 2012). Several studies have attempted to use biomarkers of host response to infection as an alternative means of Number of Differentially Expressed Genes between Cases and Controls, diagnosing TB, but these studies have not included many children, Indian patients, or microbiologically by False Discovery Rate (FDR) and Log2-Fold Change Any Log2 >0.5 Log2 >1.0 Log2 >1.5 Log2 >2.0 confirmed patients. -

The Requirement of SEPT2 and SEPT7 for Migration and Invasion in Human Breast Cancer Via MEK/ERK Activation
www.impactjournals.com/oncotarget/ Oncotarget, Vol. 7, No. 38 Research Paper The requirement of SEPT2 and SEPT7 for migration and invasion in human breast cancer via MEK/ERK activation Nianzhu Zhang1,*, Lu Liu2,*, Ning Fan2, Qian Zhang1, Weijie Wang1, Mingnan Zheng3, Lingfei Ma4, Yan Li2, Lei Shi1,5 1Institute of Cancer Stem Cell, Cancer Center, Dalian Medical University, Dalian, 116044, Liaoning, P.R.China 2College of Basic Medical Sciences, Dalian Medical University, Dalian, 116044 Liaoning, P.R.China 3Department of Gynecology and Obstetrics, Dalian Municipal Central Hospital Affiliated to Dalian Medical University, Dalian, 116033, Liaoning, P.R.China 4The First Affiliated Hospital of Dalian Medical University, Dalian, 116011, Liaoning, P.R.China 5State Key Laboratory of Drug Research, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, Shanghai, 201203, P.R.China *These authors contributed equally to this work Correspondence to: Lei Shi, email: [email protected] Yan Li, email: [email protected] Lingfei Ma, email: [email protected] Keywords: septin, forchlorfenuron, breast cancer, invasion, MAPK Received: April 24, 2016 Accepted: July 28, 2016 Published: August 19, 2016 ABSTRACT Septins are a novel class of GTP-binding cytoskeletal proteins evolutionarily conserved from yeast to mammals and have now been found to play a contributing role in a broad range of tumor types. However, their functional importance in breast cancer remains largely unclear. Here, we demonstrated that pharmaceutical inhibition of global septin dynamics would greatly suppress proliferation, migration and invasiveness in breast cancer cell lines. We then examined the expression and subcellular distribution of the selected septins SEPT2 and SEPT7 in breast cancer cells, revealing a rather variable localization of the two proteins with cell cycle progression. -
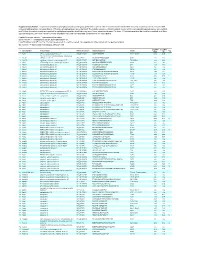
Supplementary Table 1
Supplementary Table 1. Large-scale quantitative phosphoproteomic profiling was performed on paired vehicle- and hormone-treated mTAL-enriched suspensions (n=3). A total of 654 unique phosphopeptides corresponding to 374 unique phosphoproteins were identified. The peptide sequence, phosphorylation site(s), and the corresponding protein name, gene symbol, and RefSeq Accession number are reported for each phosphopeptide identified in any one of three experimental pairs. For those 414 phosphopeptides that could be quantified in all three experimental pairs, the mean Hormone:Vehicle abundance ratio and corresponding standard error are also reported. Peptide Sequence column: * = phosphorylated residue Site(s) column: ^ = ambiguously assigned phosphorylation site Log2(H/V) Mean and SE columns: H = hormone-treated, V = vehicle-treated, n/a = peptide not observable in all 3 experimental pairs Sig. column: * = significantly changed Log 2(H/V), p<0.05 Log (H/V) Log (H/V) # Gene Symbol Protein Name Refseq Accession Peptide Sequence Site(s) 2 2 Sig. Mean SE 1 Aak1 AP2-associated protein kinase 1 NP_001166921 VGSLT*PPSS*PK T622^, S626^ 0.24 0.95 PREDICTED: ATP-binding cassette, sub-family A 2 Abca12 (ABC1), member 12 XP_237242 GLVQVLS*FFSQVQQQR S251^ 1.24 2.13 3 Abcc10 multidrug resistance-associated protein 7 NP_001101671 LMT*ELLS*GIRVLK T464, S468 -2.68 2.48 4 Abcf1 ATP-binding cassette sub-family F member 1 NP_001103353 QLSVPAS*DEEDEVPVPVPR S109 n/a n/a 5 Ablim1 actin-binding LIM protein 1 NP_001037859 PGSSIPGS*PGHTIYAK S51 -3.55 1.81 6 Ablim1 actin-binding -

The Potential Role of SEPT6 in Liver Fibrosis and Human Hepatocellular Carcinoma. Arch Gastroenterol Res
https://www.scientificarchives.com/journal/archives-of-gastroenterology-research Archives of Gastroenterology Research Commentary The Potential Role of SEPT6 in Liver Fibrosis and Human Hepatocellular Carcinoma Yuhui Fan1,2,3*, Mei Liu3* 1Department of Medicine II, Liver Center Munich, University Hospital, Ludwig-Maximilians-University of Munich, Munich, Germany 2Comprehensive Cancer Center München TUM, Klinikum rechts der Isar, Technical University of Munich (TUM), Munich, Germany 3Institute of Liver and Gastrointestinal Diseases, Department of Gastroenterology, Tongji Hospital of Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei 430030, China *Correspondence should be addressed to Yuhui Fan; [email protected], Mei Liu; [email protected] Received date: May 19, 2020, Accepted date: May 26, 2020 Copyright: © 2020 Fan Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Liver fibrosis is a reversible wound-healing response in and chemotactic has been regarded as the main driver which a variety of cells and factors are involved in and of liver fibrosis [7], thus ECM production is enhanced results in excessive deposition of extracellular matrix [8]. Paracrine signals from damaged epithelial cells, (ECM) [1]. Cirrhosis is one of the significant causes of fibrotic tissue microenvironment, disorders of immunity portal hypertension and end-stage liver disease, and it is and systemic metabolism, intestinal dysbiosis and the 14th most common cause of death around the world. hepatitis virus products can directly or indirectly induce Approximately 1.03 million people worldwide die from HSCs activation [9].