Tris (1,3(Dihydroxy(2(Propyl) Amine, a Planar Trialkylamine
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Lactide/^Caprolactone Polymerization Behavior of Monomeric Aryloxytitanatrane
698 Bull. Korean Chem. Soc. 2007, Vol. 28, No. 4 Notes Synthesis, X-ray Structure, and /-Lactide/^Caprolactone Polymerization Behavior of Monomeric Aryloxytitanatrane Sang-deok Mun, Younjin Hong, and Youngjo Kim* Department of Chemistry, Chungbuk National University, Cheongju, Chungbuk 361-763, Korea. *E-mail: 가[email protected] Received February 2, 2007 Key Words : Titanatrane, Mononuclear, Ring opening polymerization, Polylactide, Polycaprolactone The chemistry of atrane coordinated by the central nitro polymerization (ROP) of l-lactide (l-LA) or ^caprolactone gen atom as well as all three arms of deprotonated triethanol 任-CL) are now one of popular research fields in the homo amine ligand, imino-2,2',2''-triethanolate, has been inten geneous catalysis. In this regard, a new mononuclear tita- sively studied over the past few decades and its examples are natrane obtained was used as a catalyst for the ROP of l-LA now known across the periodic table.1 Most studies have and £-CL. focused on the use of main group elements such as silicon, The treatment of Ti(O-i-Pr)4 with 1 equivalent of 2,6-di- phosphorus, aluminum, and tin in the formation of atrane.1 tert-butylphenol and 1 equivalent of triethanolamine in THF In view of the well known significant number of similarities gave, after workup, novel mononuclear titanatranes 1 as in the chemistries of tin and titanium, relatively few reports orange-yellow crystals in 81% isolated yield. After recrystal have appeared concerning metallic titanatranes with a lization using toluene, 1 was used as a catalyst for making transannular N—Ti bond from bridgehead N atom in polylactide (PLA) and polycaprolactone (PCL). -

Electron Transfer and Modification of Oligosilanylsilatranes and Related
This is an open access article published under a Creative Commons Attribution (CC-BY) License, which permits unrestricted use, distribution and reproduction in any medium, provided the author and source are cited. Article pubs.acs.org/Organometallics Electron Transfer and Modification of Oligosilanylsilatranes and Related Derivatives † ‡ § Mohammad Aghazadeh Meshgi, Judith Baumgartner,*, Viatcheslav V. Jouikov,*, † and Christoph Marschner*, † Institut für Anorganische Chemie, Technische Universitaẗ Graz, Stremayrgasse 9, 8010 Graz, Austria ‡ Institut für Chemie, Universitaẗ Graz, Stremayrgasse 9, 8010 Graz, Austria § UMR 6226, Chimie et Photonique Moleculaires,́ Universitéde Rennes 1, 35042 Rennes, France *S Supporting Information ABSTRACT: Several silatranyl -substituted oligosilanes were prepared starting from bis(trimethylsilyl)silatranylsilanide. Electrochemical and theoretical investigations of some oligosilanes revealed that electrooxidation occurs by formation of a short-lived cation radical. This species undergoes structural relaxation to form a pair of conformers, with endo and exo relationships with respect to the Si−N interaction. Reaction of a 1,4-disilatranyl-1,4-disilanide with 1,2- dichlorotetramethyldisilane gave a mixture of cis and trans diastereomers of a cyclohexasilane with the trans isomer showing a diminished Si−N distance. ■ INTRODUCTION along the Si−N dative bond reflects the nature of Si−N − Among hypercoordinated silicon compounds silatranes (Figure bonding. In fact the Si N bonding neither is covalent nor is − 8 1) occupy a prominent position.1 4 The suffix “atrane” was based on intermolecular charge transfer. What happens with this unusual bond upon electron withdrawal, for instance during electrooxidation, is of a great interest (for classical bonds see ref 9) but is not known so far. -
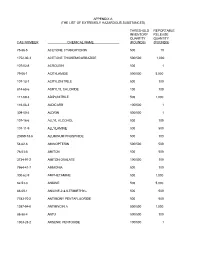
The List of Extremely Hazardous Substances)
APPENDIX A (THE LIST OF EXTREMELY HAZARDOUS SUBSTANCES) THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 75-86-5 ACETONE CYANOHYDRIN 500 10 1752-30-3 ACETONE THIOSEMICARBAZIDE 500/500 1,000 107-02-8 ACROLEIN 500 1 79-06-1 ACRYLAMIDE 500/500 5,000 107-13-1 ACRYLONITRILE 500 100 814-68-6 ACRYLYL CHLORIDE 100 100 111-69-3 ADIPONITRILE 500 1,000 116-06-3 ALDICARB 100/500 1 309-00-2 ALDRIN 500/500 1 107-18-6 ALLYL ALCOHOL 500 100 107-11-9 ALLYLAMINE 500 500 20859-73-8 ALUMINUM PHOSPHIDE 500 100 54-62-6 AMINOPTERIN 500/500 500 78-53-5 AMITON 500 500 3734-97-2 AMITON OXALATE 100/500 100 7664-41-7 AMMONIA 500 100 300-62-9 AMPHETAMINE 500 1,000 62-53-3 ANILINE 500 5,000 88-05-1 ANILINE,2,4,6-TRIMETHYL- 500 500 7783-70-2 ANTIMONY PENTAFLUORIDE 500 500 1397-94-0 ANTIMYCIN A 500/500 1,000 86-88-4 ANTU 500/500 100 1303-28-2 ARSENIC PENTOXIDE 100/500 1 THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 1327-53-3 ARSENOUS OXIDE 100/500 1 7784-34-1 ARSENOUS TRICHLORIDE 500 1 7784-42-1 ARSINE 100 100 2642-71-9 AZINPHOS-ETHYL 100/500 100 86-50-0 AZINPHOS-METHYL 10/500 1 98-87-3 BENZAL CHLORIDE 500 5,000 98-16-8 BENZENAMINE, 3-(TRIFLUOROMETHYL)- 500 500 100-14-1 BENZENE, 1-(CHLOROMETHYL)-4-NITRO- 500/500 500 98-05-5 BENZENEARSONIC ACID 10/500 10 3615-21-2 BENZIMIDAZOLE, 4,5-DICHLORO-2-(TRI- 500/500 500 FLUOROMETHYL)- 98-07-7 BENZOTRICHLORIDE 100 10 100-44-7 BENZYL CHLORIDE 500 100 140-29-4 BENZYL CYANIDE 500 500 15271-41-7 BICYCLO[2.2.1]HEPTANE-2-CARBONITRILE,5- -

Template for Electronic Submission to ACS Journals
Understanding the role of Ti-rich domains in the stabilization of gold nanoparticles on mesoporous silica-based catalysts Alaina Moragues a, Begoña Puértolas b, Álvaro Mayoral c,d, Raúl Arenal c,d, Ana B. Hungría e, Sonia Murcia-Mascarós a, Stuart H. Taylor f, Benjamín Solsona g,*, Tomás Garcíab,*, Pedro Amorós a,* a Institut de Ciència dels Materials, Universitat de València, P.O. Box 22085, 46071 Valencia, Spain. b Instituto de Carboquímica (ICB-CSIC), C/ Miguel Luesma Castán 4, 50018 Zaragoza, Spain. c Laboratorio de Microscopias Avanzadas (LMA), Instituto de Nanociencia de Aragon (INA), Universidad de Zaragoza, Mariano Esquillor, 50018 Zaragoza, Spain. d Fundación Agencia Aragonesa para la Investigación y el Desarrollo (ARAID), María de Luna 11, 50018 Zaragoza, Spain. e Departamento de Ciencia de Materiales, Ingeniería Metalúrgica y Química Inorgánica, Universidad de Cádiz, Campus Río San Pedro, 11510 Puerto Real, Spain. f Cardiff Catalysis Institute, School of Chemistry, Cardiff University, Main Building, Park Place, CF10 3AT Cardiff, United Kingdom. g Departamento de Ingenieria Quimica, Universitat de València, Avenida de la Universitat, 46071 Valencia, Spain. *Corresponding authors at: Institut de Ciència dels Materials, Universitat de València, P.O. Box 22085, 46071 Valencia, Spain (Pedro Amorós). E-mail addresses: [email protected], [email protected], [email protected] 1 ABSTRACT The preparation and stabilization of gold nanoparticles with a precise control of size and dispersion is highly attractive for a variety of applications, and a key aspect is thermal stability of the nanoparticles. This paper focuses on understanding the effect of TiO2-based nanodomains, dispersed on mesoporous silicas, and how they control gold nanoparticle stability. -

Oxatrane Is a Parent Compound of a New Atrane Family: Crystal and Molecular Structure of Triethanolamine N�Oxide Academician M
ISSN 00125008, Doklady Chemistry, 2014, Vol. 458, Part 1, pp. 172–175. © Pleiades Publishing, Ltd., 2014. Original Russian Text © M.G. Voronkov, E.A. Zel’bst, A.D. Vasil’ev, M.S. Molokeev, G.A. Kuznetsova, 2014, published in Doklady Akademii Nauk, 2014, Vol. 458, No. 3, pp. 287–290. CHEMISTRY Oxatrane Is a Parent Compound of a New Atrane Family: Crystal and Molecular Structure of Triethanolamine NOxide Academician M. G. Voronkova †, E. A. Zel’bstb, A. D. Vasil’evc,d, M. S. Molokeevc,d, and G. A. Kuznetsovaa Received March 27, 2014 DOI: 10.1134/S0012500814090031 Tertiary amine Noxides of general formula A particular interest in tertiary amine Noxides has + increased in recent years. Triethanolamine Noxide, O ⎯ NR3 (previously O=NR3 or O → NR3) continue to attract interest of synthetic, theoretical, and applied which was considered to have structure chemists. They are already used in cosmetics, as bio O=N(CH2CH2OH)3 [3], was ascribed to them by medical agents, and as oxidants in a number of organic analogy, but its molecular structure was not estab reactions (catalytic epoxidation and dihydroxylation lished. Therefore, we determined the structure by X of alkenes, catalytic oxidation of alcohols, etc.) [1, 2]. ray diffraction, presuming that the oxide oxygen atom The most general method of their synthesis is the oxi in this molecule is bound to the nitrogen atom and sur dation of tertiary amines with hydrogen peroxide, per rounded by three CH CH OH groups like hydrogen oxy acids, dioxiranes, and oxaazapyridines [1]. 2 2 atom in protatranes [4]. The obtained data proved to Triethanolamine Noxide (TEA) formulated as be unexpected and, nonetheless, allowed us to name + O –N (CH2CH2OH)3 and obtained by the oxidation TEA Noxide as oxatrane (OA) by analogy with pro of triethanolamine with hydrogen peroxide [3] is also tatranes, Fig. -
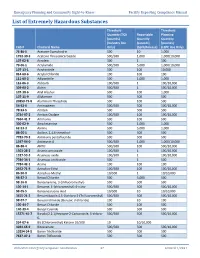
List of Extremely Hazardous Substances
Emergency Planning and Community Right-to-Know Facility Reporting Compliance Manual List of Extremely Hazardous Substances Threshold Threshold Quantity (TQ) Reportable Planning (pounds) Quantity Quantity (Industry Use (pounds) (pounds) CAS # Chemical Name Only) (Spill/Release) (LEPC Use Only) 75-86-5 Acetone Cyanohydrin 500 10 1,000 1752-30-3 Acetone Thiosemicarbazide 500/500 1,000 1,000/10,000 107-02-8 Acrolein 500 1 500 79-06-1 Acrylamide 500/500 5,000 1,000/10,000 107-13-1 Acrylonitrile 500 100 10,000 814-68-6 Acrylyl Chloride 100 100 100 111-69-3 Adiponitrile 500 1,000 1,000 116-06-3 Aldicarb 100/500 1 100/10,000 309-00-2 Aldrin 500/500 1 500/10,000 107-18-6 Allyl Alcohol 500 100 1,000 107-11-9 Allylamine 500 500 500 20859-73-8 Aluminum Phosphide 500 100 500 54-62-6 Aminopterin 500/500 500 500/10,000 78-53-5 Amiton 500 500 500 3734-97-2 Amiton Oxalate 100/500 100 100/10,000 7664-41-7 Ammonia 500 100 500 300-62-9 Amphetamine 500 1,000 1,000 62-53-3 Aniline 500 5,000 1,000 88-05-1 Aniline, 2,4,6-trimethyl- 500 500 500 7783-70-2 Antimony pentafluoride 500 500 500 1397-94-0 Antimycin A 500/500 1,000 1,000/10,000 86-88-4 ANTU 500/500 100 500/10,000 1303-28-2 Arsenic pentoxide 100/500 1 100/10,000 1327-53-3 Arsenous oxide 100/500 1 100/10,000 7784-34-1 Arsenous trichloride 500 1 500 7784-42-1 Arsine 100 100 100 2642-71-9 Azinphos-Ethyl 100/500 100 100/10,000 86-50-0 Azinphos-Methyl 10/500 1 10/10,000 98-87-3 Benzal Chloride 500 5,000 500 98-16-8 Benzenamine, 3-(trifluoromethyl)- 500 500 500 100-14-1 Benzene, 1-(chloromethyl)-4-nitro- 500/500 -

40 CFR Ch. I (7–1–11 Edition) Pt. 355, App. B
Pt. 355, App. B 40 CFR Ch. I (7–1–11 Edition) [Alphabetical Order] Reportable Threshold plan- CAS No. Chemical name Notes quantity * ning quantity (pounds) (pounds) 7550–45–0 ............ Titanium Tetrachloride ................................................ ..................... 1,000 100 584–84–9 .............. Toluene 2,4-Diisocyanate ........................................... ..................... 100 500 91–08–7 ................ Toluene 2,6-Diisocyanate ........................................... ..................... 100 100 110–57–6 .............. Trans-1,4-Dichlorobutene ........................................... ..................... 500 500 1031–47–6 ............ Triamiphos .................................................................. ..................... 500 500/10,000 24017–47–8 .......... Triazofos ..................................................................... ..................... 500 500 76–02–8 ................ Trichloroacetyl Chloride .............................................. ..................... 500 500 115–21–9 .............. Trichloroethylsilane ..................................................... d .................. 500 500 327–98–0 .............. Trichloronate ............................................................... e .................. 500 500 98–13–5 ................ Trichlorophenylsilane .................................................. d .................. 500 500 1558–25–4 ............ Trichloro(Chloromethyl)Silane .................................... ..................... 100 -

Synthesis, Structure Features and Application of Pro-Azaphsphatranes As Catalysts and Strong Non-Ionic Bases in Organic Synthesis " (1994)
Iowa State University Capstones, Theses and Retrospective Theses and Dissertations Dissertations 1994 Synthesis, structure features and application of pro- azaphsphatranes as catalysts and strong non-ionic bases in organic synthesis Jiansheng Tang Iowa State University Follow this and additional works at: https://lib.dr.iastate.edu/rtd Part of the Medicinal and Pharmaceutical Chemistry Commons, Medicinal Chemistry and Pharmaceutics Commons, Medicinal-Pharmaceutical Chemistry Commons, Organic Chemistry Commons, and the Polymer Chemistry Commons Recommended Citation Tang, Jiansheng, "Synthesis, structure features and application of pro-azaphsphatranes as catalysts and strong non-ionic bases in organic synthesis " (1994). Retrospective Theses and Dissertations. 10651. https://lib.dr.iastate.edu/rtd/10651 This Dissertation is brought to you for free and open access by the Iowa State University Capstones, Theses and Dissertations at Iowa State University Digital Repository. It has been accepted for inclusion in Retrospective Theses and Dissertations by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. INFORMATION TO USERS This manuscript has been reproduced from the microfilm master. UMI films the text directly from the original or copy submitted. Thus, some thesis and dissertation copies are in typewriter face, while others may be from any type of computer printer. The quality of this reproduction is dependent upon the quality of the copy submitted. Broken or indistinct print, colored or poor quality illustrations and photographs, print bleedthrough, substandard margins, and improper alignment can adversely affect reproduction. In the unlikely event that the author did not send UMI a complete manuscript and there are missing pages, these will be noted. -

Environmental Protection Agency Pt. 355, App. A
Environmental Protection Agency Pt. 355, App. A agreement between a State and a sion over which the United States has Tribe, the SERC shall be the entity jurisdiction and Indian Country. identified in the agreement. Threshold planning quantity means, State means any State of the United for a substance listed in Appendices A States, the District of Columbia, the and B of this part, the quantity listed Commonwealth of Puerto Rico, Guam, in the column ‘‘threshold planning American Samoa, the United States quantity’’ for that substance. Virgin Islands, the Northern Mariana [73 FR 65462, Nov. 3, 2008, as amended at 73 Islands, any other territory or posses- FR 76960, Dec. 18, 2008] APPENDIX A TO PART 355—THE LIST OF EXTREMELY HAZARDOUS SUBSTANCES AND THEIR THRESHOLD PLANNING QUANTITIES [Alphabetical Order] Reportable Threshold plan- CAS No. Chemical name Notes quantity * ning quantity (pounds) (pounds) 75–86–5 ................ Acetone Cyanohydrin ................................................. ..................... 10 1,000 1752–30–3 ............ Acetone Thiosemicarbazide ....................................... ..................... 1,000 1,000/10,000 107–02–8 .............. Acrolein ....................................................................... ..................... 1 500 79–06–1 ................ Acrylamide .................................................................. f ................... 5,000 1,000/10,000 107–13–1 .............. Acrylonitrile ................................................................. f .................. -

Environmental Protection Agency Pt. 355, App. B
Environmental Protection Agency Pt. 355, App. B [Alphabetical Order] Reportable Threshold plan- CAS No. Chemical name Notes quantity * ning quantity (pounds) (pounds) 5344–82–1 ............ Thiourea, (2-Chlorophenyl)- ....................................... ..................... 100 100/10,000 614–78–8 .............. Thiourea, (2-Methylphenyl)- ....................................... ..................... 500 500/10,000 7550–45–0 ............ Titanium Tetrachloride ................................................ ..................... 1,000 100 584–84–9 .............. Toluene 2,4-Diisocyanate ........................................... ..................... 100 500 91–08–7 ................ Toluene 2,6-Diisocyanate ........................................... ..................... 100 100 110–57–6 .............. Trans-1,4-Dichlorobutene ........................................... ..................... 500 500 1031–47–6 ............ Triamiphos .................................................................. ..................... 500 500/10,000 24017–47–8 .......... Triazofos ..................................................................... ..................... 500 500 76–02–8 ................ Trichloroacetyl Chloride .............................................. ..................... 500 500 115–21–9 .............. Trichloroethylsilane ..................................................... d .................. 500 500 327–98–0 .............. Trichloronate ............................................................... e ................. -

Environmental Protection Agency Pt. 355, App. A
Environmental Protection Agency Pt. 355, App. A Release means any spilling, leaking, the facility is located. In the absence pumping, pouring, emitting, emptying, of a SERC for a State or Indian Tribe, discharging, injecting, escaping, leach- the Governor or the chief executive of- ing, dumping, or disposing into the en- ficer of the tribe, respectively, shall be vironment (including the abandonment the SERC. Where there is a cooperative or discarding of barrels, containers, agreement between a State and a and other closed receptacles) of any Tribe, the SERC shall be the entity hazardous chemical, EHS, or CERCLA identified in the agreement. hazardous substance. Solution means any aqueous or or- Reportable quantity means, for any ganic solutions, slurries, viscous solu- CERCLA hazardous substance, the tions, suspensions, emulsions, or quantity established in Table 302.4 of 40 pastes. CFR 302.4, for such substance. For any State means any State of the United EHS, reportable quantity means the States, the District of Columbia, the quantity established in Appendices A Commonwealth of Puerto Rico, Guam, and B of this part for such substance. American Samoa, the United States Unless and until superseded by regula- Virgin Islands, the Northern Mariana tions establishing a reportable quan- Islands, any other territory or posses- tity for newly listed EHSs or CERCLA sion over which the United States has hazardous substances, a weight of 1 jurisdiction and Indian Country. pound shall be the reportable quantity. Threshold planning quantity means, SERC means the State Emergency for a substance listed in Appendices A Response Commission for the State in and B of this part, the quantity listed which the facility is located except in the column ‘‘threshold planning where the facility is located in Indian quantity’’ for that substance. -

A New Class of Silatranes: Structure and Dynamic NMR Behavior1
1066 J. Am. Chem. Soc. 2000, 122, 1066-1072 A New Class of Silatranes: Structure and Dynamic NMR Behavior1 A. Chandrasekaran, Roberta O. Day, and Robert R. Holmes* Contribution from the Department of Chemistry, LGRT 701, Box 34510, UniVersity of Massachusetts, Amherst, Massachusetts 01003-4510 ReceiVed July 26, 1999 Abstract: A new class of silatranes N[CH2(Me2C6H2)O]3SiR was prepared by the reaction of organosilanes with tris(2-hydroxy-4,6-dimethylbenzyl) amine (5) in the case of 1, 3, and 4, and by the reaction of Si(OMe)4 with the same amine (5) to give 2. This is the first series of silatranes containing all six-membered rings. Their structures were established by X-ray analysis and correlated with 29Si NMR data. VT 1H NMR spectra reflected fluxional behavior for the chiral molecules 1-3, whereas 4 was a rigid molecule. The activation energies for enantiomeric conversion of the clockwise and anticlockwise orientations of the propeller-like silatranes 1-3 were statistically the same at about 10 kcal/mol. A simultaneous pseudorotation of all three six-membered rings is suggested to cause the racemization process. The Si-N distances for 1-4 show a large dependence on the exocyclic substituent. Over an approximate electronegativity range of 2.3-3.0, the Si-N distance varies from 2.745(4) Å for 1 to 2.025(4) Å for 4. Thus, the influence of substituent effects on nitrogen-silicon donor-acceptor interactions can be readily studied in the absence of constraints imposed by the use of the smaller ring-containing silatranes.