Calcium Hypochlorite Granular Safety Data Sheet
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Billing Code: 3510-Ds-P Department Of
This document is scheduled to be published in the Federal Register on 01/30/2015 and available online at http://federalregister.gov/a/2015-01795, and on FDsys.gov BILLING CODE: 3510-DS-P DEPARTMENT OF COMMERCE International Trade Administration C-570-009 Calcium Hypochlorite from the People’s Republic of China: Countervailing Duty Order AGENCY: Enforcement and Compliance, International Trade Administration, Department of Commerce SUMMARY: Based on affirmative final determinations by the Department of Commerce (“Department”) and the International Trade Commission (“ITC”), the Department is issuing a countervailing duty order on calcium hypochlorite from the People’s Republic of China (“PRC”). EFFECTIVE DATE: [(Insert date of publication in the Federal Register]. FOR FURTHER INFORMATION CONTACT: Katie Marksberry, AD/CVD Operations, Office V, Enforcement and Compliance, International Trade Administration, U.S. Department of Commerce, 14th Street and Constitution Avenue, NW, Washington, DC 20230; telephone: (202) 482-7906. SUPPLEMENTARY INFORMATION: Background In accordance with section 705(d) of the Tariff Act of 1930, as amended (“the Act”), on December 15, 2014, the Department published its final determination that countervailable subsidies are being provided to producers and exporters of calcium hypochlorite from the PRC.1 On January 21, 2015, the ITC notified the Department of its final determination pursuant to 1 See Calcium Hypochlorite from the People’s Republic of China: Final Affirmative Countervailing Duty Determination; 79 FR -

THE JOURNAL of INDUSTRIAL and ENGINEERING CHEMISTRY 953 with Stirring, and the Flask Allowed to Stand
OCt., 1917 THE JOURNAL OF INDUSTRIAL AND ENGINEERING CHEMISTRY 953 with stirring, and the flask allowed to stand. The ascertaining the presence of aniline, the calcium hypo- solution, when the precipitate has settled, is filtered chlorite test is employed. The quantitative deter- through a Gooch crucible, using a bell jar arrangement, mination is carried out by precipitating the aniline into a 250 to 300 cc. Erlenmeyer flask. The crucible as tribromoaniline with bromine water. The precipi- is fitted preferably with a small circular filter paper tate is caught on a tared filter paper, dried in vacuo, (cut to size) instead of asbestos. The flask should be and weighed. washed with wash-ether (100parts absolute ether and In this connection, it occurred to the writer that it 2 parts alcoholic HC1) while filtering so as not to allow might be possible to work out, for estimating the any ferric chloride to dry on the white precipitate or aniline, a colorimetric method based on the qualita- on the crucible. The usual precautions of washing the tive test with hypochlorite. If this were possible, flask, crucible and funnel are taken to insure complete it certainly would be desirable, for not only would transfer of iron. It is not necessary, however, to trans- considerable time and effort be saved but probably fer all of the aluminum chloride precipitate to the a considerably smaller sample than IO liters would be Gooch crucible. The aluminum precipitate is removed sufficient and quantitative measurements could also with the paper from the crucible by tapping it into a be applied in cases where the amount of aniline in 25 cc. -

Chloroform 18.08.2020.Pdf
Chloroform Chloroform, or trichloromethane, is an organic compound with formula CHCl3. It is a colorless, sweet-smelling, dense liquid that is produced on a large scale as a precursor to PTFE. It is also a precursor to various refrigerants. It is one of the four chloromethanes and a trihalomethane. It is a powerful anesthetic, euphoriant, anxiolytic and sedative when inhaled or ingested. Formula: CHCl₃ IUPAC ID: Trichloromethane Molar mass: 119.38 g/mol Boiling point: 61.2 °C Density: 1.49 g/cm³ Melting point: -63.5 °C The molecule adopts a tetrahedral molecular geometry with C3v symmetry. Chloroform volatilizes readily from soil and surface water and undergoes degradation in air to produce phosgene, dichloromethane, formyl chloride, carbon monoxide, carbon dioxide, and hydrogen chloride. Its half-life in air ranges from 55 to 620 days. Biodegradation in water and soil is slow. Chloroform does not significantly bioaccumulate in aquatic organisms. Production:- In industry production, chloroform is produced by heating a mixture of chlorine and either chloromethane (CH3Cl) or methane (CH4). At 400–500 °C, a free radical halogenation occurs, converting these precursors to progressively more chlorinated compounds: CH4 + Cl2 → CH3Cl + HCl CH3Cl + Cl2 → CH2Cl2 + HCl CH2Cl2 + Cl2 → CHCl3 + HCl Chloroform undergoes further chlorination to yield carbon tetrachloride (CCl4): CHCl3 + Cl2 → CCl4 + HCl The output of this process is a mixture of the four chloromethanes (chloromethane, dichloromethane, chloroform, and carbon tetrachloride), which can then be separated by distillation. Chloroform may also be produced on a small scale via the haloform reaction between acetone and sodium hypochlorite: 3 NaClO + (CH3)2CO → CHCl3 + 2 NaOH + CH3COONa Deuterochloroform[ Deuterated chloroform is an isotopologue of chloroform with a single deuterium atom. -

Chemistry of Natural Resources *OCE/26193* Candidates Answer on the Question Paper
ADVANCED SUBSIDIARY GCE CHEMISTRY B (SALTERS) F332/TEST Chemistry of Natural Resources *OCE/26193* Candidates answer on the question paper. Friday 27 May 2011 Afternoon OCR supplied materials: • Data Sheet for Chemistry B (Salters) (inserted) Duration: 1 hour 45 minutes • Advance Notice: ‘Polymers on the move’ (inserted) Other materials required: • Scientific Calculator *F332TEST* INSTRUCTIONS TO CANDIDATES • The insert will be found in the centre of this document. • Write your name, centre number and candidate number in the boxes above. Please write clearly and in capital letters. • Use black ink. Pencil may be used for graphs and diagrams only. • Read each question carefully. Make sure you know what you have to do before starting your answer. • Write your answer to each question in the space provided. If additional space is required, you should use the lined pages at the end of this booklet. The question number(s) must be clearly shown. • Answer all the questions. • Do not write in the bar codes. INFORMATION FOR CANDIDATES • The number of marks is given in brackets [ ] at the end of each question or part question. • Where you see this icon you will be awarded marks for the quality of written communication in your answer. This means for example you should: • ensure that text is legible and that spelling, punctuation and grammar are accurate so that meaning is clear; • organise information clearly and coherently, using specialist vocabulary when appropriate. • You may use a scientific calculator. • The insert ‘Polymers on the move’ is provided for use with question 5. • A copy of the Data Sheet for Chemistry B (Salters) is provided as an insert with this question paper. -

CHEMICAL STORAGE SEGREGATION GUIDELINES Incompatible Chemicals Should Always Be Handled and Stored So That They Do Not Accidentally Come in Contact with Each Other
Laboratory Safety Reminders January 2007 ♦ Mount Holyoke College – Environmental Health and Safety CHEMICAL STORAGE SEGREGATION GUIDELINES Incompatible chemicals should always be handled and stored so that they do not accidentally come in contact with each other. This list is not complete, nor are all compatibilities shown. These materials can react to produce excessive heat, harmful vapors, and/or other deadly reactions. Always know the hazards and incompatibilities of a chemical before using it. Chemicals Avoid Accidental Contact With Acetic acid Chromic acid, nitric acid, permanganates, peroxides Hydroxyl-containing compounds such as perchloric acid, Acetic anhydride ethylene glycol Concentrated nitric acid and sulfuric acid mixtures, peroxides (i.e. Acetone peracetic acid solution, hydrogen peroxide) Acetylene Chlorine, bromine, copper, silver, fluorine, mercury Alkali, alkaline earth and strongly electropositive metals (powered Carbon dioxide, carbon tetrachloride and other chlorinated aluminum, magnesium, sodium, hydrocarbons potassium) Mercury, chlorine, calcium hypochlorite, iodine, bromine, hydrogen Ammonia (anhydrous) fluoride Acids, metal powders, flammable liquids, chlorates, nitrates, sulfur, Ammonium nitrate finely divided organics, combustibles Aniline Nitric acid, hydrogen peroxide Arsenical compounds Any reducing agent Azides Acids Ammonia, acetylene, butadiene, butane, other petroleum gases, Bromine sodium carbide, turpentine, benzene, finely divided metals Calcium oxide Water Carbon activated Calcium hypochlorite, other -

Kinetics of the Process of Conversion of Production Of
European Journal of Molecular & Clinical Medicine ISSN 2515-8260 Volume 07, Issue 07, 2020 Kinetics Of The Process Of Conversion Of Production Of Calcium Chlorate With The Use Of A Filtrate Of Hydrocaldic Acid Decomposition Of Washed Ck Phosphonic Concentrate Khamdamova Shohida Sherzodovna1, Rozikova Dilshoda Abdullajanovna2 1Doctor of Technical Sciences Associate Professor of the Department of Chemical Technology, Fergana Polytechnic Institute, Fergana Uzbekistan 2Basic doctoral student of the Namangan Engineering and Technological Institute, Namangan, Uzbekistan Abstract: The process of conversion of 20-25% solutions of calcium chloride obtained by decomposition of phosphorites of Central Kizikumes with sodium chlorate at 50, 75, 100 and 125 ° C with evaporation and without evaporation of conversion solutions has been studied. The process activation energies, the order and the rate constant of the conversion reaction as well as kinetic values of the reagent consumption are determined depending on temperature, time and carrying conditions of processes. The order of the conversion process of calcium chloride with sodium chlorate equals one. It is confirmed by the fact that the conversion rate constant calculated on the basis of experimental data remains practically constant for each temperature. Also, the linear dependence of lg (C0-Cτ) on τ also indicates the first order of the conversion process of calcium chloride with sodium chlorate. The rate constant of conversion increases with a rise in temperature. Its dependence on temperature obeys Arrhenius law. To establish values of the conversion rate constant for different temperatures, the constants (К0) for 20 and 25% solutions of calcium chloride have been calculated by Arrhenius equation and the dependence equation of lg K on 1 / T has been derived. -

Calcium Hypochlorite
Common Name: CALCIUM HYPOCHLORITE CAS Number: 7778-54-3 DOT Number: UN 1748 (Dry or with more than 39% Chlorine) UN 2880 (Hydrated) UN 2208 (Dry with between 10% to 39% RTK Substance number: 0323 Chlorine) Date: December 1996 Revision: April 2003 ------------------------------------------------------------------------- ------------------------------------------------------------------------- HAZARD SUMMARY * Calcium Hypochlorite can affect you when breathed in. * If you think you are experiencing any work-related health * Contact can severely irritate and burn the eyes and skin. problems, see a doctor trained to recognize occupational * Breathing Calcium Hypochlorite can irritate the nose and diseases. Take this Fact Sheet with you. throat. * Breathing Calcium Hypochlorite can irritate the lungs WORKPLACE EXPOSURE LIMITS causing coughing and/or shortness of breath. Higher No occupational exposure limits have been established for exposures may cause a build-up of fluid in the lungs Calcium Hypochlorite. This does not mean that this (pulmonary edema), a medical emergency, with severe substance is not harmful. Safe work practices should always shortness of breath. be followed. IDENTIFICATION WAYS OF REDUCING EXPOSURE Calcium Hypochlorite is a white powder, granule, or pellet * Where possible, enclose operations and use local exhaust with a strong Chlorine-like odor. It is used to kill algae and ventilation at the site of chemical release. If local exhaust bacteria, in bleach and in pool chemical products. ventilation or enclosure is not used, respirators should be worn. REASON FOR CITATION * Wear protective work clothing. * Calcium Hypochlorite is on the Hazardous Substance * Wash thoroughly immediately after exposure to Calcium List because it is cited by DOT, NFPA and EPA. Hypochlorite. * Definitions are provided on page 5. -

Safe Handling and Disposal of Chemicals Used in the Illicit Manufacture of Drugs
Vienna International Centre, PO Box 500, 1400 Vienna, Austria Tel.: (+43-1) 26060-0, Fax: (+43-1) 26060-5866, www.unodc.org Guidelines for the Safe handling and disposal of chemicals used in the illicit manufacture of drugs United Nations publication USD 26 Printed in Austria ISBN 978-92-1-148266-9 Sales No. E.11.XI.14 ST/NAR/36/Rev.1 V.11-83777—September*1183777* 2011—300 Guidelines for the Safe handling and disposal of chemicals used in the illlicit manufacture of drugs UNITED NATIONS New York, 2011 Symbols of United Nations documents are composed of letters combined with figures. Mention of such symbols indicates a reference to a United Nations document. ST/NAR/36/Rev.1 UNITED NATIONS PUBLICATION Sales No. E.11.XI.14 ISBN 978-92-1-148266-9 eISBN 978-92-1-055160-1 © United Nations, September 2011. All rights reserved. The designations employed and the presentation of material in this publication do not imply the expression of any opinion whatsoever on the part of the Secretariat of the United Nations concerning the legal status of any country, territory, city or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries. Requests for permission to reproduce this work are welcomed and should be sent to the Secretary of the Publications Board, United Nations Headquarters, New York, N.Y. 10017, U.S.A. or also see the website of the Board: https://unp.un.org/Rights.aspx. Governments and their institutions may reproduce this work without prior authoriza- tion but are requested to mention the source and inform the United Nations of such reproduction. -
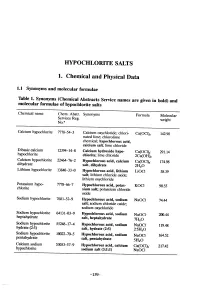
Hypochlorite Salts, As Weil As Chlorine Itself, in Aqueous Solution Produce Equilbrium Mixures of Hypochlorous Acid, Hypochlorite Ion and Chlorine
HYOCHLORITE SALTS 1. Chemical and Physical Data 1.1 Synonyms and molecular rormulae Table 1. Synonyms (Chemical Abstracts Service names are given in bold) and molecular rormulae or hyphlorite salts Chemical name Chem. Abstr. Synonyms Formula Molecular Servces Reg. weight No.u Calcium hyphlorite 7778-54-3 Calcium oxychloride; chlori- Ca(OCI)2 142.98 nated lime; chlorolime chemical; hypochlorous acid, calcium salt; lime chloride Dibasic calcium 12394-14-8 Calcium hydroxide hyp Ca(OCI)2" 291.14 hyphlorite chlonte; lime chloride 2Ca(OH)2 Calcium hyphlorite 22476-2 Hypochlorous acid, calcium Ca(OCI)2" 174.98 dihydrate salt, dihydrate 2H2O Lithium hyphlorite 1384-33-0 Hypchlorous acid, lithium LiOCI 58.39 salt; lithium chloride oxide; lithium oxychloride Potasium hyp 7778-6-7 Hypchlorous acid, potas- KOCI 90.55 chlorite sium salt; potasium chloride oxide Sodium hyphlorite 7681-52-9 Hyphlorous acid, sodium NaOCl 74.44 salt; soium chloride oxide; soium oxychloride Soium hyphlorite 64131-03-9 Hypchlorous acid, sodium heptahydrate NaOCl- 20.44 salt, heptahydrate 7H2O Sodium hyphlorite 5524-17-4 Hyphlorous acid, sodium NaOCI. 119.48 hydrate (2:5) salt, hydrate (2:5) 2-5H2O Sodium hyphlorite 10022-70-5 Hyphlorous acid, sodium NaOCI" 164.52 pentahydrate salt, pentahydrate 5H2O Calcium soium 53053-57-9 Hypchlorous acid, calcium Ca(OCI)2- 217.42 hyphlorite sodium salt (3:1:1) NaOCI -159- 160 lARe MONOGRAHS VOLUME 52 1.2 Chernical and physical properties or the pure substances From Weast (1989) unless otherwise specified Calciurn hyphlorite (a) Description: White powder or flat plates (b) Melting-point: Decomposes at 100°C (c) Density Specific gravity = 2.35 (d) Solubility: Soluble in cold water, 21.4% soluble at 25°C (Wojtowicz, 1979); insoluble in ethanol (e) Stability: Solid form decomposes exothermically when heated to 175°C, releasing oxygen (Mannsvile Chemical Products Corp., 1987). -

Six-Year Review 3 Technical Support Document for Chlorate
Six-Year Review 3 Technical Support Document for Chlorate Office of Water (4607M) EPA-810-R-16-013 December 2016 Disclaimer This document is not a regulation. It is not legally enforceable, and does not confer legal rights or impose legal obligations on any party, including EPA, states, or the regulated community. While EPA has made every effort to ensure the accuracy of any references to statutory or regulatory requirements, the obligations of the interested stakeholders are determined by statutes, regulations or other legally binding requirements, not this document. In the event of a conflict between the information in this document and any statute or regulation, this document would not be controlling. This page intentionally left blank. Table of Contents 1 Introduction ................................................................................................................. 1-1 2 Contaminant Background .......................................................................................... 2-1 2.1 Chemical and Physical Properties ................................................................................. 2-1 2.2 Production, Use and Release ......................................................................................... 2-2 2.2.1 Commercial Production and Use in Industry and Agriculture ........................... 2-2 2.2.2 Incidental Production and Release ...................................................................... 2-6 2.3 Environmental Fate ...................................................................................................... -

ECR Calcium Hypochlorite Granules EPA Reg
STORAGE AND DISPOSAL ® Read before using ECR CALCIUM Do not contaminate food or feed by storage, disposal, or cleaning of equipment. Pesticide Storage- Keep this product dry in a tightly closed container when not in use. HYPOCHLORITE Store in a cool, dry, well ventilated area away from heat or open flame. In case of decom- position, isolate container (if possible) and flood area with large amounts of water to dis- solve all materials before discarding this container. PRECAUTIONARY STATEMENTS Pesticide Disposal - Pesticide wastes may be hazardous. Improper disposal of excess HAZARDS TO HUMAN AND DOMESTIC ANIMALS GRANULES pesticide, spray mixture or rinsate is a violation of Federal Law. If these wastes cannot be disposed of use according to label instructions, contact you State Pesticide or Environ- DANGER : Highly corrosive. Causes irreversible eye damage and skin burns. Do not get in Active Ingredient: mental Control Agency, or the Hazardous Waste Representative at the nearest EPA Re- eyes, on skin, or on clothing. Wear goggles or face shield and rubber gloves when handling gional Office for guidance. this product. Wash thoroughly with soap and water after handling and before eating, drink- Calcium Hypochlorite ………………………………… 68.0% Container Disposal - Non-Refillable container. Triple rinse as follows: Empty the remain- ing, chewing gum, using tobacco, or using the toilet. Remove and wash contaminated cloth- Inert Ingredients ……………………………………… 32.0% ing contents into application equipment or a mix tank. Fill the container 1/4 full with water. ing and shoes before reuse. May be Fatal if swallowed. Irritating to nose and throat. Avoid Replace and tighten closures. -

Material Safety Data Sheet
MATERIAL SAFETY DATA SHEET CALCIUM HYPOCHLORITE, SOLID 1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION Brenntag Canada Inc. WHMIS#: 00020005 43 Jutland Rd. Index: GCD0313/15D Toronto, ON Effective Date: 2015 October 02 M8Z 2G6 (416) 259-8231 Date of Revision: 2015 October 02 Website: http://www.brenntag.ca EMERGENCY TELEPHONE NUMBER (For Emergencies Involving Chemical Spills or Releases) 1 855 273 6824 PRODUCT IDENTIFICATION Product Name: Calcium Hypochlorite, Solid. Chemical Name: Hypochlorous acid, calcium salt. Synonyms: HTH Dry Chlorine Granular; Calcium Hypochlorite Granular; Calcium Hypo; Calcium Hypo Pitchl Gran 65%; Cal Hypo Comm 65% Tabs; Calcium Hypo PPG 3" Tabs; Accu-Tab White, Blue, SI, Blue SI, White SI Tabs. Chemical Family: Hypochlorous acid salt. Molecular Formula: Ca (ClO) 2. Product Use: Swimming pool water disinfectant. Water treatment. Oxidizing agent. Chemical intermediate. WHMIS Classification / Symbol: C: Oxidizer E: Corrosive READ THE ENTIRE MSDS FOR THE COMPLETE HAZARD EVALUATION OF THIS PRODUCT. 2. COMPOSITION, INFORMATION ON INGREDIENTS (Not Intended As Specifications) Ingredient CAS# ACGIH TLV (TWA) % Concentration Calcium Hypochlorite 7778-54-3 --- 60 - 80 Sodium Chloride 7647-14-5 --- 10 - 30 Calcium Carbonate 471-34-1 --- 1 - 5 Calcium Hydroxide 1305-62-0 5 mg/m³ 1 - 5 Calcium Chlorate 10137-74-3 --- 0 - 3 3. HAZARDS IDENTIFICATION Calcium Hypochlorite, Solid Brenntag Canada Inc. WHMIS Number : 00020005 Date of Revision: 2015 October 02 Page 2 of 9 EMERGENCY OVERVIEW: Corrosive! Causes severe skin and eye burns. Dust is extremely irritating to respiratory tract. See "Other Health Effects" Section. Oxidizing material. Contact with other combustible material can cause fire. This material is a strong oxidizer which is stable under normal conditions, but can decompose if contaminated.